Friday, January 26, 2018
Printer Friendly Version in PDF Format (3 PDF pages)
Research Subcommittee Agenda: The Journey from Targets to Treatments
Brad Hyman and Allan Levey, Co-chairs
National Alzheimer's Project Act (NAPA) and the National Plans to Address Alzheimer's Disease
- Prevent and Effectively Treat Alzheimer's Disease by 2025.
- Optimize Care Quality and Efficiency.
- Expand Supports for People with Alzheimer's Disease and Their Families.
- Enhance Public Awareness and Engagement.
- Track Progress and Drive Improvement.
NIH AD and ADRD Research Summits - Recommendations
- AD 2015
- Interdisciplinary Research to Understand the Heterogeneity and Multifactorial Etiology of Disease
- Transforming AD Therapy Development: From Targets to Trials
- New Strategies for Prevention
- Innovating Disease Monitoring, Assessment and Care
- Empowering Patients, Engaging Citizens
- Enabling Partnerships for Open Innovation
- ADRD 2016
- Multiple Etiology Dementias -- Diagnosing Dementia in the 21st Century
- Non-Governmental Organizations
- AD/ADRD Health Disparities
- Lewy Body Dementias
- Frontotemporal Lobar Degeneration
- Vascular Contributions to Cognitive Impairment and Dementia
The Progression of Alzheimer's Disease and Clinical Trials
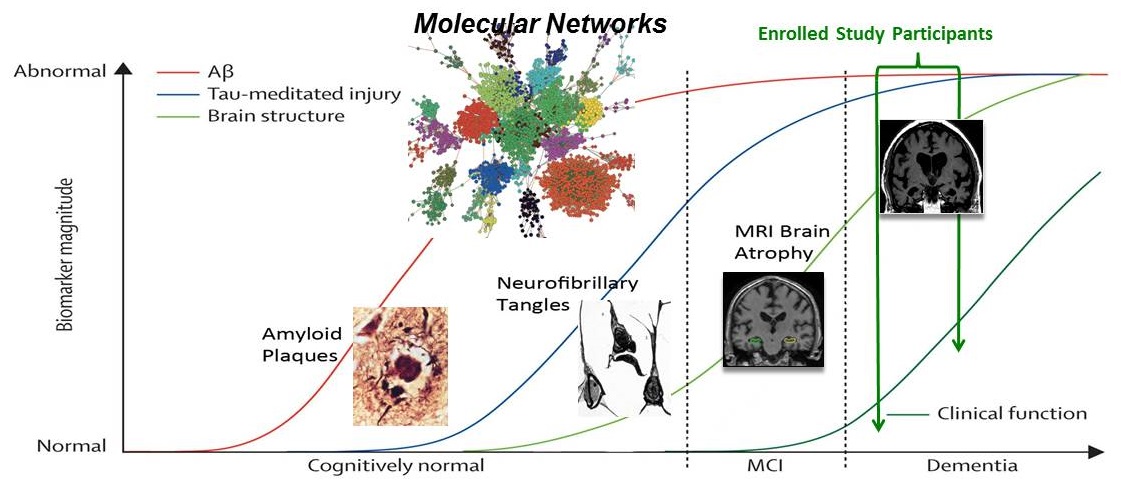 |
| Modified from Jack et al., Lancet Neurol (2009) |
Challenges of developing treatments for AD
- Prevention, disease modifying, and symptomatic treatments may have substantially different therapeutic targets, and certainly require different clinical trial designs
- Disease heterogeneity -- varied rates of progression -- make trials complicated to run with a typical trial having thousands of patients followed for 2 or more years, at a cost of hundreds of millions per trial
- Complicated disease with potentially different "phases" -- may need a "right person, right time, right drug" approach, in analogy to chronic diseases like atherosclerosis or even cancer -- where different therapies are used to prevent illness or treat established disease
- Recent high profile failures lead to caution in new investments
January 26, 2018 -- Advisory Council Meeting #27
The meeting was held on Friday, January 26, 2018, in Washington, DC. The Research Subcommittee took charge of this meeting's theme, focusing on the process from targets to treatments. The Council heard speakers on the preclinical pipeline, the clinical trial pipeline, and the industry perspective. The meeting also included discussion of a driver diagram to guide the Council's future work, updates and a report from the October Care Summit, and federal workgroup updates. Material available from this meeting is listed below and at https://aspe.hhs.gov/advisory-council-alzheimers-research-care-and-services-meetings#Jan2018.
Comments and questions, or alerts to broken links, should be sent to napa@hhs.gov.
General Information
-
Agenda -- [HTML Version] [PDF Version]
-
Meeting Announcement -- [HTML Version] [PDF Version]
-
Meeting Summary -- [HTML Version] [PDF Version]
-
Public Comments -- [HTML Version]
Handouts
-
Care Summit Report Themes -- [PDF Version]
-
NAPA Driver Diagram Draft Examples -- [PDF Version]
-
Outline for Care Summit Final Report -- [PDF Version]
Presentation Slides
-
AbbVie's R&D Vision for Alzheimer's Disease -- [HTML Version] [PDF Version]
-
Care Summit Report -- [HTML Version] [PDF Version]
-
Clinical Subcommittee Update -- [HTML Version] [PDF Version]
-
Initiatives, Partnerships and Collaboration to Help Patients with the Highest Unmet Need: Dominantly Inherited Alzheimer's Disease Trials Unit (DIAN-TU) as a Case Example -- [HTML Version] [PDF Version]
-
Long-Term Services and Supports Committee Update -- [HTML Version] [PDF Version]
-
NAPA Driver Diagram -- [HTML Version] [PDF Version]
-
Overview of the Clinical Trial Pipeline for AD -- [HTML Version] [PDF Version]
-
Overview on NIA Preclinical Pipeline -- [HTML Version] [PDF Version]
-
Participating in an Alzheimer's Clinical Study: Perspectives on Involvement of a Person Living with Dementia and Her Study Partner -- [HTML Version] [PDF Version]
-
Progress Since October -- [HTML Version] [PDF Version]
-
Research Progress on Alzheimer's Disease and Related Dementias -- [HTML Version] [PDF Version]
-
Research Subcommittee Agenda: The Journey from Targets to Treatments -- [HTML Version] [PDF Version]
Videos
-
Updates since October meeting -- [Video]
-
NAPA Driver Diagram -- [Video]
-
Federal Updates -- [Video]
-
Public Comments -- [Video]
-
Research Subcommittee Agenda -- [Video]
-
Care Summit Update -- [Video]
