Friday, January 26, 2018
Printer Friendly Version in PDF Format (12 PDF pages)
AbbVie's R&D Vision for Alzheimer's disease
Eric Karran, Ph.D.
AbbVie R&D Strategy -- science and innovation applied to high unmet medical need
- Unmet Medical Need
- Novel Biology
- New Technologies
- Coe Capabilities
- Clinical Translation
Alzheimer's Disease is an Emerging Global Crisis
- 115 million AD patients by 2050
- Cost of care in the U.S. was $259 billion in 2017; will be $1.1 trillion by 2050
- Therapeutic options for AD are limited; progress lags well behind successes in oncology, inflammation, metabolic diseases and cardiology
Compelling reasons for AbbVie to focus research efforts in neurodegenerative diseases
- Science
- Emergence of compelling approaches beyond amyloid beta peptide.
- Biomarkers of Disease
- New technologies and techniques emerging to identify the right patients and assess pathology in living patients.
- Clinical Trials
- Ability to identify and intervene earlier in the disease process, at very mild symptomatic stage or even before cognitive symptoms emerge.
- Regulatory Environment Engaged regulatory bodies are open to innovative approaches and actively discussing path to approval
Foundational Neuroscience Center -- new investment
- 200 Sidney Street, Cambridge MA
- Officially opened May 18th 2016
- 43,000 sq. ft.
- 50 life scientists
AbbVie's Global AD Research
- Cambridge
- Tau
- Proteostasis
- Neuro-inflammation
- Ludwigshafen
- Tau
- Proteostasis
- Neuro-inflammation
- Lake County
- Tau
- Proteostasis
A personal view - how to translate basic science into patient benefit.
- Access to excellent scientists and fundamental scientific research into disease mechanisms
- Access to clinicians who know the disease and treat patients
- Access to patients and patient material (genome, fluids, samples, biomarkers, brain banking)
- In my experience, 4 conditions are pre-requisites for drug discovery success
- Hire (access) talented people and treat them well
- Provide well equipped labs and sufficient budget
- Set a clear mission
- Give it time
- And finally........high quality science and data drives everything
Drug targets -- what are they and where do they come from?
Genetics has informed AD research -- but how much genetic risk is associated with disease onset vs disease progression?
 |
Drug targets are sometimes lost in translation from academia to pharma
- As a broad generalization, a pharma scientist's drug target:
- Can be traced to pathways with genetic evidence
- Has druggable sites or activities
- Has an evident polarity of modulation
- Is without obvious side effects or toxicity liabilities
- Has a favourable tissue distribution
- Is preferably not a member of a gene superfamily where selectivity will be impossible
- AMP-AD has been an excellent vehicle to bring together academic and pharma scientists to educate each other.
Choosing the right target is critical
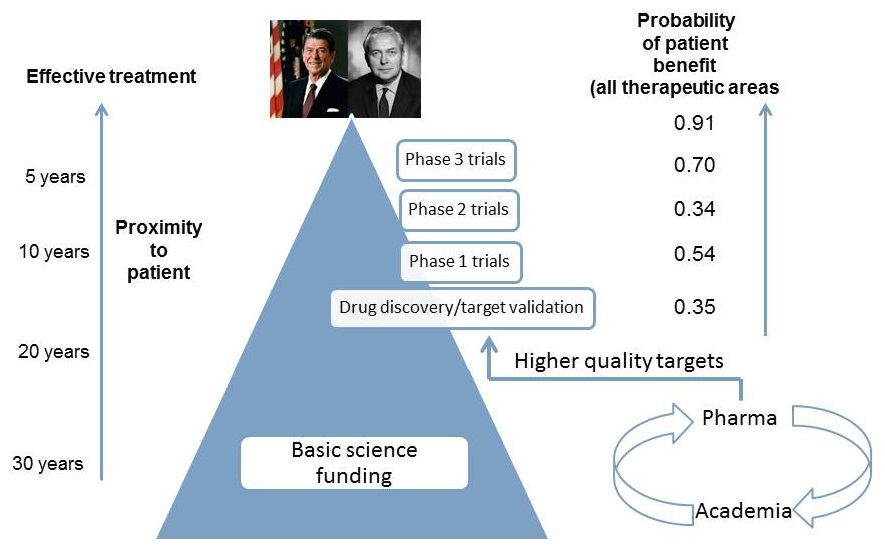 |
Funding landscape.. From the NIH website
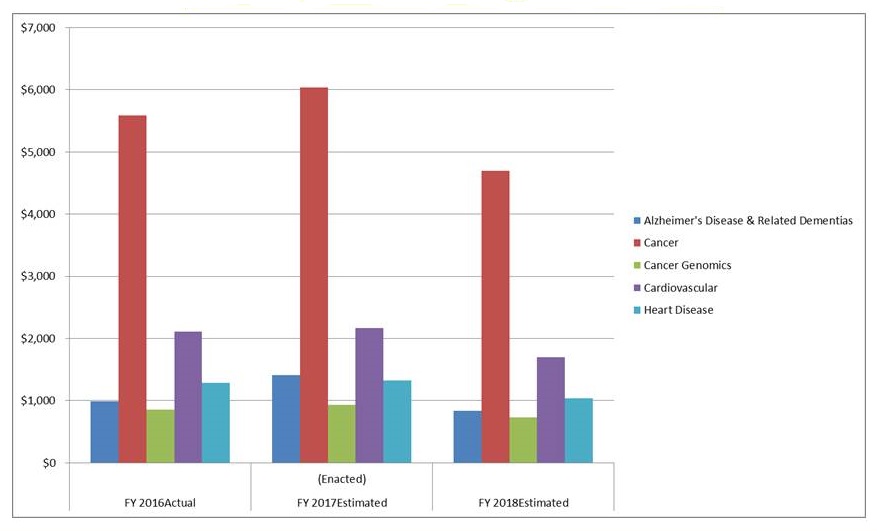 |
| https://report.nih.gov/categorical_spending.aspx |
2017 46 FDA Approvals
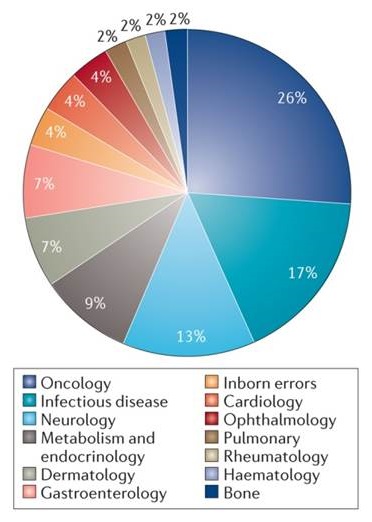 |
| Nature Reviews | Drug Discovery |
In vivo models of AD -- is it true that they are not fit for purpose?
George Box and H. L. Mencken
"The most that can be expected from any model is that it can supply a useful approximation to reality: all models are wrong; some models are useful".
~George Box
"To every complex problem there is an answer that is clear, simple and wrong".
~H.L. Mencken
Mice and humans are different
 |
What does pharma use in vivo models for?
- Proof of pharmacology/mechanism
- Biomarker/pharmacodynamic marker discovery
- Assessment of safety vs efficacy of therapeutics
- Projecting doses to man
- Target 'validation'/hypothesis testing
- Sometimes, but rarely, used to understand disease processes de novo.
Does pharma need more in vivo models?
- Would it be useful to have a model of Alzheimer's disease in mice?
- Yes
- Is that a realistic objective?
- No
- Do we need additional in vivo systems that model different aspects of neurodegenerative disease?
- Yes
- Is that a realistic objective?
- Yes
- Mechanisms of neuronal death
- Relationship between synaptic & neuronal dysfunction, neuronal death, and animal functional decrement (in a disease relevant manner)
- Differential neuronal selectivity to pathology
- Yes
Clinical development
Why have there been multiple failures in clinical development?
AD Disease Modifying Clinical Pipeline
- Amyloid plaques and tau pathology (leading to cognitive impairment) define Alzheimer's disease.
- There is no correlation between amyloid and cognitive impairment.
- Tau pathology correlates well with cognitive impairment.
- We all hope one of these agents will work...
- But 'hope' should not replace science.
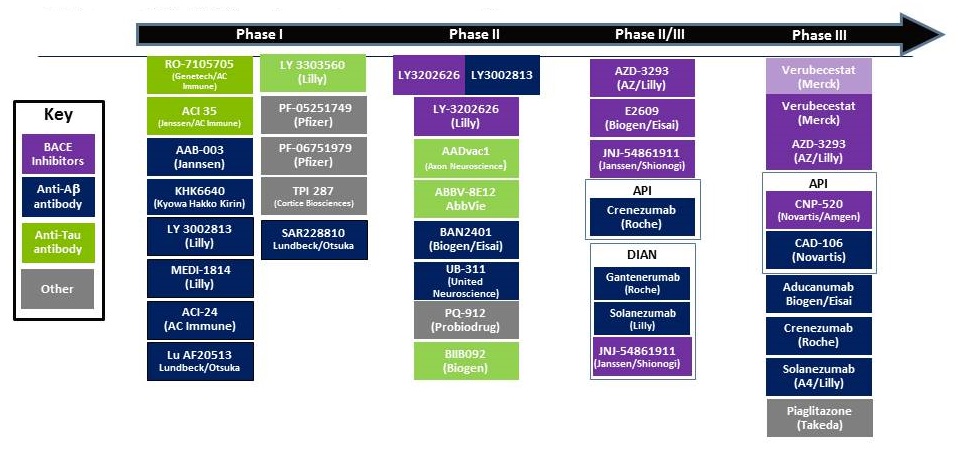 |
The Jack model & clinical development
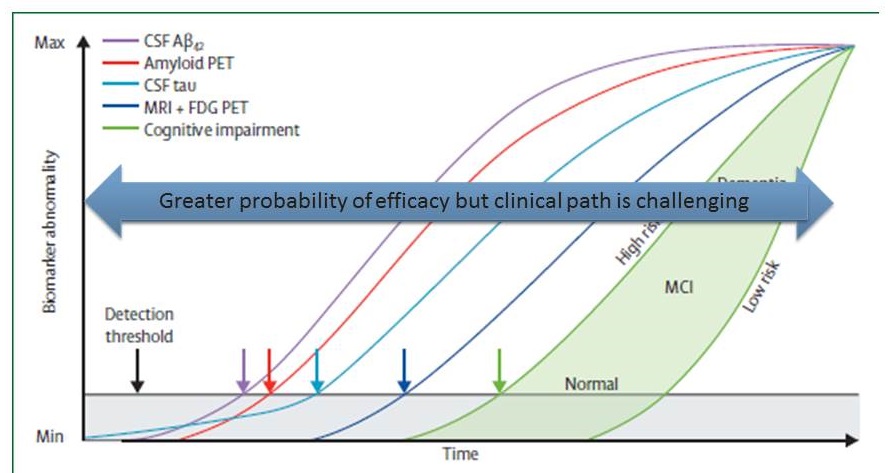 |
Conclusions
- A number of highly successful public-private consortia have been established that have advanced our understanding of AD significantly.
- Sustaining and increasing the interaction of all stake-holders --funders, academics, advocacy groups, patients, regulators and pharma -- will increase the probability that we shall succeed.
- The current degree of collaboration and sense of a shared mission is unprecedented in my experience.
January 26, 2018 -- Advisory Council Meeting #27
The meeting was held on Friday, January 26, 2018, in Washington, DC. The Research Subcommittee took charge of this meeting's theme, focusing on the process from targets to treatments. The Council heard speakers on the preclinical pipeline, the clinical trial pipeline, and the industry perspective. The meeting also included discussion of a driver diagram to guide the Council's future work, updates and a report from the October Care Summit, and federal workgroup updates. Material available from this meeting is listed below and at https://aspe.hhs.gov/advisory-council-alzheimers-research-care-and-services-meetings#Jan2018.
Comments and questions, or alerts to broken links, should be sent to napa@hhs.gov.
General Information
-
Agenda -- [HTML Version] [PDF Version]
-
Meeting Announcement -- [HTML Version] [PDF Version]
-
Meeting Summary -- [HTML Version] [PDF Version]
-
Public Comments -- [HTML Version]
Handouts
-
Care Summit Report Themes -- [PDF Version]
-
NAPA Driver Diagram Draft Examples -- [PDF Version]
-
Outline for Care Summit Final Report -- [PDF Version]
Presentation Slides
-
AbbVie's R&D Vision for Alzheimer's Disease -- [HTML Version] [PDF Version]
-
Care Summit Report -- [HTML Version] [PDF Version]
-
Clinical Subcommittee Update -- [HTML Version] [PDF Version]
-
Initiatives, Partnerships and Collaboration to Help Patients with the Highest Unmet Need: Dominantly Inherited Alzheimer's Disease Trials Unit (DIAN-TU) as a Case Example -- [HTML Version] [PDF Version]
-
Long-Term Services and Supports Committee Update -- [HTML Version] [PDF Version]
-
NAPA Driver Diagram -- [HTML Version] [PDF Version]
-
Overview of the Clinical Trial Pipeline for AD -- [HTML Version] [PDF Version]
-
Overview on NIA Preclinical Pipeline -- [HTML Version] [PDF Version]
-
Participating in an Alzheimer's Clinical Study: Perspectives on Involvement of a Person Living with Dementia and Her Study Partner -- [HTML Version] [PDF Version]
-
Progress Since October -- [HTML Version] [PDF Version]
-
Research Progress on Alzheimer's Disease and Related Dementias -- [HTML Version] [PDF Version]
-
Research Subcommittee Agenda: The Journey from Targets to Treatments -- [HTML Version] [PDF Version]
Videos
-
Updates since October meeting -- [Video]
-
NAPA Driver Diagram -- [Video]
-
Federal Updates -- [Video]
-
Public Comments -- [Video]
-
Research Subcommittee Agenda -- [Video]
-
Care Summit Update -- [Video]
