Friday, October 27, 2017
Printer Friendly Version in PDF Format (8 PDF pages)
Research Progress for Alzheimer's Disease and Related Dementias
Richard J. Hodes, M.D.
Director
National Institute on Aging
Program integration toward achieving the 2025 goal of NAPA (from discovery to the clinic)
- Basic R01's, ADSP, NCRAD
- Discovery Stage
- Identify Target
- Identify Compounds
- Discovery Stage
- AMP-AD, MOVE2
- Discovery Stage
- Establish Activity
- Select Clinical Candidates
- Discovery Stage
- Translational Animal center
- Development Stage
- Test Safety
- Development Stage
- AD Centers, NACC, ADNI/Biomarkes
- Development Stage
- Submit Application for Clinical Trial (IND/CTX/CTA)
- Human Clinical Trial Phase I
- Human Clinical Trial Phase II
- Development Stage
- AD Centers, AD Clinical Trials, Consortium
- Development Stage
- Submit Registration for Regulatory Approval (NDA/BLA/MAA)
- Human Clinical Trial Phase III
- Human Clinical Trial Phase IV
- Development Stage
NIA-AD accomplishments FY16-17
- R01's
- Who
- Alzheimer's Disease Genetics Consortium (ADGC) [RENEWED FY16 AND 17]
- National Institute on Aging Genetics of Alzheimer's Disease Data Storage Site (NIAGADS) [RENEWED FY16 AND 17]
- National Cell Repository for Alzheimer's Disease (NCRAD) [RENEWED FY16 AND 17]
- R01's [NEW PROGRAM]
- Inflam
- ApoE
- Stem cells
- Synapses
- Genetics
- Gau
- Abeta
- Who
- AMP-AD, MOVE2 [RENEWED FY16 AND 17]
- Who
- Alzheimer's Disease Sequencing Project (ADSP) [RENEWED FY16 AND 17]
- Accelerating Medicines Partnership
- Who
- Translational MODEL-AD [NEW PROGRAM]
- Who
- NIH Blueprint for Neuroscience Research
- Who
- Biomarkes
- Who
- National Alzheimer's Coordinating Center (NACC)
- Alzheimer's Disease Neuroimaging Initiative (ADNI) [RENEWED FY16 AND 17]
- Dominantly Inherited Alzheimer Network (DIAN)
- R01's [NEW PROGRAM]
- Who
- AD Centers [NEW PROGRAM]
- Who
- Trial-Ready Cohort for Preclinical/Prodromal Alzheimer's Disease (TRC-PAD) [NEW PROGRAM]
- Alzheimer's Disease Cooperative Study (ADCS)
- Alzheimer's Prevention Initiative [RENEWED FY16 AND 17]
- Dominantly Inherited Alzheimer Network Trials Unit (DIAN TU) [RENEWED FY16 AND 17]
- ACTC [NEW PROGRAM]
- Wake Forest University
- Michigan University
- BU
- Who
Behavioral and Social Approaches to AD/ADRD Research
| Research Area | Example Topics |
|---|---|
| Dementia Care |
|
| Caregiver Research and Interventions |
|
| Epidemiology |
|
| Behavioral and Social Pathways to ADRD |
|
| Early Psychological Changes in AD |
|
| Prevention |
|
| Disparities |
|
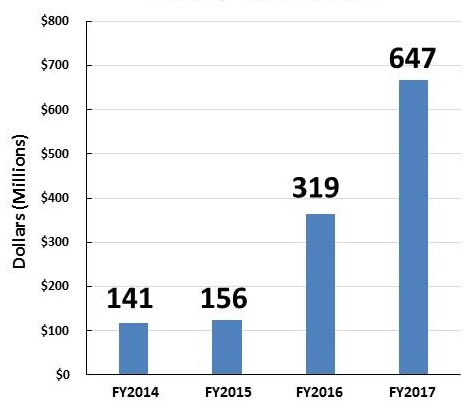
AD and ADRD Research Investments FY14-FY17
| Individual Research Grants | Clinical Trials |
|---|---|
 |
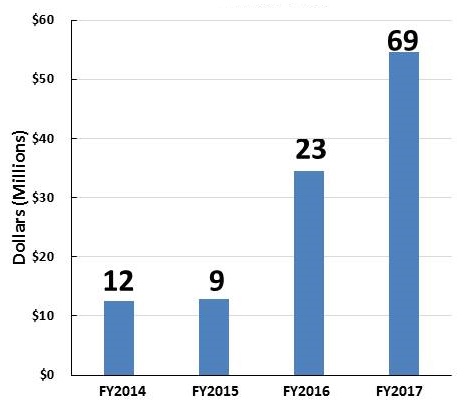 |
| These figures show grants awarded with NIA funding. Some of the grants are in response to initiatives sponsored by NIH ICs other than NIA (e.g., NINDS, NIBIB, NIEHS, NIDCD). | |
Interactions among NIA sponsored centers
 |
Opportunities for Small Business -- AD/ADRD specific
- Advancing Research on Alzheimer's Disease (AD) and Alzheimer's-Disease-Related Dementias (ADRD) (R41/R42/R43/R44)
- Tools for Clinical Care and Management of Alzheimer's Disease (AD) and its Comorbidities (R41/R42/R43/R44)
- Development of Socially-Assistive Robots (SARs) to Engage Persons with Alzheimer's Disease (AD) and AD-Related Dementias (ADRD), and their Caregivers (R41/R42/R43/R44)
NIA SBIR/STTR Obligations
| $ in Millions |
|---|
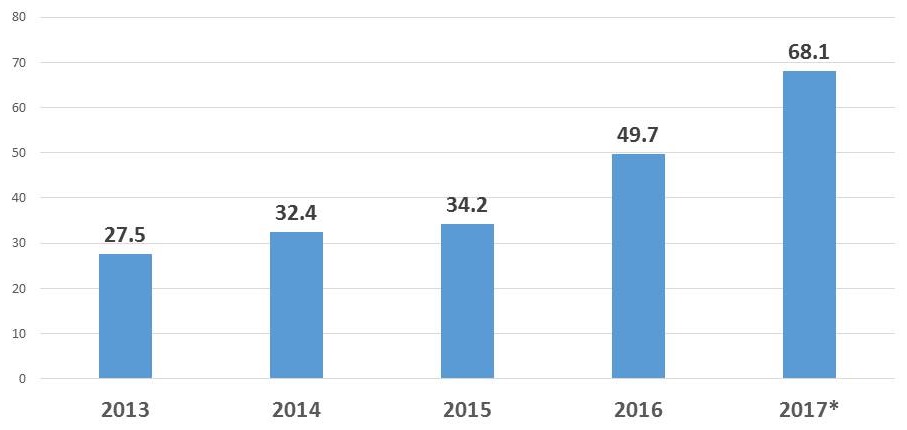 |
| * Estimate |
Save the Dates!
- Alzheimer's Disease Research Summit (III), March 1-2, 2018, Bethesda, MD
- ADRD Summit (III), March 14-15, 2019, Bethesda, MD
Other Federal Updates
7 Grants Awarded for FY2017 NINDS Funding Opportunity Announcements
- 2 Awards for Leveraging Existing Resources (i.e., ADNI/NACC, Parkinson's Disease Biomarker Program) for Research on Lewy Body Dementia (R03)
- Whole-Methylome Sequencing to Identify Unique Epigenetic Profiles in Lewy Body Dementias (Debby W. Tsuang; Seattle Institute for Biomedical/Clinical research)
- Applying DNA Methylation for Differential Diagnosis in Lewy Body Dementia (Paula A. Desplats; UCSF)
- 2 Awards for Frontotemporal Degeneration (FTD) Sequencing Consortium: Discovery, Replication, and Validation (UG3/UH3)
- Impact of Coding and Non-coding Variation in Progressive Supranuclear Palsy (Giovanni Coppola et al.; UCLA)
- Whole Genome Sequencing Consortium on Frontotemporal Dementia with Underlying TDP-43 Pathology (Rosa Rademakers; Mayo Clinic Jacksonville)
- 3 Awards for Detecting Cognitive Impairment, Including Dementia, in Primary Care and Other Everyday Clinical Settings for the General Public and in Health Disparities Populations (UG3/UH3)
- Mycog -- Rapid Detection of Cognitive Impairment in Everyday Clinical Settings (Richard Gershon et al.; Northwestern)
- The UCSF Brain Health Assessment for the Detection of Cognitive Impairment Among Diverse Populations in Primary Care (Katherine L. Possin; UCSF)
- 5-Cog Battery to Improve Detection of Cognitive Impairment and Dementia (Joe Verghese; Albert Einstein College of Medicine)
VA Research Update
- U.S. Department of Veterans Affairs (VA) partnering with National Institute on Aging(NIA) to foster new networks & leverage resources, funding in Alzheimer's Disease (AD) and Alzheimer's Disease Related Dementias (ADRD).
- Request for information (RFI) issued last year: https://grants.nih.gov/grants/guide/notice-files/NOT-AG-16-083.html.
- Strategic Partnership for Advancing Research and Knowledge (SPARK)-Dementia Research, now forming workgroups around:
- Basic mechanisms and pathogenesis of AD and ADRD;
- Genetic and population studies;
- Diagnosis and disease monitoring;
- Translational and clinical research;
- Care and caregiver research.
October 27, 2017 -- Advisory Council Meeting #26
The meeting was held on Friday, October 27, 2017, in Washington, DC. The Advisory Council welcomed its new members and invited them to share their experiences and where they see the Council going over the length of their terms. The Advisory Council also spent some time discussing the process of developing recommendations and how those recommendations relate to the National Plan. The Council then spent much of the meeting discussing the National Research Summit on Care, Services, and Supports for Persons with Dementia and Their Caregivers, held on October 16-17. Material available from this meeting is listed below and at https://aspe.hhs.gov/advisory-council-alzheimers-research-care-and-services-meetings#Oct2017.
Comments and questions, or alerts to broken links, should be sent to napa@hhs.gov.
General Information
-
Agenda -- [HTML Version] [PDF Version]
-
Meeting Announcement -- [HTML Version] [PDF Version]
-
Meeting Summary -- [HTML Version] [PDF Version]
-
Public Comments -- [HTML Version]
Handouts
-
National Plan to Address Alzheimer's Disease: 2017 Update -- [HTML Version] [PDF Version]
-
Public Members of the Advisory Council on Alzheimer's Research, Care and Services: 2017 Recommendations -- [HTML Version] [PDF Version]
Presentation Slides
-
2017 National Plan -- [HTML Version] [PDF Version]
-
Before NAPA: IADRP -- [HTML Version] [PDF Version]
-
Clinical Care Subcommittee Update -- [HTML Version] [PDF Version]
-
Clinical Subcommittee Update -- [HTML Version] [PDF Version]
-
Developing, Sustaining and Taking Innovations to Scale -- [HTML Version] [PDF Version]
-
Discussion of Future Meetings -- [HTML Version] [PDF Version]
-
Long-Term Services and Supports Committee Update -- [HTML Version] [PDF Version]
-
Overview of the NAPA Legislation and the Charge to the Council -- [HTML Version] [PDF Version]
-
Overview of the National Plan and the Process for Recommendations -- [HTML Version] [PDF Version]
-
Research Progress for Alzheimer's Disease and Related Dementias -- [HTML Version] [PDF Version]
-
Research Summit on Dementia Care Overview and Recommendations -- [HTML Version] [PDF Version]
Videos
-
Welcome through Overviews -- [Video]
-
Subcommittees and Fed Workgroups and Future Meeting Discussion -- [Video]
-
Care Summit Overview and Presentation of Recommendations -- [Video]
-
Discussion through lunch -- [Video]
-
Public Comments -- [Video]
-
Marie Schall Presentation and 2017 National Plan Discussion -- [Video]
-
Federal Workgroup Updates through adjourn -- [Video]
