Friday, January 26, 2018
Printer Friendly Version in PDF Format (14 PDF pages)
Overview on NIA preclinical pipeline
Eliezer Masliah, M.D.
Director, Division of Neuroscience,
National Institute on Aging, NIH
NIA Translational research pipeline for AD and ADRD:The Team
Genetics and Epigenetics
ADSP, GCAD, NIGADS, ADGC
Marilyn Miller
millerm@nia.nih.gov
Target Discovery and Validation
AMP-AD Targets, M2OVE-AD-AD, Resilience-AD
Suzana Petanceska
petanceskas@nia.nih.gov
Drug Discovery and Preclinical Drug Development
Drug development (U01), MODEL-AD, AlzPED, SBIR's
Lorenzo Refolo PhD
refolol@nia.nih.gov
Clinical Drug Development
AMP-AD Biomarkers, ABC-DS, ACTC, Clinical trials, DIAN-TU
Laurie Ryan PhD
ryanl@nia.nih.gov
Dementias of the aging population
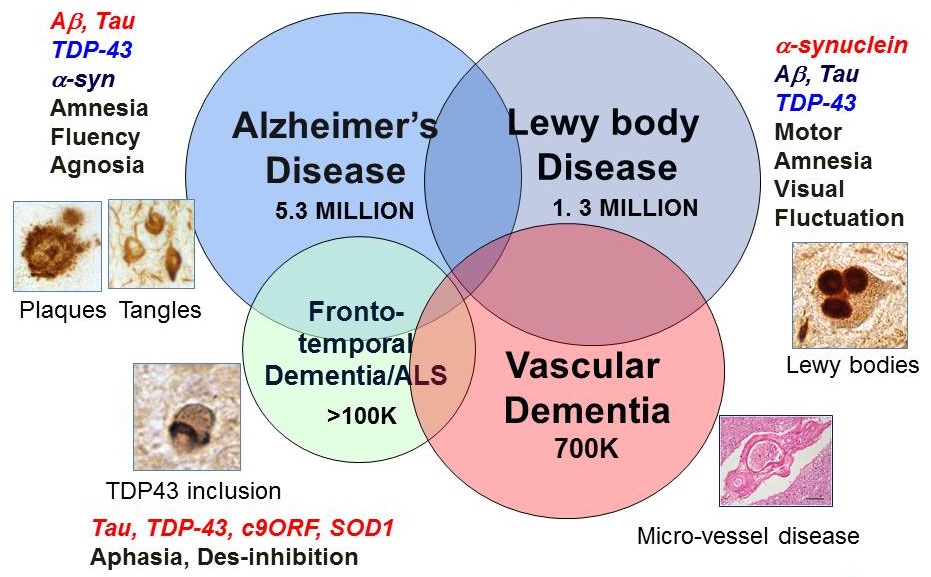 |
Mechanisms of toxicity in neurodegenerative disorders
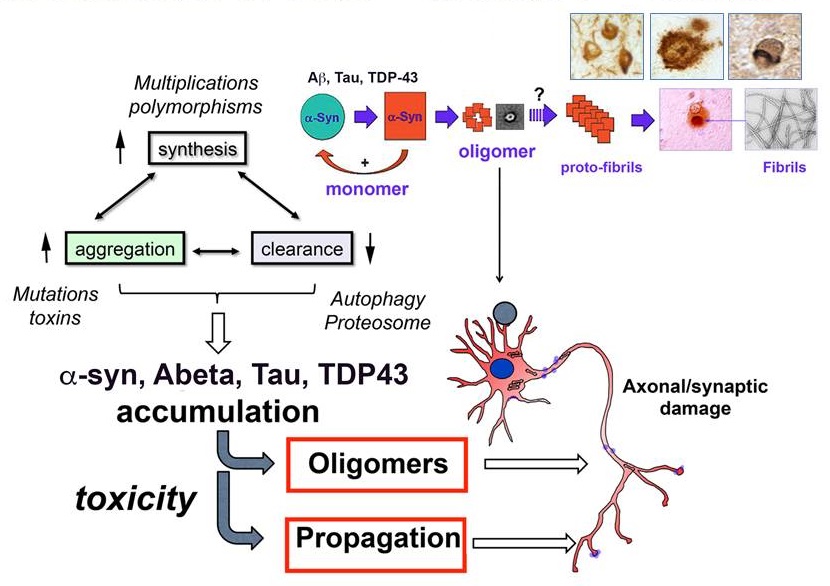 |
| Masliah and Valera MDS 2015 |
Mechanisms of neurodegeneration in Alzheimer's Disease
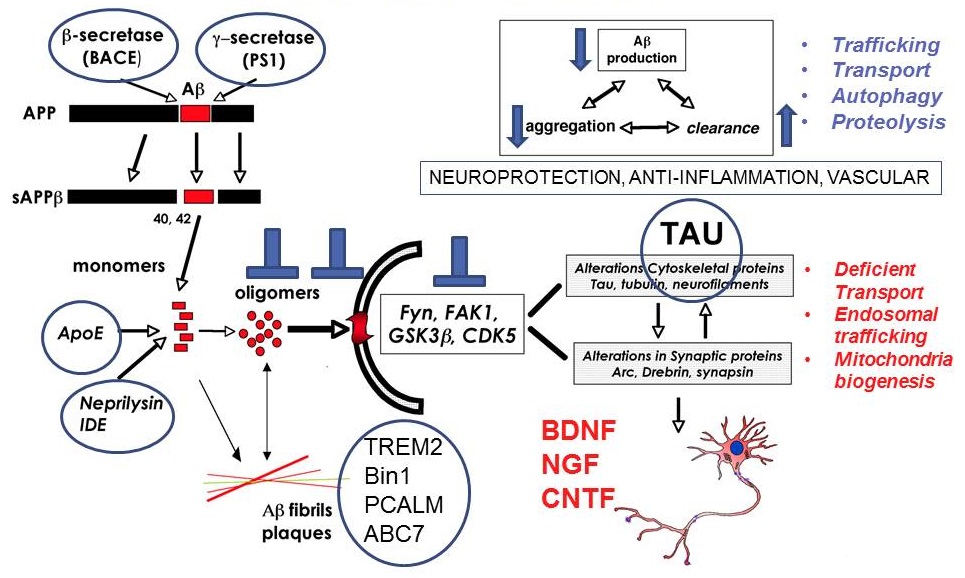 |
Alzheimer's Disease drug development pipeline-2017
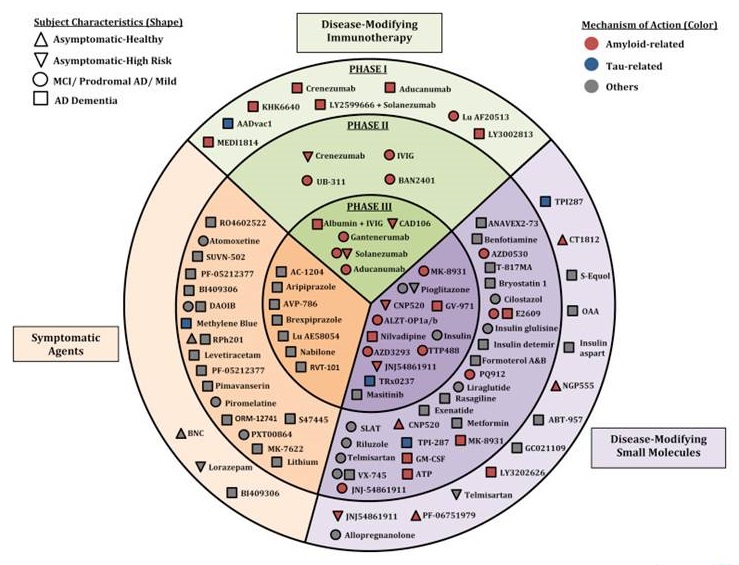 |
| Cummings et al Alz Dem 2017 |
AD pipeline- how does it compare to others?
| HCV | Alzheimer disease | MRSA | Industry average* | |
|---|---|---|---|---|
| Preclinical | 30.2 | 119.3 | 13.0 | 14.6 |
| Phase I | 10.5 | 33.6 | 4.7 | 8.6 |
| Phase II | 6.6 | 23.0 | 2.9 | 4.6 |
| Phase III | 1.7 | 5.8 | 1.8 | 1.6 |
| Registration | 1.1 | 1.0 | 1.1 | 1.1 |
| Launch | 1.0 | 1.0 | 1.0 | 1.0 |
| Overall success rate | 2.0% | 0.5% | 4.6% | 4.1% |
| Nature Review | Drug Discovery | ||||
Failure in the Clinic- where does the fault lie?
- Too late?
- Drug interventions are started at the wrong stage of disease
- Too little?
- May need greater drug effects
- Insufficient dose
- Lack of BBB penetration
- Lack of target engagement
- Drugs do not engage with intended targets in patients
- Lack of translatable pharmaco-dynamic biomarkers
- Wrong target?
- We are targeting the wrong pathophysiological mechanisms
- We need to target networks rather than single molecules
- Unclear which toxic species to target
Sperling, Jack and Aisen, Science Translational Medicine, 2011
Key AD Summits Recommendations
- Recognize the heterogeneity and the multifactorial nature of the disease.
- Support extensive molecular profiling of existing and establish new cohorts to fill the gaps in large-scale human data needed to build predictive models of disease and wellness.
- Employ new research paradigms such as systems biology and systems pharmacology.
- Enable rapid and extensive sharing of data, disease models, and biological specimens.
- Develop computational tools and infrastructure for storage, integration, and analysis of large-scale biological and other patient-relevant data.
- Build new multidisciplinary translational teams and create virtual and real spaces where these teams can operate.
- Support and enable open science.
- Develop new precompetitive public-private partnerships.
- Change academic, publishing, and funding incentives to promote collaborative, transparent, and reproducible research.
- Engage patients, caregivers and citizens as direct partners in research.››
NAPA Research Goal #1: Treat and Prevent AD by 2025
- New Funding Opportunities and Public Private Partnerships
- Implementation Research Milestones
- NIH AD Research Summits Recommendations
NIH AD Research Summits: Path to Treatment and Prevention
- May 14-15, 2012
- Feb 9-10, 2015
- March 1-2, 2018
Integrated NIA AD-Drug Development Program
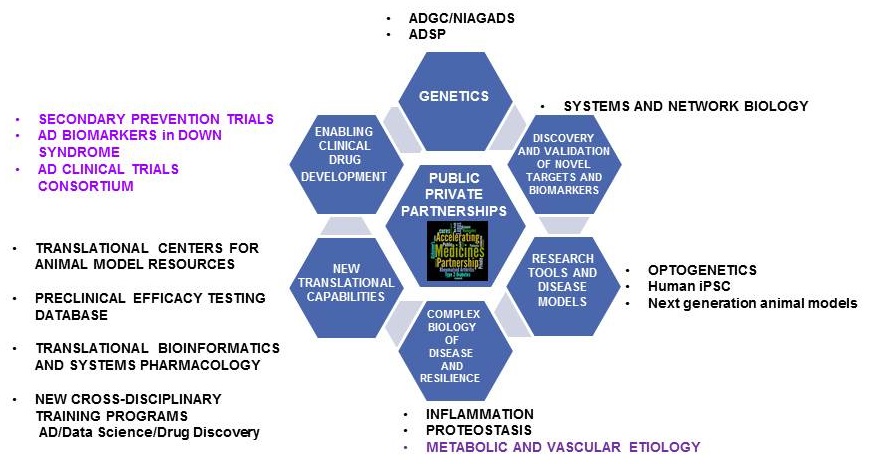 |
NIA and Trans-NIH translational pipeline for AD and ADRD
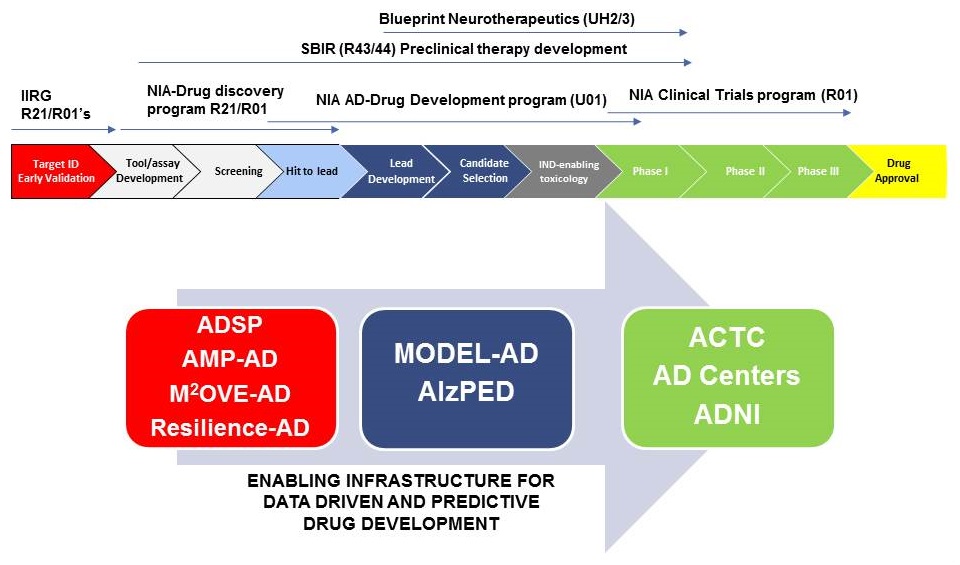 |
Alzheimer's Disease Sequencing Project (ADSP)
PI: G. Schellenberg; L. San U Penn; NIA Contact: Marilyn Miller
- New genomic variants contributing to Late-Onset AD (LOAD)
- Identify genomic variants contributing to protection against AD
- Provide insight as to why individuals with known risk factor variants escape developing AD
- Examine these factors in multi-ethnic populations to identify new pathways
Associated Programs
- ADGC- Alzheimer's Disease Genetic Consortium
- GCAD- Genome Center for AD
- NIAGDS- NIA Genetics of AD storage site
Now WGS in 10,000 controls and 10,000 AD, 10,000 diverse populations
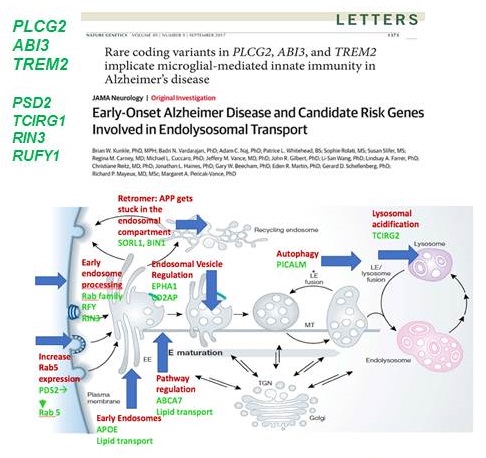 |
From single target to networks approach for AD-drug development
| Complexity of Drug Action |
|---|
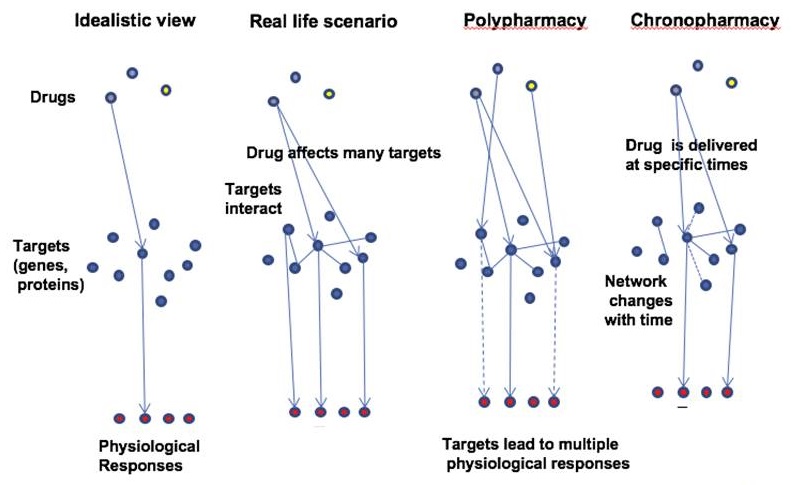 |
Accelerating Medicines Partnership (AMP)
| Alzheimer's Disease Program Foundation for the National Institutes of Health |
|---|
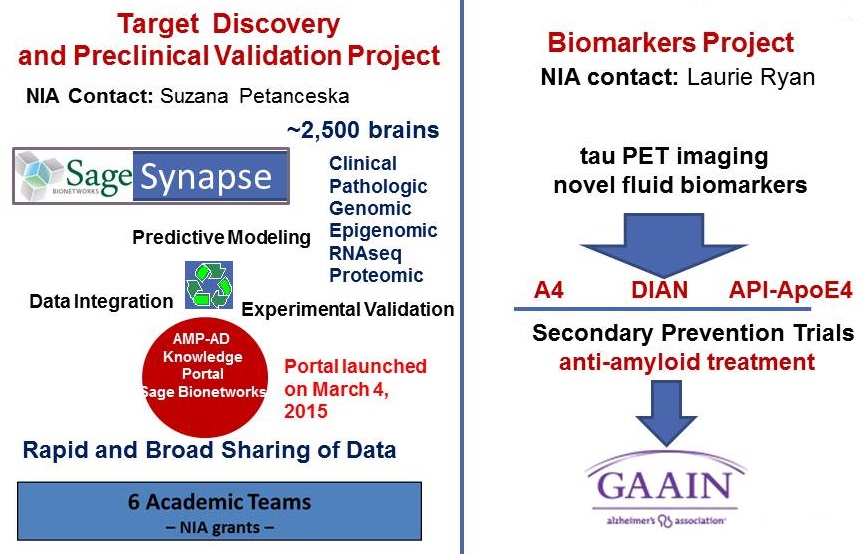 |
- Progress over 4 years:
- Centralized data resource established
- All data deliverables/milestones met
- Over 100 novel targets discovered; currently undergoing data-driven prioritization for further preclinical validation
- A variety of experimental validation models developed
- Novel biomarker discovery initiated
- AMP-AD Partners
- National Institute of Neurological Disorders and Stroke
- Food and Drug Administration
- Abbvie
- Biogen
- GlaxoSmithKline
- Lilly
- Alzheimer's Association
- Alzheimer's Drug Discovery Foundation
- USAgainst Alzheimer's
- Foundation for the National Institutes of Health
| AMP-AD Teams Candidate Targets: preliminary list |
||||||
|---|---|---|---|---|---|---|
| SNRNP70 | TGFBR1 | CCDC85C | RGS4 | |||
| U1-A | TGFBR2 | CIC | SCN2A | |||
| U1-C | BMPR1A | CSRP1 | OLFM3 | |||
| SNRPN | BMPR1B | DAB2IP | SLC22A10 | |||
| SNRPB | CRHR1 | FAM63A | ENAH | |||
| PLCD1 | TREM2 | FURIN | WWTR1 | |||
| PTRHD1 | TYROBP | HMG20B | LRP10 | |||
| SFRP1 | S100A8 | IGFBP5 | SYP | |||
| PPP1R7 | S100A9 | ISYNA1 | PCSK1 | |||
| DNM3 | P2RY2 | KIF1C | KMO | |||
| RTN4 | P2RX7 | PADI2 | PTTG1IP | |||
| EPB41L3 | P2RY12 | SLC38A2 | MLIP | |||
| TUBB3 | P2RY13 | SNAP25 | DLGAP1 | |||
| PLEC | OSMR | STX1A | MOAP1 | |||
| ANXA5 | TLR4 | STXBP3 | PRKCB | |||
| MSN | CR1 | SV2B | YAP1 | |||
| CD44 | CSF1R | SYT1 | GNA13 | |||
| LMNA | CX3CR1 | SYT12 | TRIM56 | |||
| SPI1 | ZBTB47 | |||||
| TNFRSF10A | VGF | |||||
| TNFRSF10B | PLXNB1 | |||||
|
||||||
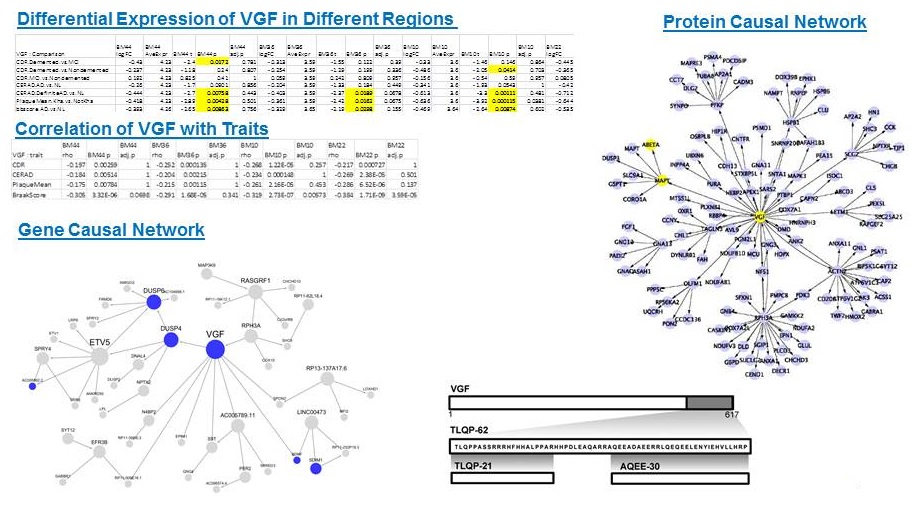
Secreted peptide VGF (non-acronymic)
Specifically expressed in a subpopulation of neuroendocrine cells, and is upregulated by nerve growth factor. The encoded secretory protein also shares similarities with the secretogranin/chromogranin family, however, its exact function is not known. Multiple VGF peptides reported to be significantly decreased between converting and non-converting MCI patients (ADNI) Spellman et al. 2015 - NIH (FNIH) Biomarkers Consortium CSF Proteomics Project Team
 |
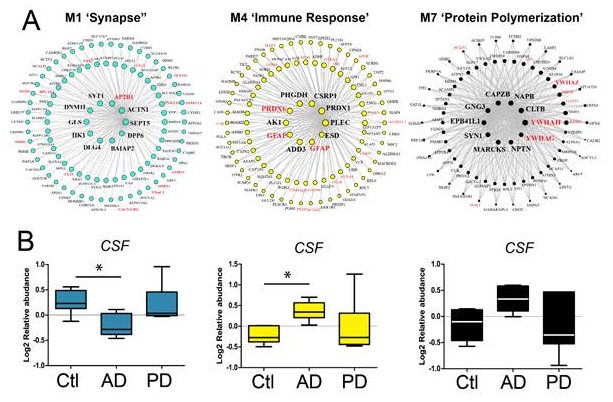
Protein Networks as Novel Biomarkers
AMP-AD Emory Team
PI: Allan Levey
Hub proteins from brain networks are found in human CSF and discriminate AD from control and PD patients. Hub proteins are defined as proteins with the highest intra-modular connectivity (i.e., proteins that are most central within the module) in the M1, M4 and M7 modules. Red symbols are proteins that were also identified in the CSF.
 |
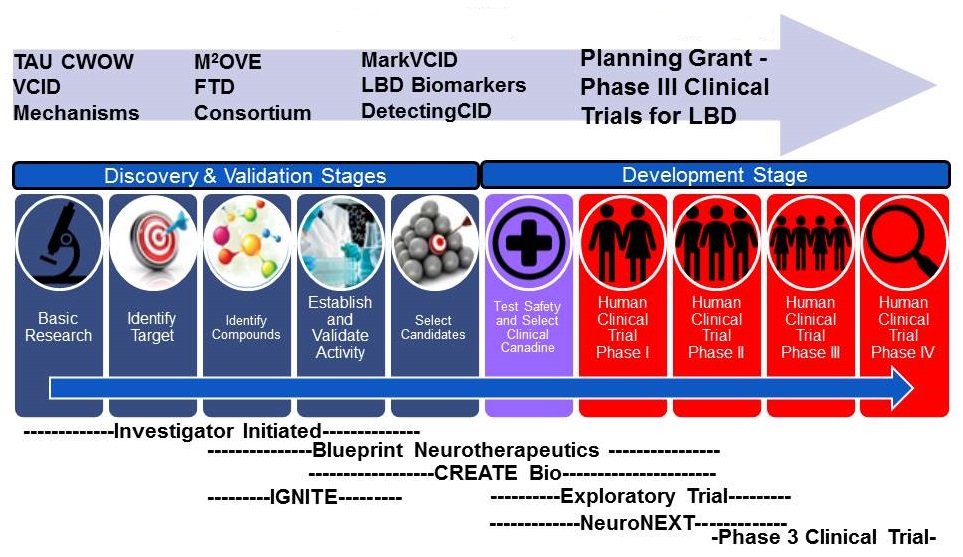
Building on and expanding the AMP-AD Target Discovery Project
Molecular Mechanisms of the Vascular Etiology of Alzheimer's Disease (M2OVE-AD)
NIA contact- Suzana Petanceska
A collaboration between NIA and NINDS
- ~$30 million over 5 years to support cross disciplinary research teams:
- 5 research teams will generate various "omics" data from brain and peripheral fluids from individuals participating in several natural history and population studies
- Predictions about molecular mechanisms will be explored in animal models (AD models and models of vascular/metabolic risk factors).
- Goals and deliverables:
- rapid and broad sharing of data
- deeper understanding of the phenotypes of risk and the mechanisms linking vascular risk factors, cerebrovascular disease and AD
- new disease-relevant therapeutic targets for prevention
- molecular signatures that can be non-invasively measured and used for patient stratification
NINDS ADRD Translation Research Initiatives
In collaboration with NIA
NINDS contact: Roderick Corriveau (roderick.corriveau@nih.gov)
 |
Some examples of NIA pipeline for AD and ADRD
 |
| AD Drug Development Progarm | ||
|---|---|---|
| PI | Institution | Project or Target |
| Tuszynski M | UCSD | BDNF Gene Delivery |
| Catalano C | Cognition Therapeutics, Inc | Receptor Antagonist CT0093 |
| Lin CL | OSU | Activators of glutamate transporter EAAT2 |
| Wagner S | UCSD | Soluble Gamma-Secretase Modulators |
| Ives J | Acumen Pharmaceuticals, Inc. | Amyloid-Beta Oligomer Selective Immunotherapy |
| Van Eldik L | U Kentucky | MW151, block cytokine |
| Agadjanvan M | Inst of Molecular Med | AV1959 anti-Amyloid vaccine |
| Thota G | Emory U | EP2 receptor antagonists |
| Watterson DM | Northwestern U | MAPK Inhibitors |
| Brinton R | USC | Allopregnanolone |
| Blueprint, Drug Discovery for CNS, and Preclinical Drug Development | ||
|---|---|---|
| PI | Institution | Project or Target |
| Rosenzweig-Lipson S | AgeneBio | GAB-A 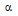 5 agonists 5 agonists |
| Brunden K | U Penn | Microtubule-stabilizers for tauopathy |
| Zhang, S | VCU | Aß oligomerization inhibitors |
| Pahan, K | Rush U | HEX, OCT and 3-hydroxy, 2, 2-dimethyl butyrate (HMB) |
| Bitan G | UCLA | 'Molecular Tweezers' CLR01 as |
| Witt K | Southern Illinois U | Somatostatin-based therapeutics |
| Varghese J | UCLA | ApoE4-targeted therapeutics |
| LU K | Beth Israel Deaconess | pT231-tau mAbs |
| Stutzmann G | Rosalind Franklin Univ | Ryanodine Calcium Channels |
| Sung Ok Yoon | OSU | JNK3 inhibitors |
| Diamond M | Wash U | Anti-Tau Antibodies |
| Cai J | USF | anti-Aß aggregation peptides |
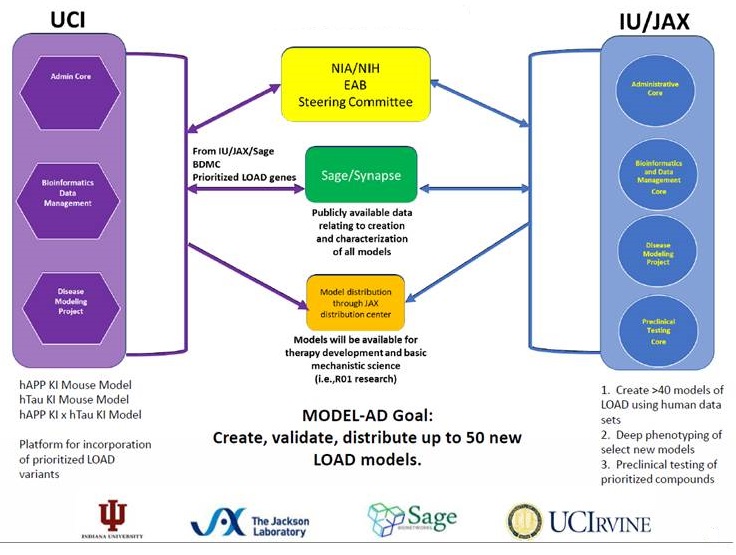
NIA Translational Center for Animal Model Resources MODEL-AD
- NIA contact, Larry Refolo
- RFA AG16-014 (U54): Indiana University/Jax Labs/Sage Bionetworks (Bruce Lamb, PI) and UCI (Frank LaFerla) http://grants.nih.gov/grants/guide/rfa-files/RFA-AG-16-014.html
- Maximize human datasets to identify putative variants, genes and biomarkers for AD
- Generate, phenotype and validate the next generation of Tg mouse models of AD (50 new models over 5 years; deep, longitudinal phenotyping)
- Develop a preclinical testing pipeline that implements rigorous study design and data analysis
- Make data and animal models available to the research community for use in therapy development without IP barrier.
NIA Translational Center for Animal Model Resources
| MODEL-AD |
|---|
 |
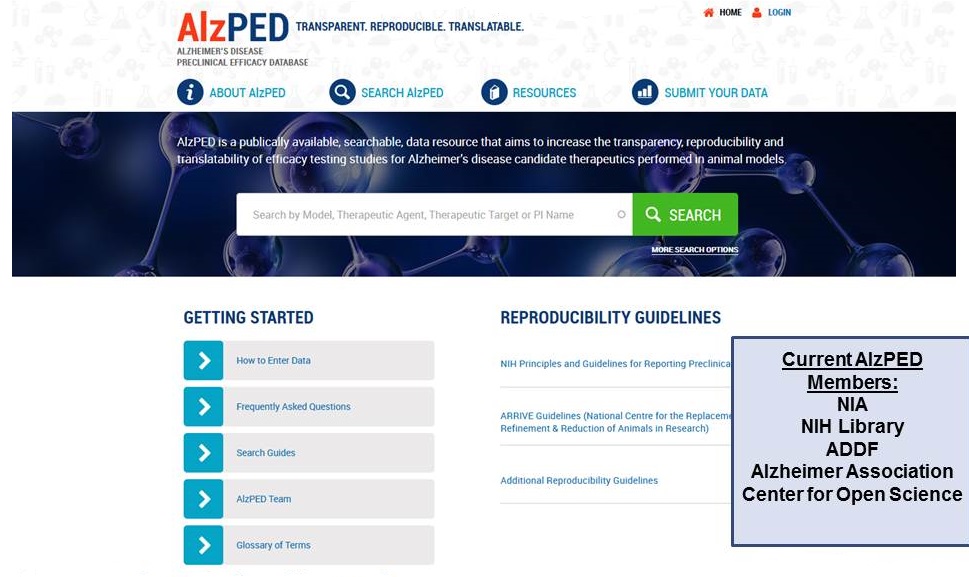
Alzheimer's Disease Preclinical Efficacy Database
NIA contact, Larry Refolo
https://alzped.nia.nih.gov/
 |
New* NIA funding opportunities for translation research
- Network biology of resilience to AD risk
- Sex-differences in AD risk and responsiveness to treatment
- Translational bioinformatics for drug repositioning and combination therapy development for AD
- Closing the expertise/skills gap in data science and drug discovery
- PAR18-524 (T32) https://grants.nih.gov/grants/guide/pa-files/PAR-18-524.html
- PAR17-052 (K18) https://grants.nih.gov/grants/guide/pa-files/PAR-17-052.html
SBIR/STTR opportunities
- Advancing Research on Alzheimer's Disease (AD) and Alzheimer's-Disease-Related Dementias (ADRD) (R41/R42/R43/R44)
- Tools for Clinical Care and Management of Alzheimer's Disease (AD) and its Comorbidities (R41/R42/R43/R44)
- Development of Socially-Assistive Robots (SARs) to Engage Persons with Alzheimer's Disease (AD) and AD-Related Dementias (ADRD), and their Caregivers (R41/R42/R43/R44)
- https://www.nia.nih.gov/research/grants-funding/small-business-innovation-research-and-technology-transfer-programs
Next steps toward developing an AD Translational pipeline
| Attaining the Goal of Precision Medicine for AD |
|---|
 |
Alzheimer's Disease Summit Program- 2018
Alzheimer's Disease Research Summit 2012: Path to Treatment and Prevention
March 1-2, 2018
National Institutes of Health
Bethesda, MD
The 2018 Summit will build on the foundation laid by the NIH AD Research Summits held in 2012 (https://www.nia.nih.gov/news/alzheimers-disease-research-summit-offers-research-recommendations) and 2015 (https://www.nia.nih.gov/research/recommendations-nih-ad-research-summit-2015). It will feature progress towards achieving the AD research implementation milestones (https://www.nia.nih.gov/research/milestones) and continue the development of an integrated multidisciplinary research agenda necessary to enable precision medicine for AD treatment and prevention.
- Novel Mechanistic Insights into the Complex Biology and Heterogeneity of AD
- Enabling Precision Medicine for AD
- Translational Tools and Infrastructure to Enable Predictive Drug Development
- Emerging Therapeutics
- Understanding the Impact of the Environment to Advance Disease Prevention
- Advances in Disease Monitoring, Assessment and Care
- Building an Open Science Research Ecosystem to Accelerate AD Therapy Development
THANKS
January 26, 2018 -- Advisory Council Meeting #27
The meeting was held on Friday, January 26, 2018, in Washington, DC. The Research Subcommittee took charge of this meeting's theme, focusing on the process from targets to treatments. The Council heard speakers on the preclinical pipeline, the clinical trial pipeline, and the industry perspective. The meeting also included discussion of a driver diagram to guide the Council's future work, updates and a report from the October Care Summit, and federal workgroup updates. Material available from this meeting is listed below and at https://aspe.hhs.gov/advisory-council-alzheimers-research-care-and-services-meetings#Jan2018.
Comments and questions, or alerts to broken links, should be sent to napa@hhs.gov.
General Information
-
Agenda -- [HTML Version] [PDF Version]
-
Meeting Announcement -- [HTML Version] [PDF Version]
-
Meeting Summary -- [HTML Version] [PDF Version]
-
Public Comments -- [HTML Version]
Handouts
-
Care Summit Report Themes -- [PDF Version]
-
NAPA Driver Diagram Draft Examples -- [PDF Version]
-
Outline for Care Summit Final Report -- [PDF Version]
Presentation Slides
-
AbbVie's R&D Vision for Alzheimer's Disease -- [HTML Version] [PDF Version]
-
Care Summit Report -- [HTML Version] [PDF Version]
-
Clinical Subcommittee Update -- [HTML Version] [PDF Version]
-
Initiatives, Partnerships and Collaboration to Help Patients with the Highest Unmet Need: Dominantly Inherited Alzheimer's Disease Trials Unit (DIAN-TU) as a Case Example -- [HTML Version] [PDF Version]
-
Long-Term Services and Supports Committee Update -- [HTML Version] [PDF Version]
-
NAPA Driver Diagram -- [HTML Version] [PDF Version]
-
Overview of the Clinical Trial Pipeline for AD -- [HTML Version] [PDF Version]
-
Overview on NIA Preclinical Pipeline -- [HTML Version] [PDF Version]
-
Participating in an Alzheimer's Clinical Study: Perspectives on Involvement of a Person Living with Dementia and Her Study Partner -- [HTML Version] [PDF Version]
-
Progress Since October -- [HTML Version] [PDF Version]
-
Research Progress on Alzheimer's Disease and Related Dementias -- [HTML Version] [PDF Version]
-
Research Subcommittee Agenda: The Journey from Targets to Treatments -- [HTML Version] [PDF Version]
