Friday, January 26, 2018
Printer Friendly Version in PDF Format (9 PDF pages)
Research Progress on Alzheimer's Disease and Related Dementias
Richard J. Hodes, M.D.
Director
National Institute on Aging
Research Implementation Milestones: Our Central Driver
| 2011 | 2012 AD Summit |
2013 ADRD Summit |
2015 AD Summit |
2016 ADRD Summit |
2017 AD/ADRD Care/Services Summit |
2018 AD Summit |
2019 AD/RD Summit |
|---|---|---|---|---|---|---|---|
| The National Alzheimer's Project Act (NAPA) was signed into law in January -- updated annually |
Recent Investments Allow Us to Accelerate Milestones - ADBB Timing and Appropriations
| FY17 AD/ADRD milestones -- $323M extra needed | FY17 ADBB Released July 2015 | $350M in FY16 supports acceleration of FY17 milestones |
|---|---|---|
| FY18 AD/ADRD milestones - $414M extra needed | FY18 ADBB Released August 2016 | $400M in FY17 supports acceleration of FY18 milestones |
| FY19 AD/ADRD milestones - $597M extra needed | FY19 ADBB Released July 2017 | FY18 TBD: 40% cut (PB) or +$400M (House/ Senate)? |
What Counts as AD/ADRD Research?
- The AD/ADRD pay line applies to applications/awards that are coded as AD or Alzheimer's disease - related dementias (ADRD)
- The ADRD RCDC categories that report related dementias specifically named in the National Plan to Address Alzheimer's Disease are:
- Lewy Body dementia (LBD)
- Frontotemporal dementia (FTD)
- Vascular Cognitive Impairment/Dementia (VCI/D)
Diversity of AD/ADRD Research
- Alzheimer's Research
- Cognitive outcomes in Population Studies
- Research on Disease Mechanisms
- Geroscience
- Aging metabolic changes in AD
- Comparative biology of neurodegeneration
- Basic Biological Processes of AD
- Biomarkers
- Research on Care and Caregiver Support
- Disparities, Sex differences, and AD risk
AD/ADRD FOAs by Fiscal Year
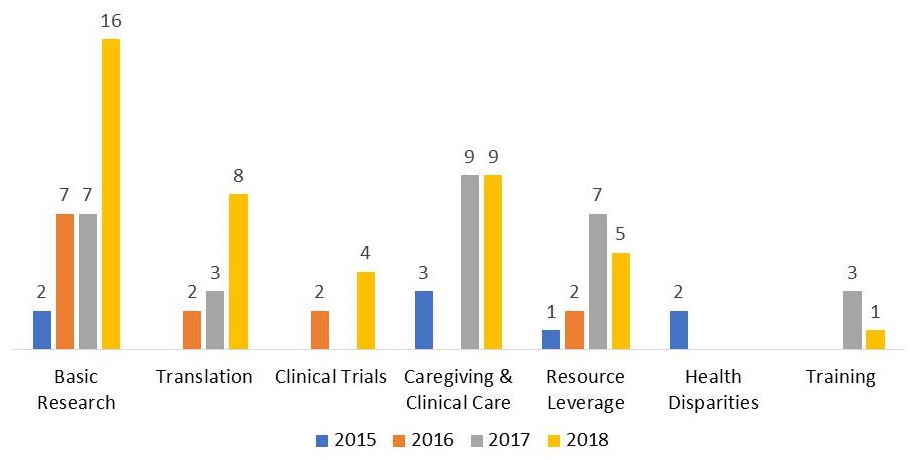 |
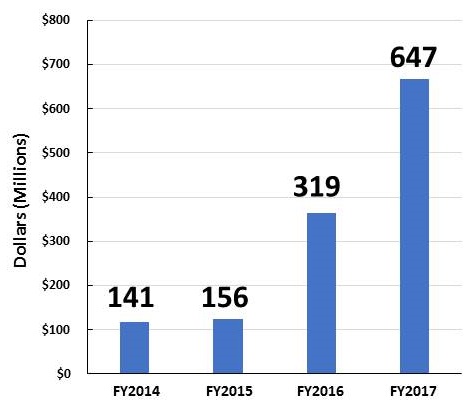
AD and ADRD Research Investments FY14-FY17
| Individual Research Grants | Clinical Trials |
|---|---|
 |
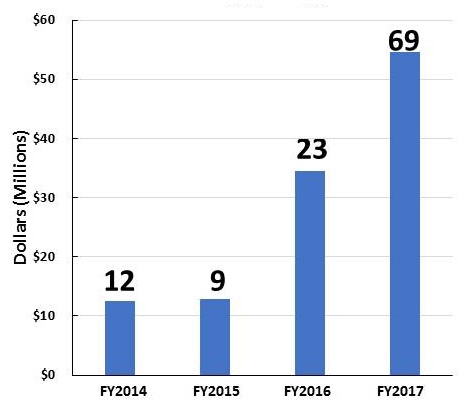 |
| These figures show grants awarded with NIA funding. Some of the grants are in response to initiatives sponsored by NIH ICs other than NIA (e.g., NINDS, NIBIB, NIEHS, NIDCD). | |
Translational Programs and Infrastructure for AD and ADRD
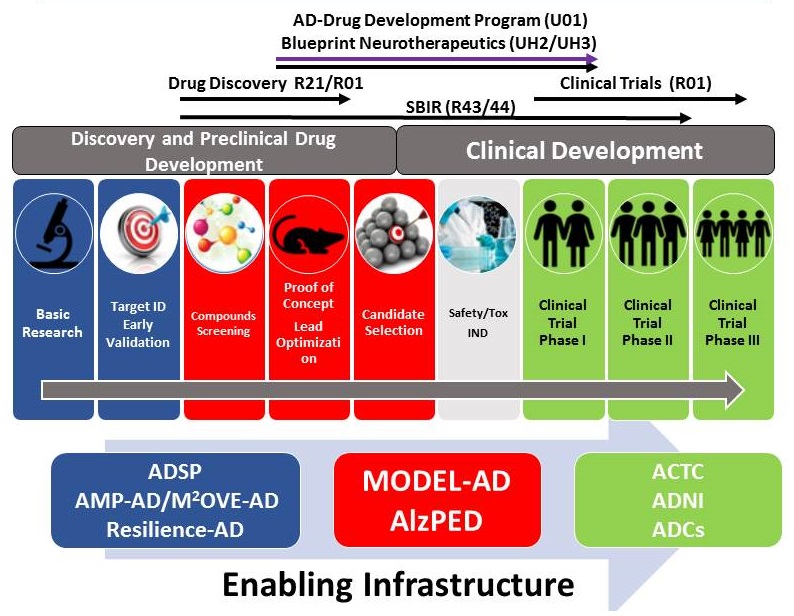 |
ENABLING INFRASTRUCTURE FOR DATA DRIVEN AND PREDICTIVE DRUG DEVELOPMENT
- AMP-AD, M2OVE-AD, Resilience-AD
- Large scale systems/network biology approach
- Predictive models for novel targets and biomarkers
- Computational methods benchmarking
- Open Data
- MODEL-AD, AlzPED
- Next-gen animal models for late onset AD
- Deep phenotyping and staging relative to human disease
- Methods development for efficacy testing/Transparent reporting
- Open data and models distribution free of IP barriers
- ACTC
- Clinical trials infrastructure (Phase I, II, III)
- Methods development for clinical trial design
- New methods for recruitment and retention (emphasis on diversity)
Ongoing NIA AD/ADRD and Related Intervention and Prevention Trials
- Over 140 active trials
- 40 Early-stage Clinical Drug Development (Phase I and Phase II Clinical Trials)
- Amyloid (9)
- Neurotransmitter Receptors (3)
- Metabolism and Bioenergetics (4)
- Vasculature (3)
- Growth Factors and Hormones (1)
- Multi-target (6)
- Oxidative Stress (1)
- 8 Late-stage Clinical Drug Development (Phase II/III and Phase III Clinical Trials)
- Amyloid (6)
- Vasculature (2)
- 62 Non-Pharmacological Interventions
- Exercise (16)
- Diet (2)
- Cognitive Training (20)
- Combination Therapy (11)
- 7 Clinical Therapy Development for the Neuropsychiatric Symptoms of AD/ADRD
- Pharmacological (5)
- Non-Pharmacological (2)
- 37 Care and Caregiver Interventions
Specific Milestone-Directed Achievements
Milestone 6.D: Identify, characterize, and complete early validation for at least six novel therapeutic targets for AD (a minimum of three targets for presymptomatic and early-stage disease and a minimum of three for advanced disease). These efforts should include therapeutic targets for the neuropsychiatric and behavioral disturbances in AD. Validation based on availability of the following for each novel target: a systems-level understanding of the gene, protein, and metabolic networks within which they operate, one or more cell-based/animal models that are freely available to the research community, a quantitative assessment of the integrative response to the modulation of the target in one or more model organisms, and identification of pharmacodynamic biomarker(s) for target engagement. [2012 AD Summit: 1A, 1B, 1D, and 5A]
| 2012 | 2013 | 2014 | 2015 | 2018 |
|---|---|---|---|---|
| RFA AG 13-013 Interdisciplinary Approach to Identification and Validation of Novel Therapeutic Targets for Alzheimer's Disease (R01) https://grants.nih.gov/grants/guide/rfa-files/RFA-AG-13-013.html | 4 R01 projects funded and converted into U01 mechanism | 2 additional R01s funded and incorporated into AMP-AD Target Discovery and Preclinical Validation Project/ Consortium | AMP-AD Knowledge Portal launched as a platform for sharing multi-omics data and analytical results/network models https://www.synapse.org/#!Synapse:syn2580853/wiki/409840 | Data generation completed, data and network models made available to researchers at large |
Milestone 6.F: Initiate first in human Phase I drug trials for therapeutic agents against at least six novel therapeutic targets. In addition to safety, these trials will provide evidence of target engagement. [2012 AD Summit: 3A, 3B, 3F, and 5E]
Milestone 6.G: Initiate Phase ll (proof-of-concept) drug trials for agents against three to six novel therapeutic targets. These trials will provide proof of mechanism and/or evidence of target engagement of the target being tested. [2012 AD Summit: 3A, 3B, 3F, and 5E]
| 2012 | 2014 | 2015 | 2018 |
|---|---|---|---|
| RFA AG 13-016 Alzheimer's Disease Phase I Clinical Trials (R01) https://grants.nih.gov/grants/guide/rfa-files/RFA-AG-13-016.html | Funded a Phase I Trial to test the safety of Allopregnanolone as a novel neuroregenerative therapeutic for AD | PAR 16-365 Pilot Clinical Trials for the Spectrum of Alzheimer's Disease and Age-related Cognitive Decline (R01) https://grants.nih.gov/grants/guide/pa-files/PAR-16-365.html
Funded a Phase I Trial to test the safety of a first in class PDE4 allosteric modulator as a novel therapeutic for MCI |
Funded a Phase II Trial to test the efficacy of a first in class P75NTR ligand as a novel neuroprotective AD therapeutic |
LM11A-31 - first in class, small molecule modulator of the P75 Neurotrophin Receptor
PI -- Frank Longo MD and NeurotrophiX
The pre-clinical drug development and part of the IND-enabling studies for LM11A-31 were supported through NIA's AD Translational Research Program
The Phase II trial is being supported through NIA's Pilot Clinical Trials Program via PAR 16-365 https://grants.nih.gov/grants/guide/pa-files/PAR-16-365.html
Opportunities for Small Business -- AD/ADRD specific
- Advancing Research on Alzheimer's Disease (AD) and Alzheimer's-Disease-Related Dementias (ADRD) (R41/R42/R43/R44)
- Tools for Clinical Care and Management of Alzheimer's Disease (AD) and its Comorbidities (R41/R42/R43/R44)
- Development of Socially-Assistive Robots (SARs) to Engage Persons with Alzheimer's Disease (AD) and AD-Related Dementias (ADRD), and their Caregivers (R41/R42/R43/R44)
NIA SBIR/STTR Obligations
| $ in Millions |
|---|
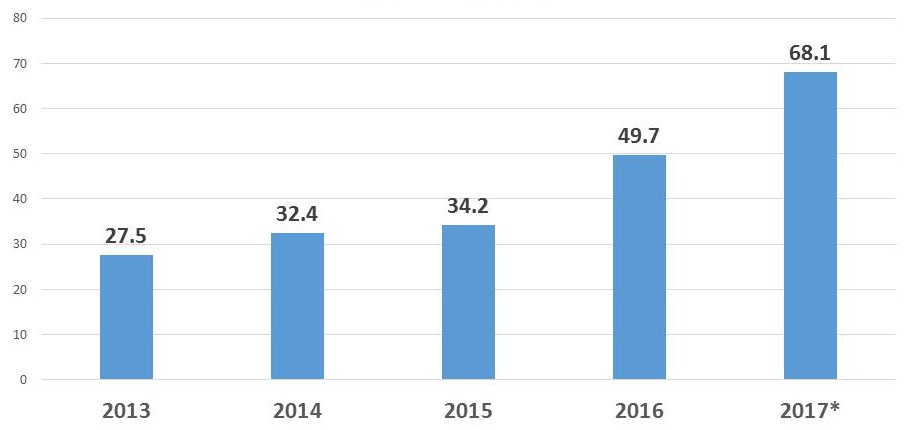 |
| * Estimate |
NIH AD Research Summit - March 1-2, 2018: Path to Treatment and Prevention
The program agenda is organized around seven sessions:
- Novel Mechanistic Insights into the Complex Biology and Heterogeneity of AD
- Enabling Precision Medicine for AD
- Translational Tools and Infrastructure to Enable Predictive Drug Development
- Emerging Therapeutics
- Understanding the Impact of the Environment to Advance Disease Prevention
- Advances in Disease Monitoring, Assessment and Care
- Building an Open Science Research Ecosystem to Accelerate AD Therapy Development
https://www.nia.nih.gov/research/nih-ad-summit-2018-program-agenda#agenda
FY2018 NINDS ADRD Funding Opportunity Announcements
- PAR-18-413 Mechanistic Basis of Diffuse White Matter Disease and Small Vessel Pathology in Vascular Contributions to Cognitive Impairment and Dementia (VCID)(R01) https://grants.nih.gov/grants/guide/pa-files/PAR-18-413.html
- To support hypothesis-testing research to elucidate cellular and molecular mechanisms that underlie diffuse white matter disease and small vessel disease in the brain, the relationships between them, and their contributions to cognitive impairment and dementia
- Application are due March 23, 2018
- RFA-NS-18-017 Planning Grant to Develop Phase III Clinical Trials for Lewy Body Dementia (R34) https://grants.nih.gov/grants/guide/rfa-files/RFA-NS-18-017.html
- To provide one year of support for planning activities necessary for initiating a Phase III clinical trial designed to treat patients with LBD
- Applications are due March 20, 2018
- NINDS is planning to release 4 additional new ADRD FOAs for FY18
- See the approved research concepts in the NINDS Director's Message on the NINDS website! https://www.ninds.nih.gov/News-Events/Directors-Messages/All-Directors-Messages/Boosting-Research-Alzheimers-Disease
| Save the date! |
|---|
| ADRD Summit (III) March 14-15, 2019 Location: Natcher on the NIH Main Campus, Bethesda MD |
January 26, 2018 -- Advisory Council Meeting #27
The meeting was held on Friday, January 26, 2018, in Washington, DC. The Research Subcommittee took charge of this meeting's theme, focusing on the process from targets to treatments. The Council heard speakers on the preclinical pipeline, the clinical trial pipeline, and the industry perspective. The meeting also included discussion of a driver diagram to guide the Council's future work, updates and a report from the October Care Summit, and federal workgroup updates. Material available from this meeting is listed below and at https://aspe.hhs.gov/advisory-council-alzheimers-research-care-and-services-meetings#Jan2018.
Comments and questions, or alerts to broken links, should be sent to napa@hhs.gov.
General Information
-
Agenda -- [HTML Version] [PDF Version]
-
Meeting Announcement -- [HTML Version] [PDF Version]
-
Meeting Summary -- [HTML Version] [PDF Version]
-
Public Comments -- [HTML Version]
Handouts
-
Care Summit Report Themes -- [PDF Version]
-
NAPA Driver Diagram Draft Examples -- [PDF Version]
-
Outline for Care Summit Final Report -- [PDF Version]
Presentation Slides
-
AbbVie's R&D Vision for Alzheimer's Disease -- [HTML Version] [PDF Version]
-
Care Summit Report -- [HTML Version] [PDF Version]
-
Clinical Subcommittee Update -- [HTML Version] [PDF Version]
-
Initiatives, Partnerships and Collaboration to Help Patients with the Highest Unmet Need: Dominantly Inherited Alzheimer's Disease Trials Unit (DIAN-TU) as a Case Example -- [HTML Version] [PDF Version]
-
Long-Term Services and Supports Committee Update -- [HTML Version] [PDF Version]
-
NAPA Driver Diagram -- [HTML Version] [PDF Version]
-
Overview of the Clinical Trial Pipeline for AD -- [HTML Version] [PDF Version]
-
Overview on NIA Preclinical Pipeline -- [HTML Version] [PDF Version]
-
Participating in an Alzheimer's Clinical Study: Perspectives on Involvement of a Person Living with Dementia and Her Study Partner -- [HTML Version] [PDF Version]
-
Progress Since October -- [HTML Version] [PDF Version]
-
Research Progress on Alzheimer's Disease and Related Dementias -- [HTML Version] [PDF Version]
-
Research Subcommittee Agenda: The Journey from Targets to Treatments -- [HTML Version] [PDF Version]
