Friday, April 27, 2018
Printer Friendly Version in PDF Format (11 PDF pages)
Alzheimer's Disease and Related Dementias Research Update
Richard Hodes, M.D.
Director, NIA
Appropriations
- 2011: The National Alzheimer's Project Act (NAPA) was signed into law in January -- updated annually
- 2012: $50 M* redirected as part of Presidential initiative
- 2013: $40 M* redirected from unallocated funds in the NIH budget
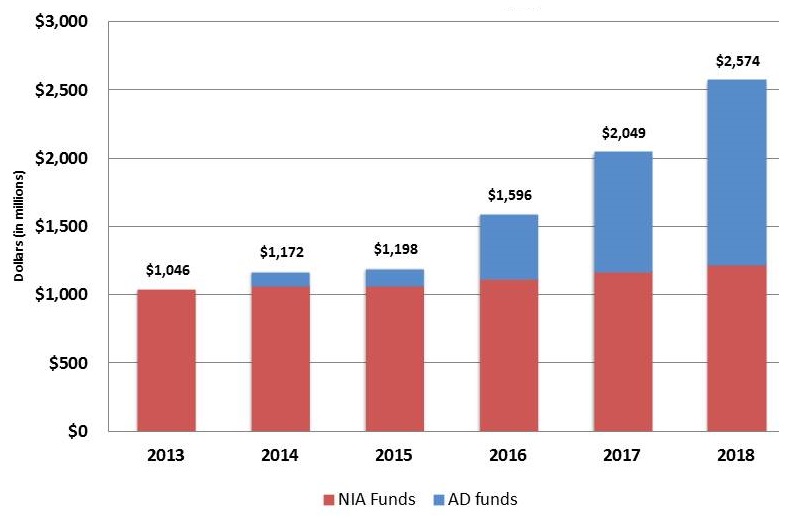
- 2014: $100 M in additional appropriations
- 2015: $25 M in additional appropriations
- 2016: $350 M in additional appropriations
- 2017: $400 M in additional appropriations
- 2018: $414 M in additional appropriations as of 3/23/18
*one-year money
Years displayed are Fiscal Years
NIA Appropriations
| Fiscal Years 2013-2018 |
|---|
 |
AD/ADRD Research Implementation Milestones and Bypass Budget Process
NIH Research Implementation Milestones are Based on Broad Input
- 2011: The National Alzheimer's Project Act (NAPA) was signed into law in January -- updated annually
- 2012: AD Summit
- 2013: ADRD Summit
- 2015: AD Summit
- 2016: ADRD Summit
- 2017: AD Care/Services Summit [Newest sources of milestone input]
- 2018: AD Summit [Newest sources of milestone input]
- 2019: ADRD Summit
- 2020: AD Care/Services Summit
https://www.nia.nih.gov/alzheimers/milestones
What Counts as AD/ADRD Research?
- The AD/ADRD payline applies to applications/awards that are coded as AD or Alzheimer's disease- related dementias (ADRD)
- The ADRD RCDC categories that report related dementias specifically named in the National Plan to Address Alzheimer's Disease are:
- Lewy Body dementia (LBD)
- Frontotemporal dementia (FTD)
- Vascular Cognitive Impairment/Dementia (VCI/D)
Combined External and Internal Input -- FY20 AD/ADRD Bypass Budget
- Input at 2012-2018 meetings:
- Academic research community
- Industry
- Non-governmental organizations
- Other Input:
- 2017 Request for Information
- Development of comprehensive milestones (NIH staff) -- based on summit and other input
- Trans-NIH (including NINDS) staff discussion; milestones edited to ensure comprehensive inclusion of priorities for FY20
- NIH staff "price" the milestones
- Final budget estimate for FY20
Timing of ADBB Releases and AD/ADRD Appropriations
- FY17 AD/ADRD milestones -- $323M extra needed
- FY17 ADBB Released July 2015
- $350M in FY16 supports acceleration of FY17 milestones
- FY18 AD/ADRD milestones - $414M extra needed
- FY18 ADBB Released August 2016
- $400M in FY17 supports acceleration of FY18 milestones
- FY19 AD/ADRD milestones - $597M extra needed
- FY19 ADBB Released July 2017
- $414M in FY18 supports acceleration of FY19 milestones
- FY20 ADBB: release planned July 2018
Complex Considerations for Future Bypass Budgets
- Past/current appropriations, including funds that have allowed us to address milestones "in advance"
- Progress on milestones -- what have we accomplished? what new milestones are emerging?
- Incorporating new care/services research milestones along with other milestones that are focused on drug development, etc.
- Large project needs -- emerging clinical trials, etc.
- Emerging investments by other groups -- what gaps are best filled by the NIA/NIH?
- Successes in recruiting new and established researchers to AD/ADRD research and capacity of the field
NIA-Alzheimer's Association Research Framework
NIA-AA Research Framework
| A-T-(N)- | Normal Alzheimer's biomarkers | |
|---|---|---|
| A+T-(N)- | Alzheimer's pathologic change | Alzheimer's continuum |
| A+T+(N)- | Alzheimer's disease | |
| A+T+(N)+ | Alzheimer's disease | |
| A+T-(N)+ | Alzheimer's and suspected non-Alzheimer's pathologic change | |
| A-T+(N)- | Non-Alzheimer's pathologic change | |
| A-T-(N)+ | Non-Alzheimer's pathologic change | |
| A-T+(N)+ | Non-Alzheimer's pathologic change | |
| A = Amyloid; T = Tau; N = Neurodegeneration Adapted from Jack et al., Alzheimer's & Dementia (2018) 14(4): 535-562. |
||
The framework will hopefully aid researchers in identifying individuals at risk for disease sufficiently early to test new prevention strategies as they emerge
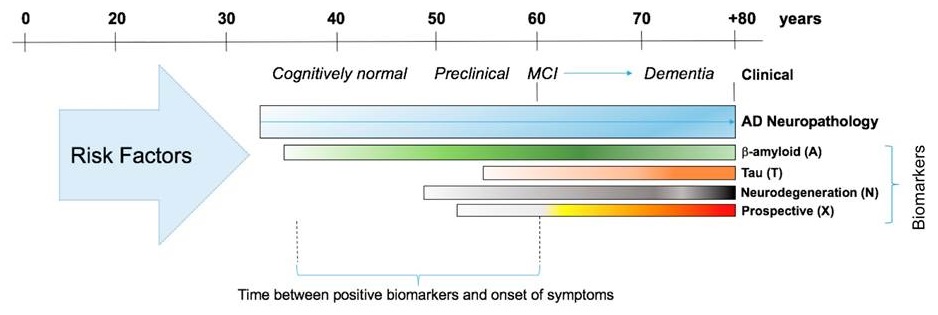 |
| Silverberg et al., Alzheimer's & Dementia (2018) 14(4):576-578. |
Summit Updates
National Research Summit on Care, Services and Supports for Persons with Dementia and Their Caregivers
- October 16-17, 2017
- NIH Campus, Natcher Building
- Convened by the National Institute on Aging at the NIH with support from the Foundation for the National Institutes of Health
- https://aspe.hhs.gov/national-research-summit-care-services-and-supports-persons-dementia-and-their-caregivers
Save the Date
- AD/ADRD Care/Services Summit (II)
- March 24-25, 2020
- Natcher Conference Center, Bethesda, MD
NEW Planned Evidence Review: Care Interventions for Individuals with Dementia and Their Caregivers
- Hundreds of studies have looked at the efficacy and effectiveness of care/caregiving interventions, but what is the quality of evidence supporting readiness for dissemination of any of these?
- NIA is funding a two-part study similar to that used for the recent dementia prevention literature review
- Initial steps:
- National Academies of Sciences, Engineering, and Medicine: Expert panel input on questions and study design www.nationalacademies.org/dementiacare
- Agency for Healthcare Research & Quality: Evidence review
- Draft study questions, and other planned study parameters will be posted for public comment
- The URL will be shared widely once available
NIH AD Research Summits: Path to Treatment and Prevention
- Dates:
- May 14-15, 2012
- Feb 9-10, 2015
- March 1-2, 218
- Goal: The 2018 Summit was built on the foundation laid by the NIH AD Research Summits held in 2012 and 2015. It featured progress towards achieving the AD research implementation milestones and continue development of an integrated multidisciplinary research agenda necessary to enable precision medicine for AD.
- https://www.nia.nih.gov/research/nih-ad-summit-2018-program-agenda
AD Research Summit Program
- The Summit Program was organized around 7 sessions and featured 86 speaker
- Novel Mechanistic Insights into the Complex Biology and Heterogeneity of AD
- Enabling Precision Medicine for AD
- Translational Tools and Infrastructure to Enable Predictive Drug Development
- Emerging Therapeutics
- Understanding the Impact of the Environment to Advance Disease Prevention
- Advances in Disease Monitoring, Assessment and Care
- Building an Open Science Research Ecosystem to Accelerate AD Therapy Development
- Central programmatic themes of the 2018 Summit:
- Understanding disease heterogeneity
- Enhancing research rigor, reproducibility and translatability
- Enabling rapid translational learning through open science systems and incentives.
AD Research Summit Program
- Day 1 hosted over 400 in-person attendees and ~2300 online viewers
- Videocast from day 1 is available: https://videocast.nih.gov/summary.asp?Live=26922&bhcp=1
- The Day 2 Program was cancelled due to inclement weather. Day 2 Program rescheduled for May 24 as a live video-cast event.
- DAY 2 of the Program will feature:
- Emerging Therapeutics -- part II
- Understanding the Impact of the Environment to Advance Disease Prevention
- Advances in Disease Monitoring, Assessment and Care
- Building an Open Science Research Ecosystem to Accelerate AD Therapy Development
Additional Save the Date
- ADRD Summit (III)
- March 14-15, 2019
- Bethesda, MD
Co-hosted by the NIA.
April 27, 2018 -- Advisory Council Meeting #28
The meeting was held on Friday, April 27, 2018, in Washington, DC. During the meeting, the Clinical Care Subcommittee took charge of the theme, focusing on advancing consensus on dementia care elements to guide new outcomes measurement. The Council heard speakers in two sessions, one focused on developing consensus about dementia care elements, and the second on models that are informing outcomes measurement. The meeting also included updates on work from the previous meetings, a presentation on the final report from the October 2017 Care Summit, and federal workgroup updates. Material available from this meeting is listed below or at https://aspe.hhs.gov/advisory-council-alzheimers-research-care-and-services-meetings#Apr2018.
Comments and questions, or alerts to broken links, should be sent to napa@hhs.gov.
General Information
-
Agenda -- [HTML Version] [PDF Version]
-
Meeting Announcement -- [HTML Version] [PDF Version]
-
Meeting Summary -- [HTML Version] [PDF Version]
-
Public Comments -- [HTML Version]
Handouts
-
Main Summit Recommendations -- [HTML Version] [PDF Version]
-
National Research Summit on Care, Services, and Supports for Persons with Dementia and Their Caregivers: Report to the National Advisory Council on Alzheimer's Research, Care, and Services -- [HTML Version] [PDF Version]
Presentation Slides
-
Age-Friendly Health Systems -- [HTML Version] [PDF Version]
-
Alzheimer's Disease and Related Dementias Research Update -- [HTML Version] [PDF Version]
-
Care Planning and Health Information Technology: How to Aid Dementia Quality Care -- [HTML Version] [PDF Version]
-
Clinical Care Subcommittee Agenda: Advancing Consensus on Dementia Care Elements to Guide New Outcomes Measurement -- [HTML Version] [PDF Version]
-
Clinical Subcommittee Update -- [HTML Version] [PDF Version]
-
Defining Quality Dementia Care -- [HTML Version] [PDF Version]
-
Final Report to the NAPA Advisory Council -- [HTML Version] [PDF Version]
-
Long-Term Services and Supports Committee Update -- [HTML Version] [PDF Version]
-
Quality Care from the Perspectives of People Living with Dementia -- [HTML Version] [PDF Version]
-
Research Summit on Dementia Care: Building Evidence for Services and Supports Process Report -- [HTML Version] [PDF Version]
-
Testing the Promise of Primary Care: Comprehensive Primary Care Plus (CPC+) -- [HTML Version] [PDF Version]
-
Updates and Follow-Up from January Meeting -- [HTML Version] [PDF Version]
