Friday, January 26, 2018
Printer Friendly Version in PDF Format (20 PDF pages)
Initiatives, Partnerships and collaboration to help patients with the highest unmet need: Dominantly Inherited Alzheimer's Disease Trials Unit (DIAN-TU) as a case example
Randall J. Bateman M.D.
Charles F. and Joanne Knight Distinguished Professor of Neurology &DIAN Trials Unit Director
Disclosure:
- Sources of Research Support:
- NIH R01NS065667
- NIH UF1AG032438 (DIAN)
- NIH ADRC (P50AG05681)
- NIH U01AG042791 (DIAN-TU)
- NIH WU CTSA award (UL1RR024)
- NIH U01AG042791-S1 (DIAN-TU)
- NIH Mass Spectrometry Resource (RR000954)
- NIH R01AG046179 (DIAN-TU-APT)
- NIH R56AG053267 (DIAN-TU NexGen)
- Alzheimer's Association, BrightFocus Foundation, GHR Foundation, Ruth K. Broad Biomedical Research Foundation, Anonymous Foundation, Cure Alzheimer's Fund
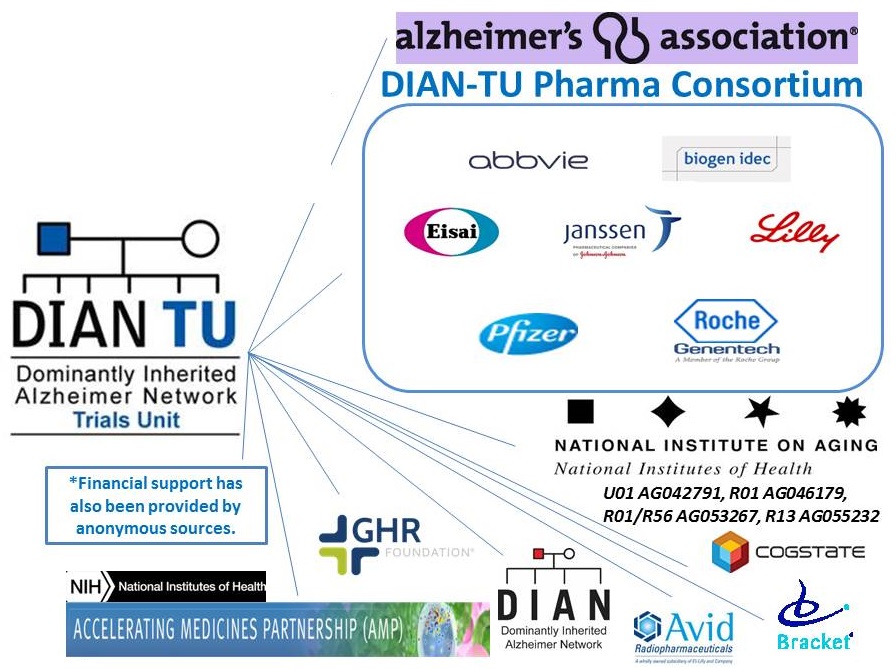
- DIAN Pharma Consortium:
- Abbvie, Amgen, AstraZeneca, Biogen, Eisai, Elan, Eli Lilly, Forum, Genentech, Hoffman La-Roche, Janssen AIP, Mithridion, Novartis Pharm AG, Pfizer, Sanofi-Aventis
- DIAN-TU:
- Washington University and Dr. David Holtzman, Department Head of Neurology where the research is being conducted, may receive royalty income for an investigational drug called solanezumab, which was developed by Dr. Holtzman and licensed by Washington University to Eli Lilly & Company. Solanezumab is evaluated in this research.
- Companies:
- Co-founder C2N Diagnostics
- Invited Speaker:
- BMS, FDA, Eli Lilly, Merck, Pfizer, Elan, Wyeth, Novartis, Abbott, Biogen, Takeda Foundation, Fidelity Biosciences Research Initiative, EMA
- Editorial Board:
- Alzheimer's Research and Therapy, The Journal of Prevention of Alzheimer's Disease
- Consulting Relationships:
- Boehringer-Ingelheim, DZNE, Probiodrug AG, Medscape, Eisai, Forum (En Vivo) Scientific Advisory Board, Merck, Sanofi, Novartis, Roche, Global Alzheimer's Platform, IMI JU-Project EMIF
A brief history of Alzheimer's disease modifying prevention
- 2018? - Primary Prevention
- 2017 - Prevention trials with oral secretase inhibitors
- 2014 - Prevention trials targeting at-risk individuals
- 2012 --first prevention trial against amyloid-beta is launched
- 2012 - Aß lowering mutation discovered which dramatically protects against Alzheimer's
- 2000's -- first drugs targeting Aß - A cause of Alzheimer's are developed
- 1991 - Mutations discovered that cause early onset Alzheimer's in families -- later discovered in Alzheimer's first patient
- 1906 - Dr. Alois Alzheimer describes first Alzheimer's disease patient -- disease of brain -- plaques and tangles
- Senility known throughout history
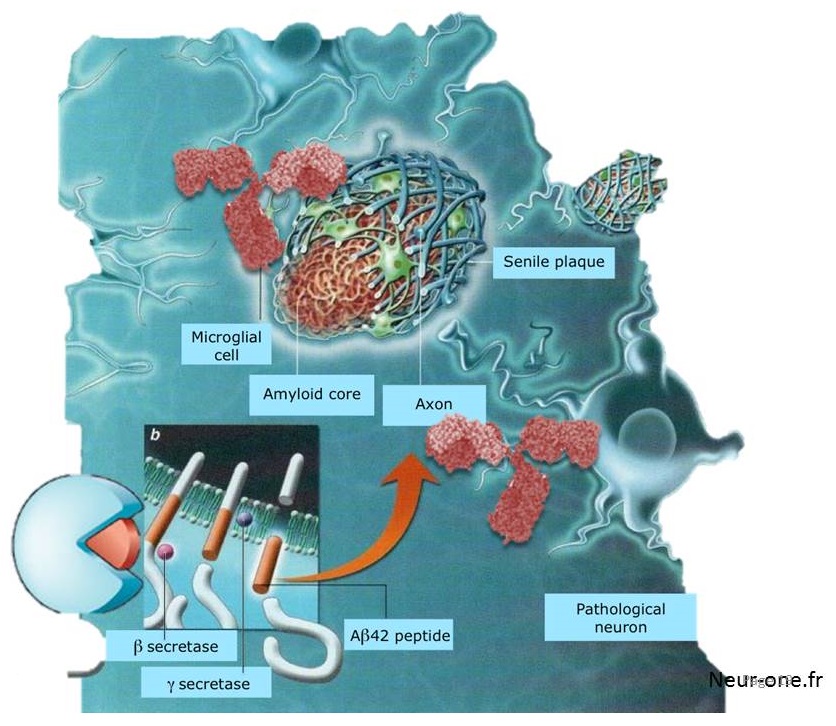
Autosomal Dominant Alzheimer's Disease (ADAD)
- Less than 1% of AD cases result from autosomal dominant mutations in three genes directly involved in amyloid beta (Aß) production
- Amyloid precursor protein (APP)
- Presenilin 1 (PSEN1)
- Presenilin 2 (PSEN2)
- Auguste D., the first AD patient ever described by Alois Alzheimer, was later found to carry an ADAD mutation in presenilin 1 (F176L)
Comparison of Dominantly Inherited and Sporadic Alzheimer's Disease
| Dominantly Inherited AD | Sporadic AD | |
|---|---|---|
| Clinical presentation | Amnestic | Amnestic |
| Cognitive deterioration | Memory, frontal/executive, generalized cognitive decline | Memory, frontal/executive, generalized cognitive decline |
| MRI | Hippocampal atrophy and whole brain atrophy | Hippocampal atrophy and whole brain atrophy |
| PiB PET | Cortex plus basal ganglia | Cortex |
| FDG PET | Parieto-occipital hypometabolism | Parieto-occipital hypometabolism |
| CSF Aß 42 | Decreased by 50% | Decreased by 50% |
| CSF tau | Increased by 2-fold | Increased by 2-fold |
DIAN Observational: An International, Multi-Center Study
- Biomarker collection rate +80-90%
- >500 enrolled since 2008, 275 active participants, 52% 10 years or more prior to EYO
- 104 publications
- 16 presentations at 2017 AAIC, London, England
- DIAN resource requests to date:
- Data (n=125) Tissue (n=43)
- Data used to design and implement 3 arms in the DIAN-TU trial
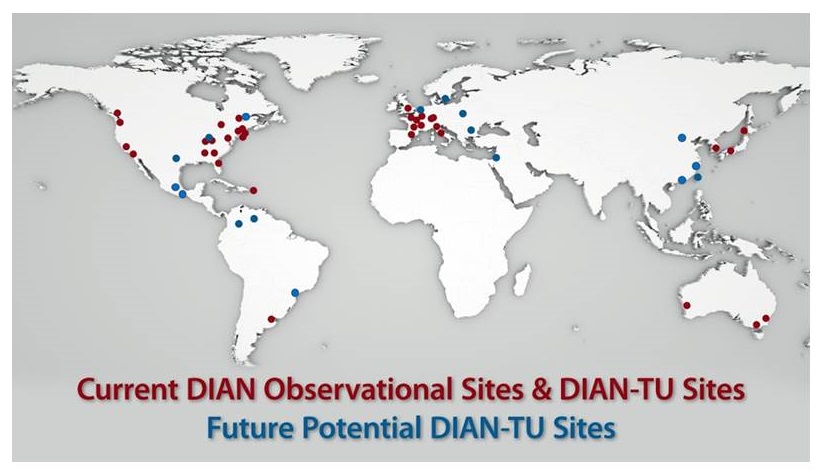
- Planned Expansion: Korea, Spain, Canada
- Renewal submission in 2018 for funding 2019-2024
- increased recruitment
- Focus on molecular cause of Alzheimer's
- Tau PET imaging
- Harmonization of DIAN-TU and DIAN EXR
Through public/private support and partnership, the DIAN-TU has launched trials to provide advancement of treatments, scientific understanding and improvements in the approach to Alzheimer's disease drug developments.
 |
DIAN Observational and DIAN-TU Trial sites
 |
DIAD amyloid deposition by estimated age of onset
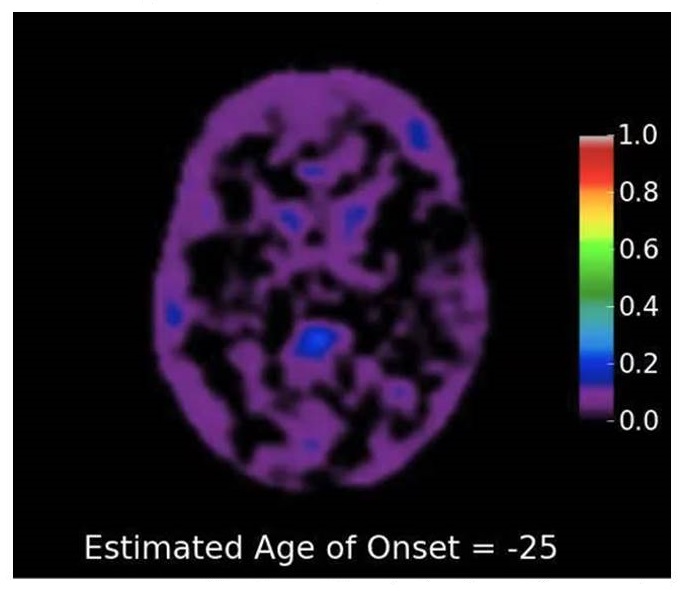 |
| Courtesy of Tammie Benzinger; Bateman et. al NEJM 2012 |
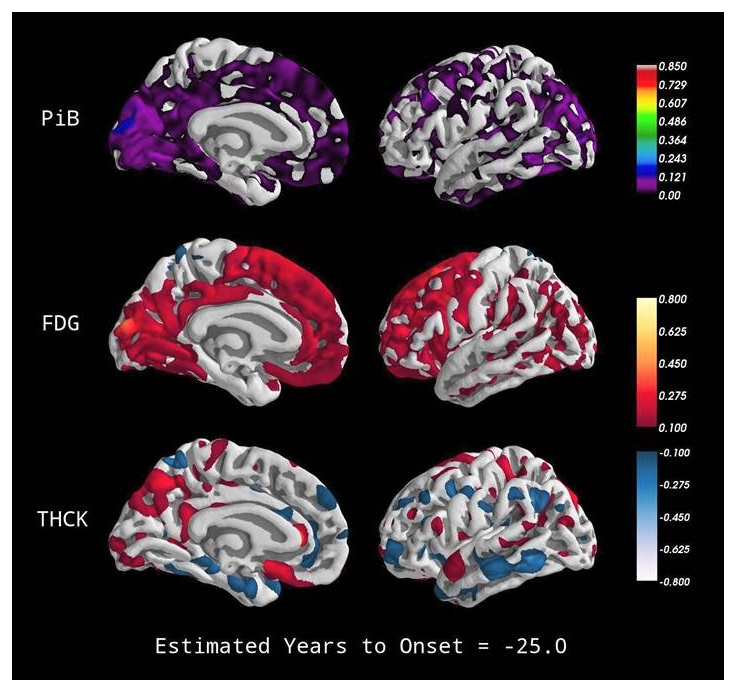 |
Stages of Proteinopathy in ADAD
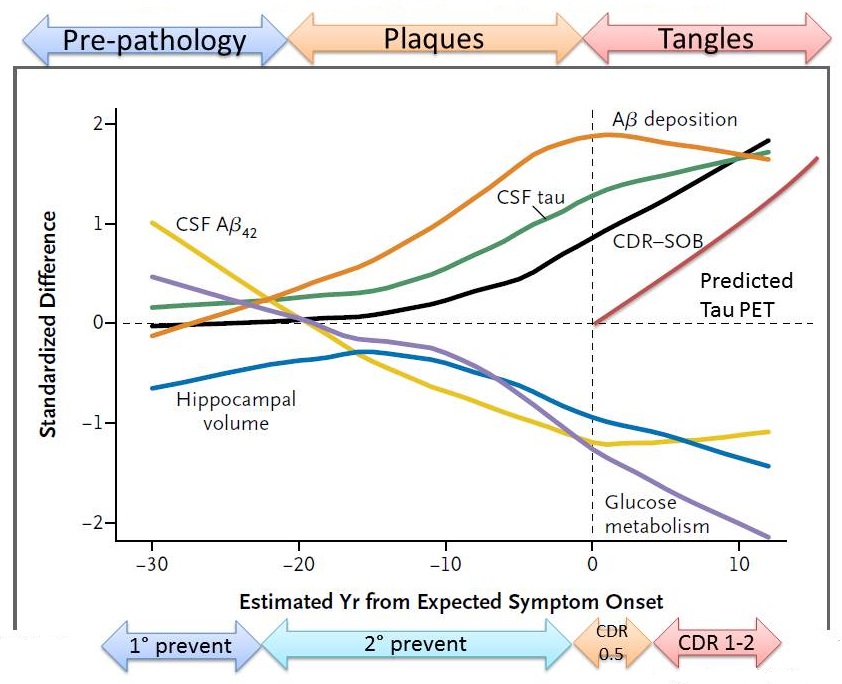 |
| The DIAN; Bateman et. al NEJM 2012 |
DIAN Therapeutic Trials Rationale
- Clinical onset of symptoms can be predicted at any point in lifespan, allowing therapeutic trials years or even decades before the clinical onset.
- DIAD has a clear cause of disease due to amyloid-beta: 'pure AD' without comorbid confounds
- Uniquely informative scientific information of disease progression, biomarkers and changes due to therapeutic treatments
- Common pathophysiology support general AD indication
- Successful implementation of prevention and symptomatic studies will inform as to the cause of AD and provide guidance for future therapeutic development.
Engaging patients: a DIAN-TU participant perspective
Embedded video that could not be included here.
Participant Interaction and Partnership
- DIAN Expanded Registry
- Serves as a key information and referral source for the DIAN Observational and DIAN-TU trials
- Register:
- https://dian.wustl.edu/our-research/registry/
- Call: 1-844-DIAN-EXR (342-6397)
- Email: dianexr@wustl.edu
| Total Registrants (15 Dec 2017) | 1628 |
|---|---|
| Total Family Registrants | 1415 |
| - DIAD Individuals | 381 |
| - At-Risk Individuals | 365 |
| Total Researcher Registrants | 213 |
| Exploratory Genetic Counseling and Testing Program (as of Dec 2017) |
|
|---|---|
| Pedigrees Reviewed | 147 |
| Approved for testing | 75 |
| Positive for mutation on PS1, PS2, APP | 45 (60%) |
| Pathogenic mutations | 41 (91%) |
| Variants of uncertain significance | 3 (7%) |
| Not Pathogenic | 1 (2%) |
DIAD Family Conference
- DIAD family networking opportunity
- Opportunity for DIAD families to have a dialogue with AD researchers, pharma, NIH and regulators
- Discussions:
- Scientific, medical, regulatory, advocacy and disease burden matters
- Support sessions specific to asymptomatic and symptomatic individuals, and family members
- Sponsored by the DIAN-TU, the Alzheimer's Association, and NIH (R13 AG 055232)
DIAD Family Conferences* to discuss scientific, medical, regulatory, advocacy and disease burden and also provide support sessions for asymptomatic and symptomatic individuals and their families
- July 18th, 2015 AAIC, Washington, D.C. Historic, first-time meeting of DIAD families
- 99 DIAD individuals and family members attended
- July 23rd, 2016 AAIC, Toronto, Ontario, "Too Young to Forget"
- 117 DIAD individuals and family members attended
- July 15th, 2017 AAIC, London, United Kingdom, "Key to the Cure"
- 152 DIAD individuals and family members attended
*Funding for this conference was made possible in part by the Alzheimer's Association and by 1 R13 AG 055232-01, PI McDade, from the National Institute on Aging. The views expressed in written conference materials or publications and by speakers and moderators do not necessarily reflect the official policies of the Department of Health and Human Services; nor does mention by trade names, commercial practices, or organizations imply endorsement by the U.S. Government.
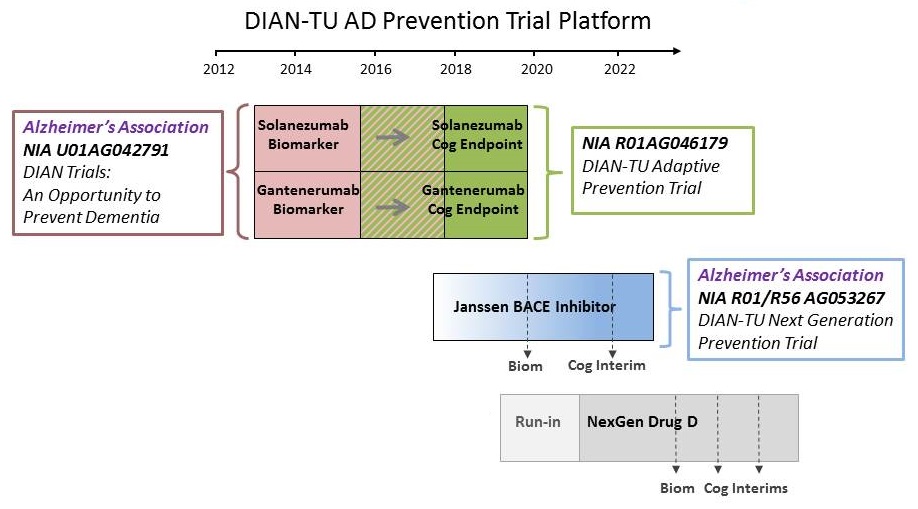
DIAN-TU NexGen Trial Design
- DIAN-TU NexGen:
- New drug arms
- 4 years of treatment
- Novel biomarkers
- Uses DIAD-specific Disease Progression Model based on DIAN Obs. data.
- Cognitive interim analysis
- Dose adjustment for maximal effect
- Home-based cognitive testing
- Population: with or at-risk for a DIAD mutation; -15 to +10 years of EYO; CDR 0, 0.5, 1
 |
 |
Making a Difference
- Pioneering prevention studies in DIAD utilizing observational data from DIAN, disease progression models, comprehensive biomarkers, input from participants and family members, and inclusive discussions with stakeholders.
- "It is really cutting edge, and it is the right thing to do -- the trial, the observational study......" Janet Woodcock, 2015 DIAD Family Conference, https://dian-tu.wustl.edu/en/2015-family-conference/
- First trial in dominantly inherited AD and the first AD prevention trial using anti-amyloid drugs
- Identification of effective therapeutics for DIAD with the goal of registration for DIAD and SAD patient use.
- Established foundation/groundwork and is a model for multiple initiatives: AMP, IMI EPAD and GAP
- FDA comment "exemplar of next generation trials"
- Multi-drug arm DIAN-TU is ideal platform for testing combination treatments (e.g. anti-tau and anti-amyloid)
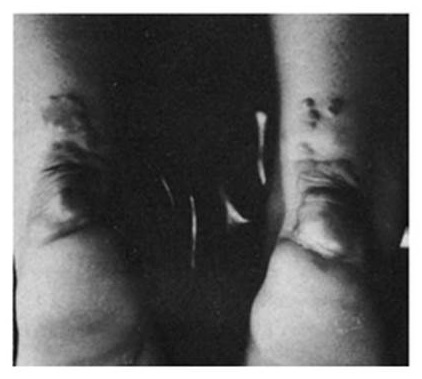
Historical Precedent:
Treatment of familial hypercholesterolemiawith compactin, the first statin drug
| Pre-treatment | Post-treatment |
|---|---|
 |
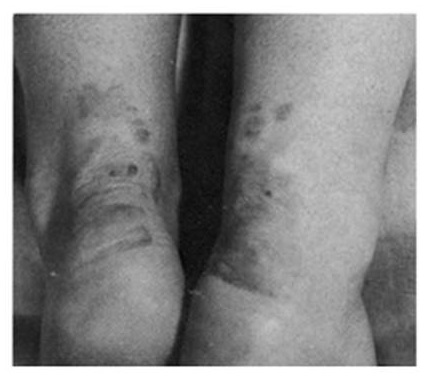 |
Similar to cholesterol, amyloid deposition and AD biomarkers can be used in studies to inform about drug engagement
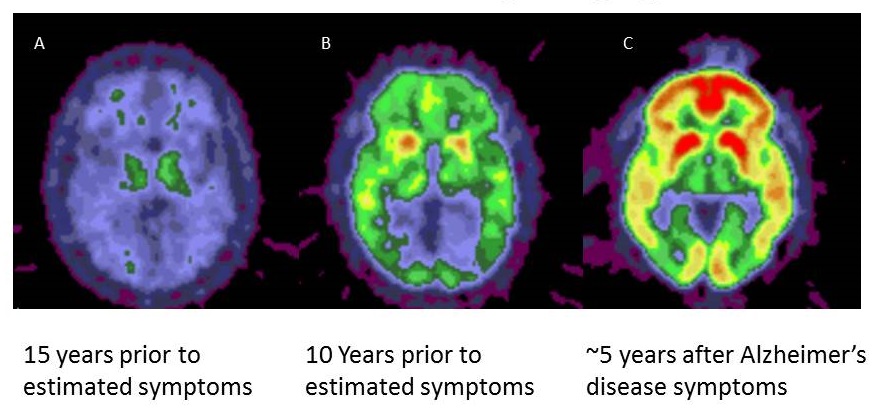 |
| Courtesy of Mark Mintun and Randy Bateman |
Proportional/Time Slowing Treatment Effect
Treated at EYO -15
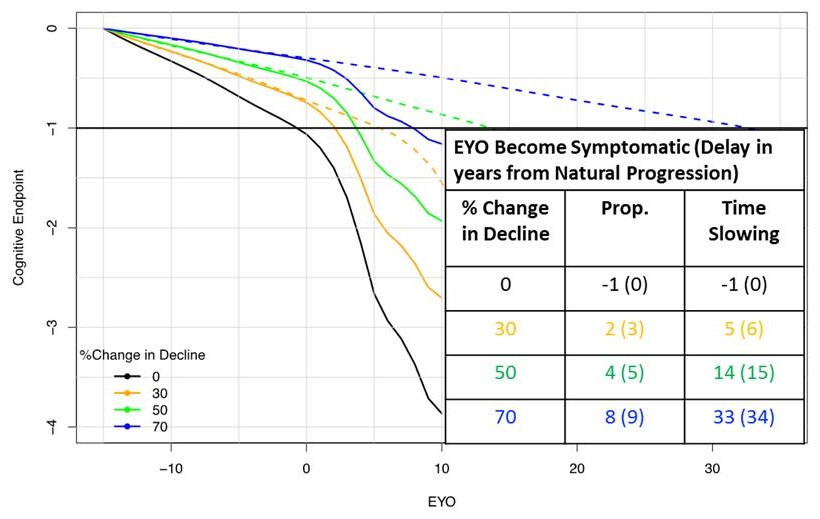 |
Rationale for Primary Prevention
ADAD cause is known, pathophysiology clear, risk of disease certain, and predictability of onset is high. The ADAD families have very high unmet need.
- Beta-secretase inhibitors and gamma-secretase modulators target molecular defect of ADAD mutations.
- Evidence of safety, convenience, relative low cost to manufacture and well-tolerated
- The DIAN -TU has demonstrated the feasibility of conducting complex clinical trials of long duration in a the ADAD population.
- Strong support for primary prevention by participants, families, and AD researchers, with regulatory pathways being established.
- Grant review scheduled January 25th
 |
| http://www.nature.com/news/stop-alzheimer-s-before-it-starts-1.22274 |
How early to go? Primary Prevention and timing of AD prevention trials related to core pathology and symptom onset
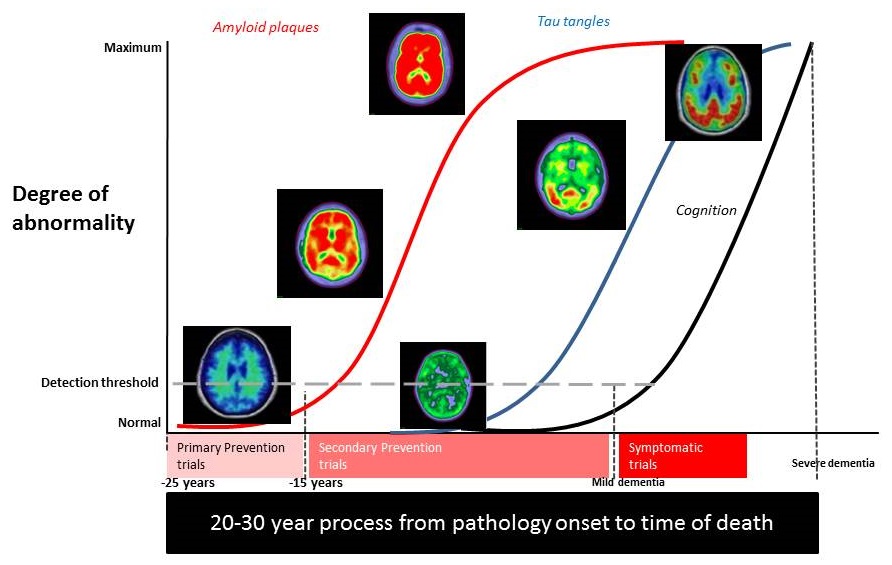 |
DIAN-TU Milestones
- 2011: DIAN-TU awarded grant by the Alzheimer's Association to launch trials
- 2011-2014: DIAN-TU Pharma Consortium (1st Term, 2011; 2nd Term, 2014): Pharma companies collaborating to develop dominantly inherited AD prevention trials (9 members)
- Multiple meetings with patient advocacy, academia, expert consultants, regulatory groups, DIAN participants and families, & Pharma partners to develop advanced prevention AD trials
- 26 therapies nominated to date
- 2012: DIAN-TU Trial biomarker phase launched with two treatments.
- FDA approval for first trial in dominantly inherited AD and the first AD prevention trial using anti-amyloid drugs
- Washington University serves as sponsor for the DIAN-TU trial
- 2013-2014: DIAN-TU awarded $6M and $26.7M NIA grants to support the DIAN-TU Trials
- 2014: Trial protocol amended to extend treatment to 4 years for a cognitive endpoint registration trial.
- 2014: DIAN-TU awarded funds from philanthropic foundations to continue efforts
- Foundation/groundwork established to enable pioneering prevention studies in DIAD and associated sites: Model for AMP, IMI EPAD and GAP. FDA comment "exemplar of next generation trials"
- 2015: Funds received for DIAN-TU NexGen trial design and DIAN-TU NexGen grant submitted
- 2016: Received Alzheimer's Association grant to launch the DIAN-TU NexGen drug arms.
- 2016: Received partial, start-up funding from the NIA to launch the NexGen drug arms.
- 2016: Received foundation funds for Primary Prevention trial design.
- 2016: Received foundation funds for Ambulatory Research in Cognition (ARC) testing.
- 2017: Received full NIA funding for and launched the NexGen (third) drug arm.
Resources
- Websites:
- DIAN Observational http://www.dian-info.org
- DIAN Expanded Registry http://www.dianexr.org
- DIAN-TU http://www.dian-tu.org
- Contact Information:
- DIAN-EXR email: dianexr@wustl.edu
- DIAN Expanded Registry Coordinator 844-DIAN-EXR (844-342-6397)
- DIAN Global Coordinator, 314-286-2643
The DIAN (NIH UF1AG032438)
The DIAN participants and family members
The Alzheimer's Association, ADAD Forum, DIAN Pharma Consortium
- Performance Sites
- Argentina
- Fundación para la Lucha contra las Enfermedades Neurológicas de la Infancia (FLENI), Ricardo Allegri
- Australia
- Mental Health Research Institute, Melbourne, Colin Masters
- Edith Cowan Univ , Perth, Martins
- Prince of Wales Medical Research Institutes, Sydney, Peter Schofield
- Germany
- Ludwig-Maximilians-Universität München, Adrian Danek
- University of Tübingen, Matthias Jucker
- Japan: DIAN-Japan, Mori
- Hirosaki University, Mikio Shoji
- Niigata University, Takeshi Ikeuchi
- Osaka City University, Hiroyuki Shimada
- University of Tokyo, Kazushi Suzuki
- Korea
- DIAN-Korea, JH Lee: Asan Medical Center, JH Roh
- United Kingdom
- Institute of Neurology, Univ College London, Martin Rossor
- United States
- University of Southern California, John Ringman
- University of California, San Diego, Doug Galasko
- Mayo Clinic, Jacksonville, Neil Graff-Radford
- Indiana University, Bernardino Ghetti
- Mass General Hospital/Brigham & Women's Hospital, Reisa Sperling
- Washington University, Randall J. Bateman
- Columbia University, Richard Mayeux
- University of Pittsburgh, William Klunk
- Butler Hospital/Brown University, Steve Salloway
- Argentina
DIAN-TU Trial Performance Sites
- Australia
- Neuroscience Research Australia, William Brooks
- The McCusker Foundation, Roger Clarnette
- Mental Health Research Institute, Colin Masters
- Canada
- McGill University, Serge Gauthier
- UBC Hospital, Robin Hsiung
- Sunnybrook Health Sci Centre, Mario Masellis
- France
- Hopital Roger Salengro, Florence Pasquier
- Hopital Neurologique Pierre Wertheimer, Maité Formaglio
- CHU de Rouen, Didier Hannequin
- CHU de Toulouse, Jérémie Pariente
- Groupe Hospitalier Pitie, Bruno Dubois
- Italy
- IRCCS Centro San Giovanni di Dio Fatebenefratelli, Giovanni Frisoni
- Azienda Ospedaliera Universitaria Careggi, Sandro Sorbi
- Spain
- Hospital Clinic I Provincial de Barcelona, Raquel Sanchez Valle
- United Kingdom
- The National Hospital for Neurology & Neurosurgery, Catherine Mummery
- United States
- Columbia University, Lawrence Honig
- University of Puerto Rico, Ivonne Jimenez-Velazques
- Indiana University, Jared Brosch
- University of Pittsburgh, Sarah Berman
- Washington University, Joy Snider
- University of Alabama, Erik Roberson
- Butler Hospital, Ghulam Surti
- Emory University, James Lah
- Yale University, Christopher Van Dyck
- UCSD, Doug Galasko
- University of Washington, Seattle, Suman Jayadev
DIAN-TU Administrative and Clinical Operations Team
Randall Bateman -- Director and PI, Eric McDade -- Associate Director
Sarah Adams, Stephanie Belyew, Erin Cattoor, David Clifford, Garland Edmonds, Angela Farrar, Erica Fowler, Amanda Fulbright, Angela Fuqua, Cortaiga Gant, Kurtis Hanks, Ron Hawley, Dottie Heller, Latoya Jones, Michelle Jorke, Nicole Kelley, Paulette MacDougall, Jacki Mallmann, Tayona Mayhew, Karen McCann, Susan Mills, Jennifer Petranek, Anna Santacruz, Jessi Smith, Annette Stiebel, Shannon Sweeney, Linda Watkins-Imhof, Mary Wolfsberger, Ellen Ziegemeier
- DIAN-TU Cores
- Administrative: Randall Bateman and team
- Biomarkers: Anne Fagan and team
- Biostatistics: Chengjie Xiong, Guoqiao Wang and team
- Cognition: Jason Hassenstab and team
- Genetics: Alison Goate, Carlos Cruchaga and team
- Imaging: Tammie Benzinger and team
- Neuropathology: Nigel Cairns and team
- DIAN-TU Collaborators
- Project Arm Leaders: Steve Salloway, Martin Farlow, Lon Schneider
- Consultants : Berry Consultants, Cornelia Kamp, Cardinal Health Regulatory Sciences, Granzer Regulatory Consulting
- DIAN-TU Therapy Evaluation Committee: Paul Aisen, Randall Bateman, Dave Clifford, David Cribbs, Bart De Strooper, Kelly Dineen, David Holtzman, Mathias Jucker, Jeffrey Kelly, Virginia Lee, Cynthia Lemere, Eric McDade, Susan Mills, John Morris, James Myles, Laurie Ryan, Matthias Staufenbiel, Raymond Tait, Robert Vassar
- DSMB Members: Gary Cutter, Steve Greenberg, Scott Kim, David Knopman, Willis Maddrey, Kristine Yaffe
- ADCS: Ron Thomas
- ATRI: Paul Aisen
- University of Michigan: Robert Koeppe
- Mayo Clinic: Clifford Jack
We gratefully acknowledge the DIAN and DIAN-TU participants and family members, DIAN Team, DIAN Steering Committee, Knight ADRC, Alzheimer's Association, ADAD Forum, NIH U01 AG042791, R01 AG046179, R01/R56 AG053267, R13 AG055232, DIAN-TU Pharma Consortium, GHR, Anonymous Foundation, Industry Partners (Eli Lilly & Co., Hoffman-LaRoche, Janssen, Avid Radiopharmaceuticals, Bracket, Cogstate), and Regulatory Representatives.
Backup Slides
How to improve screening for prevention trials?
Blood test for amyloid plaques in the brain
Blood plasma ß-amyloid 42/40 concentrations are consistently lower with Alzheimer's amyloid plaques, similar to cerebrospinal fluid.
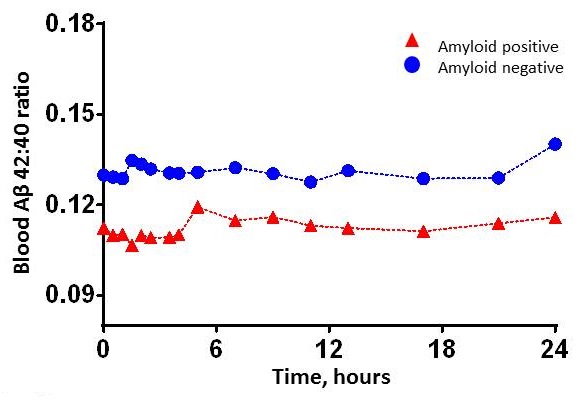 |
| Ovod, Ramsey, et. al Alzheimer's & Dementia 2017 |
Plasma ß-amyloid concentrations have sufficient specificity to be used as a potential screening test
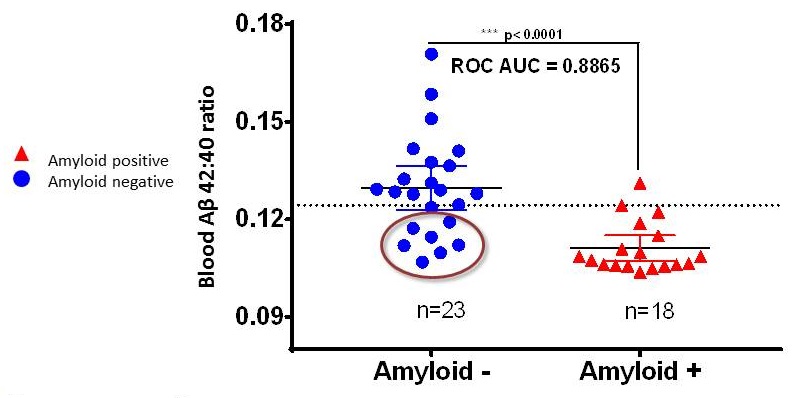 |
| Ovod, Ramsey, et. al Alzheimer's & Dementia 2017 |
Plasma ß-amyloid correlates with CSF and PIB PET ß-amyloid
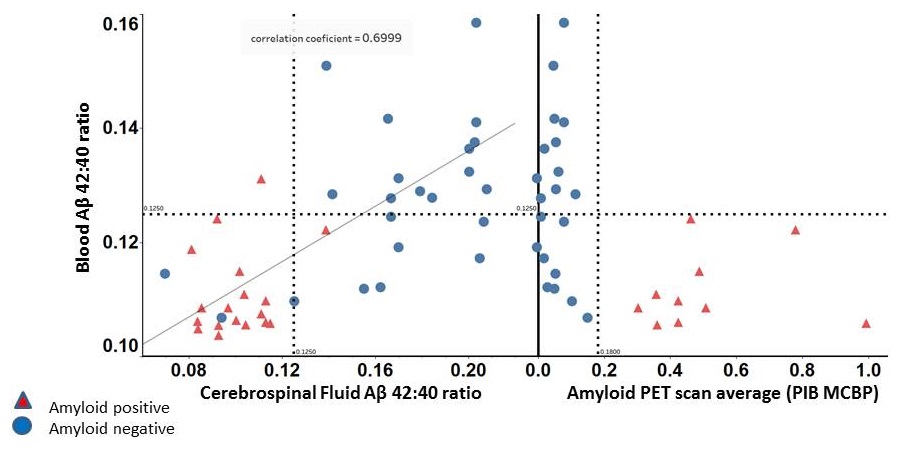 |
| Ovod, Ramsey, et. al Alzheimer's & Dementia 2017 |
Receiver Operating Characteristic of Single Time Points is Accurate with Average AUC of 0.86
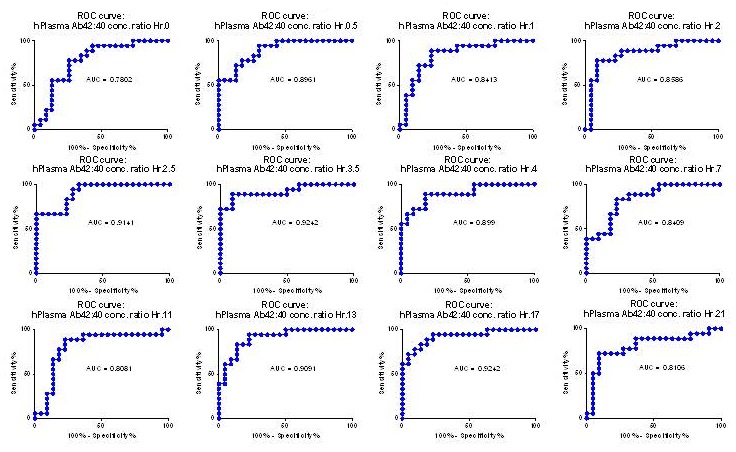 |
| Ovod, Ramsey, et. al Alzheimer's & Dementia 2017 |
Second Validation Cohort from Knight ADRC plasma samples (p=6E-8)
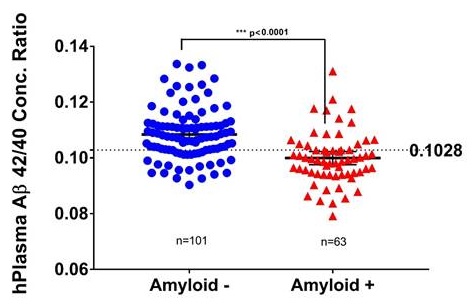 |
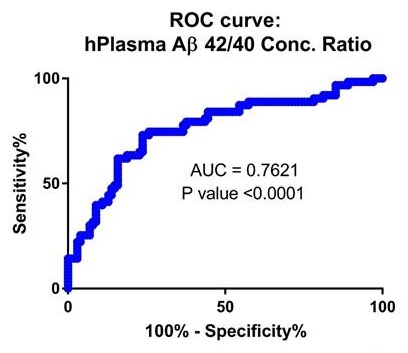 |
Third Validation Cohort from LOAD60 plasma samples
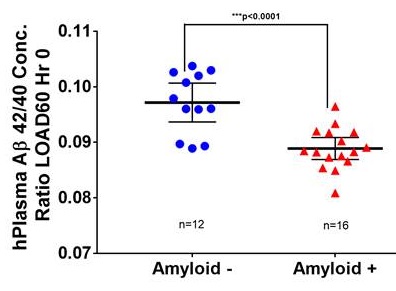 |
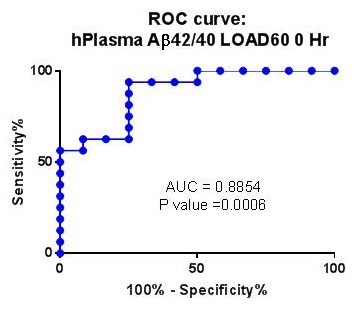 |
Blood plasma ß-amyloid as a test for Alzheimer's disease plaques
- Were validated using longitudinal samples in a pre-specified (clinicaltrials.gov) prospective study.
- Are related to gold-standard measures of amyloid PET scans and cerebrospinal fluid ß-amyloid for Alzheimer's disease amyloid plaques.
- The blood ß-amyloid test can be used to detect Alzheimer's disease amyloid plaques in individuals before symptom onset.
- The blood ß-amyloid test can accelerate ongoing and future clinical trials to speed drug discovery for Alzheimer's disease prevention and treatments.
- When effective drugs are found, the blood ß-amyloid test can be used to screen millions of people in the general public to identify who is at risk for Alzheimer's disease and start treatments even before memory loss and brain damage begins.
Ovod, Ramsey, et. al Alzheimer's & Dementia July 2017
January 26, 2018 -- Advisory Council Meeting #27
The meeting was held on Friday, January 26, 2018, in Washington, DC. The Research Subcommittee took charge of this meeting's theme, focusing on the process from targets to treatments. The Council heard speakers on the preclinical pipeline, the clinical trial pipeline, and the industry perspective. The meeting also included discussion of a driver diagram to guide the Council's future work, updates and a report from the October Care Summit, and federal workgroup updates. Material available from this meeting is listed below and at https://aspe.hhs.gov/advisory-council-alzheimers-research-care-and-services-meetings#Jan2018.
Comments and questions, or alerts to broken links, should be sent to napa@hhs.gov.
General Information
-
Agenda -- [HTML Version] [PDF Version]
-
Meeting Announcement -- [HTML Version] [PDF Version]
-
Meeting Summary -- [HTML Version] [PDF Version]
-
Public Comments -- [HTML Version]
Handouts
-
Care Summit Report Themes -- [PDF Version]
-
NAPA Driver Diagram Draft Examples -- [PDF Version]
-
Outline for Care Summit Final Report -- [PDF Version]
Presentation Slides
-
AbbVie's R&D Vision for Alzheimer's Disease -- [HTML Version] [PDF Version]
-
Care Summit Report -- [HTML Version] [PDF Version]
-
Clinical Subcommittee Update -- [HTML Version] [PDF Version]
-
Initiatives, Partnerships and Collaboration to Help Patients with the Highest Unmet Need: Dominantly Inherited Alzheimer's Disease Trials Unit (DIAN-TU) as a Case Example -- [HTML Version] [PDF Version]
-
Long-Term Services and Supports Committee Update -- [HTML Version] [PDF Version]
-
NAPA Driver Diagram -- [HTML Version] [PDF Version]
-
Overview of the Clinical Trial Pipeline for AD -- [HTML Version] [PDF Version]
-
Overview on NIA Preclinical Pipeline -- [HTML Version] [PDF Version]
-
Participating in an Alzheimer's Clinical Study: Perspectives on Involvement of a Person Living with Dementia and Her Study Partner -- [HTML Version] [PDF Version]
-
Progress Since October -- [HTML Version] [PDF Version]
-
Research Progress on Alzheimer's Disease and Related Dementias -- [HTML Version] [PDF Version]
-
Research Subcommittee Agenda: The Journey from Targets to Treatments -- [HTML Version] [PDF Version]
Videos
-
Updates since October meeting -- [Video]
-
NAPA Driver Diagram -- [Video]
-
Federal Updates -- [Video]
-
Public Comments -- [Video]
-
Research Subcommittee Agenda -- [Video]
-
Care Summit Update -- [Video]
