Friday, February 3, 2017
Printer Friendly Version in PDF Format (14 PDF pages)
Strategies to facilitate recruitment and screening for Alzheimer's clinical trials
Rachel L. Nosheny, Ph.D.
UCSF Department of Radiology and Biomedical Imaging
Co-investigator
Alzheimer's Patient & Caregiver-Powered Research Network
- Funded by:
- PCORNET
- The National Patient-Centered Clinical Research Network
- A collaboration between:
- Mayo Clinic
- Univesity of Florida
- USAgainst Alzheimer's
- BrainHealth Registry
- Leadership:
- Principal Investigator
- Ron Petersen, Mayo Clinic
- Co-Investigators
- Meryl Comer, Geoffrey Beene Foundation
- Michael Weiner, UCSF, Brain health Registry
- Glenn Smith, University of Florida
- Alex Alexander, Mayo Clinic
AD-PCPRN Goal and Specific Aims
- Overall goal: To accelerate development of effective treatments for Alzheimer's disease and related dementias
- Specific Aims
- Connect patients, caregivers, and researchers focused on developing, conducting, and disseminating patient-centered dementia research
- Recruit, engage, and educate individuals at risk for or with Alzheimer's Disease and related dementias and caregivers in a patient-centered network
- Screen, refer, and enroll individuals in Alzheimer's trials and observational studies
- Increase diversity in Alzheimer's trials through outreach to minority populations, especially African Americans and Latinos
AD-PCPRN Structure
 |
BrainHealthRegistry.org:Web portal for recruitment, engagement & longitudinal monitoring
 |
| http://brainhealthregistry.org/ |
Brain Health Registry: How It Works
- Sign up
- Register, consent online
- Tell us about yourself
- 13 questionnaires (health history, family history, medications, memory problems, mood, lifestyle)
- Take some tests
- 3 Online cognitive tests
- Invite a study partner
- Study Partner can separately register and answer questions about participant and self
- Come back...and come back again
- Participants and study partners return every 6 months to complete follow-up tasks
AD-PCPRN participants in the Brain Health Registry
- Total of 30,944 participants enrolled
- Age 55+
- Interested in learning about future research opportunities
- Longitudinal data collected from 12,975
- Retention rate of ~40% over 2 years of follow up
- 31% complete all tasks
- 64% provide enough information to assess whether they are likely to be eligible for AD trials
Geographic distribution of participants
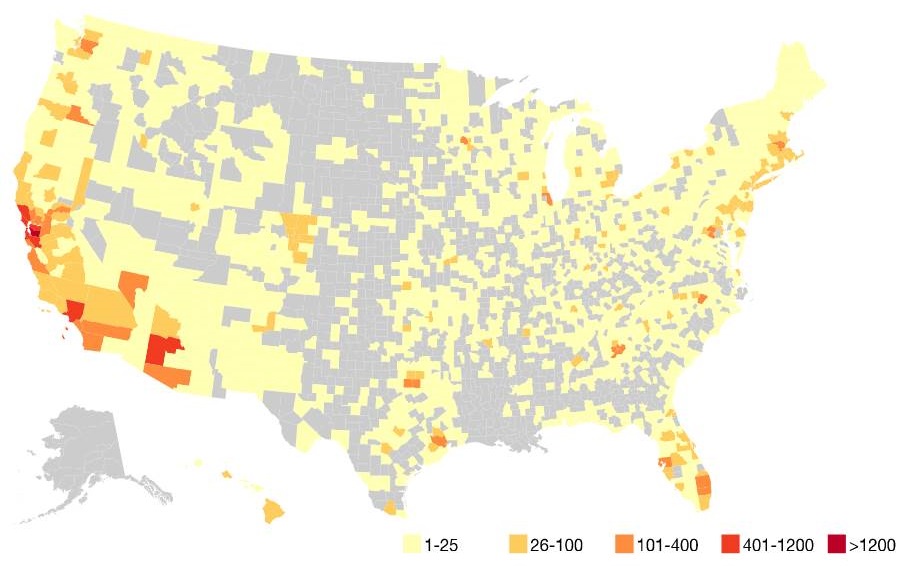 |
Demographics
| Age Distribution |
|---|
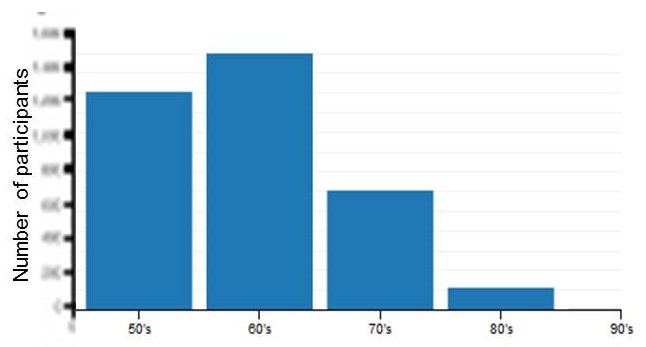 |
| Gender |
|---|
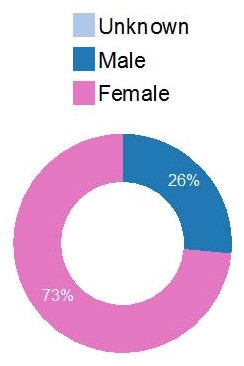 |
Race & Ethnicity
- 4% African American
- 2.1% Asian
- 86.7% Caucasian
- 1.2% Declined To state
- 2.8% Mixed
- 0.3 Native American
- 2.3% Other
- 0.1% Pacific Islander
 |
Educational Attainment
- 2.2% Grammar School
- 5.8% High School
- 18.3% Some College
- 8.5% 2 Year Degree
- 28.8% 4 Year Degree
- 25.1% Masters Degree
- 5.1% Doctoral Degree
- 5.8% Professional Degree
Alzheimer's risk factors
| Family History of Alzheimer's |
|---|
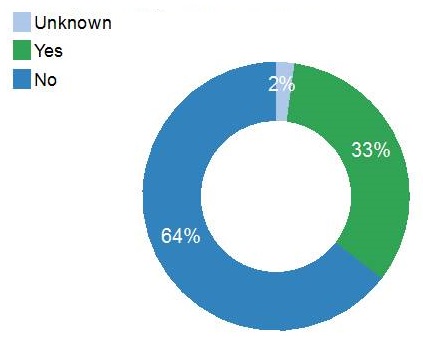 |
| Memory Concern |
|---|
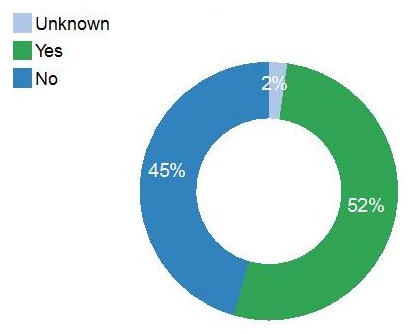 |
How does the AD-PCPRN facilitate AD clinical study recruitment?
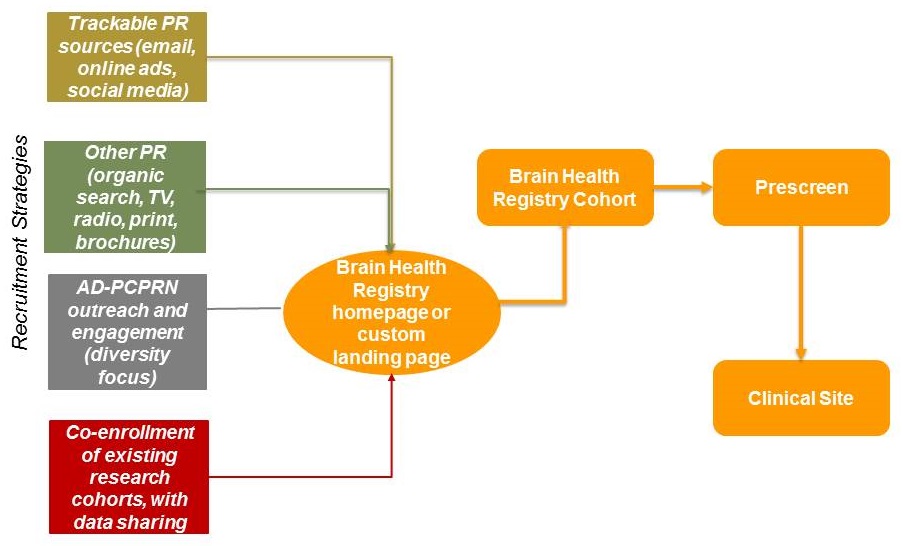 |
Prescreening participants for AD clinical trials
 |
ADPCPRN participants likely to be eligible for Alzheimer's trials
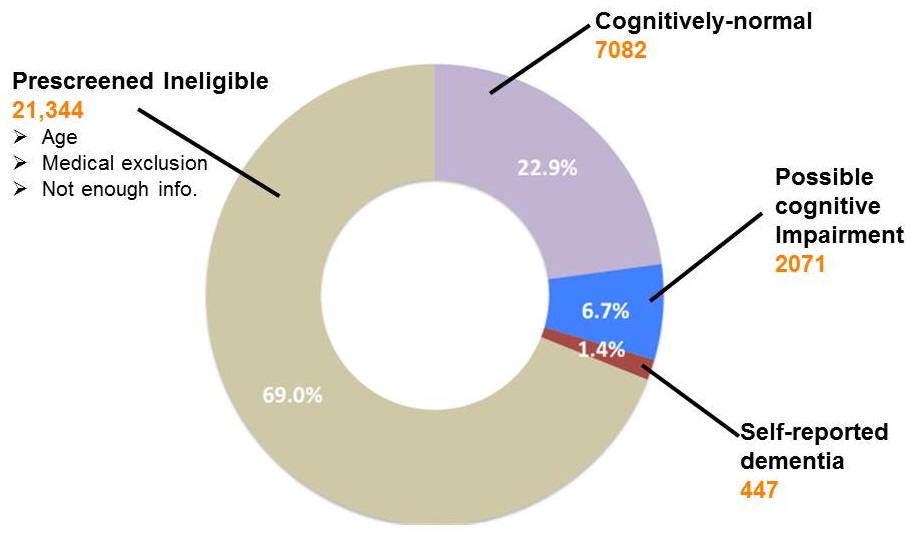 |
Referral of AD-PCPRN participants to clinical studies
- 10,001 participants have been referred to 10 Alzheimer's and aging clinical studies
- Large, observational studies (late life depression, healthy aging, olfaction)
- Randomized treatment trials
- Validation study of online tests
- 757 participants have been enrolled in studies
- Vast majority are enrolled in observational or validation studies
- A major challenge has been to successfully enroll in treatment trials
New strategies to maximize enrollment in trials
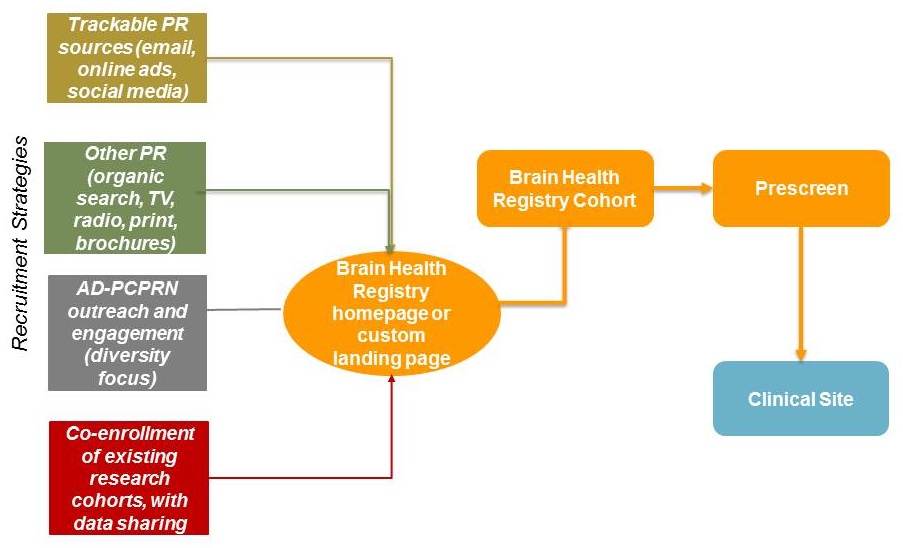 |
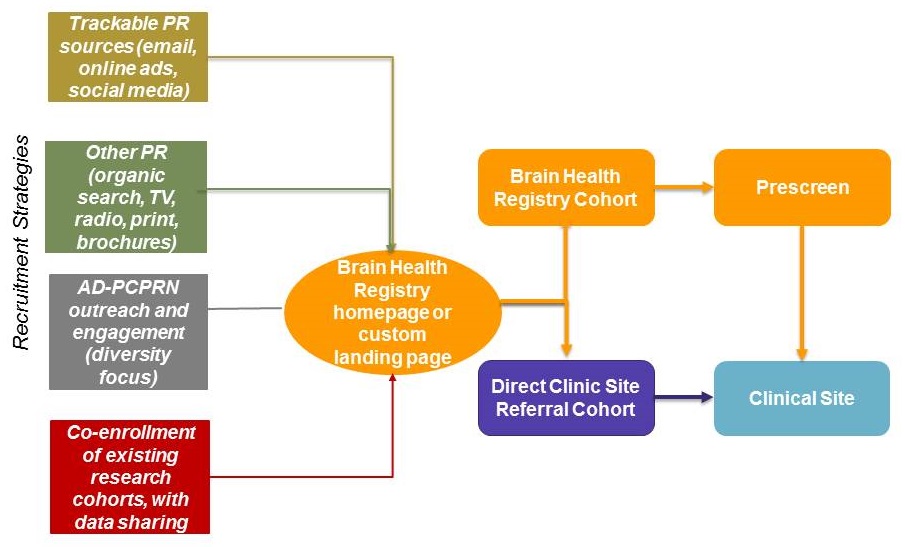 |
 |
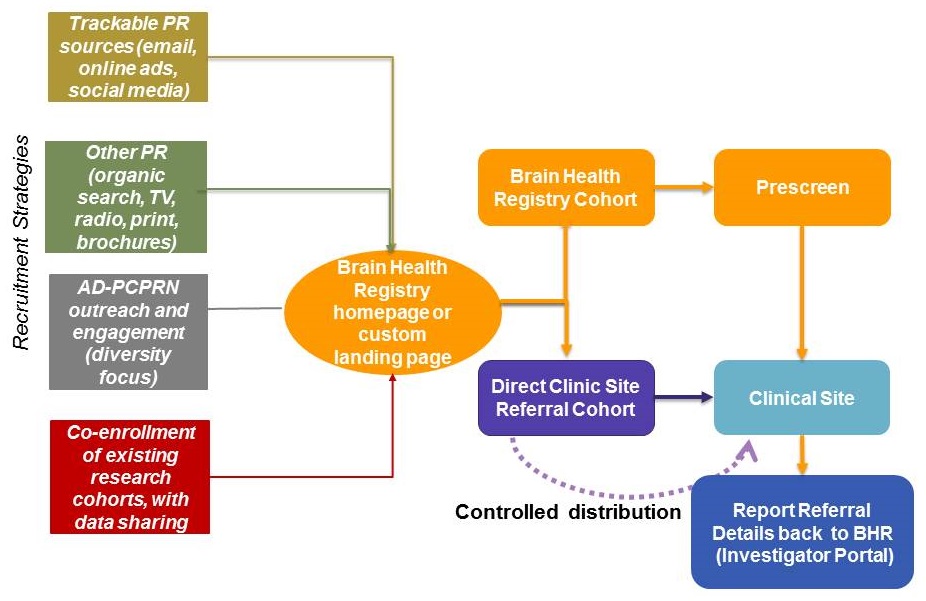 |
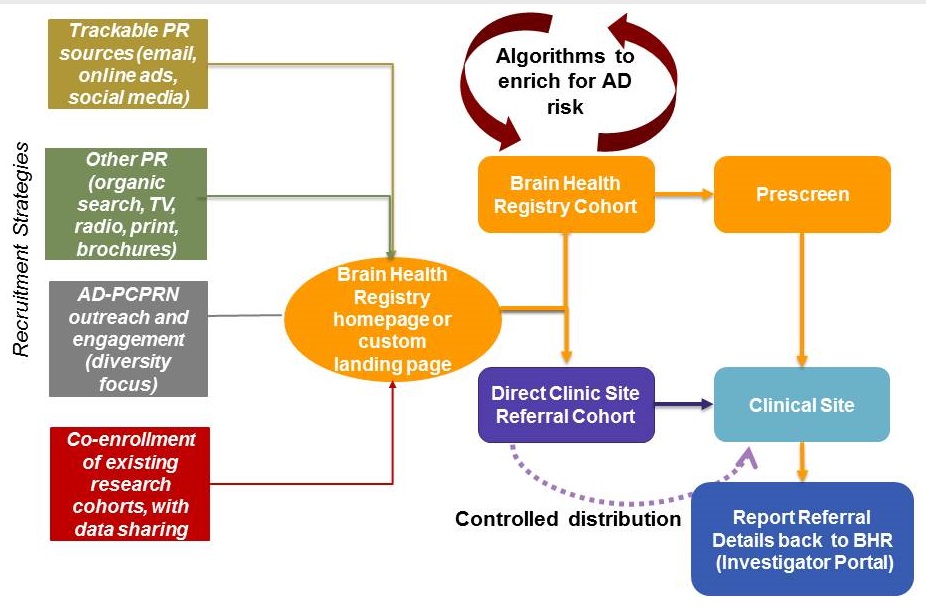 |
Facilitating recruitment through study partner engagement: The Brain Health Registry Caregiver & Study Partner Portal
 |
| http://brainhealthregistry.org/studies/caspp |
Brain Health Registry Caregiver and Study Partner Portal (CASPP)
- Infrastructure & database within BHR for a study partner to separately register and consent online
- Study partners answer questions about the participant's cognitive and daily functioning
- Unique advantages of study-partner reported information
- Associated with many disease-related outcomes
- May sometimes be more accurate than self-report
- Can identify change in function using a single data point
- Focuses on outcomes that matter to patients & families
- Can help overcome patient barriers to participation
- May help recruit participants who are too impaired to join themselves
- Study partners also report on their own health and caregiver experience
- Improve caregiver health
Demographics of AD-PCPRN Study Partners
Total: 1895 Study Partners
| Age Distribution |
|---|
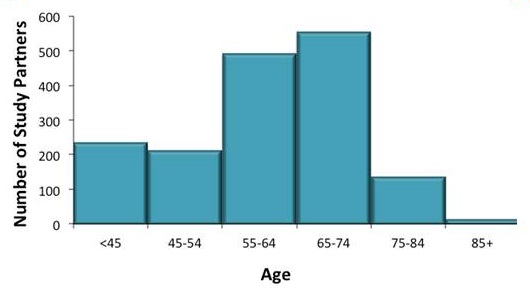 |
| Gender |
|---|
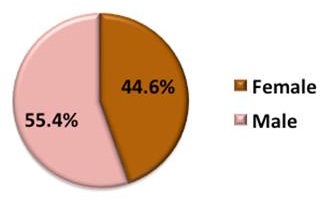 |
| Relationship to Participate |
|---|
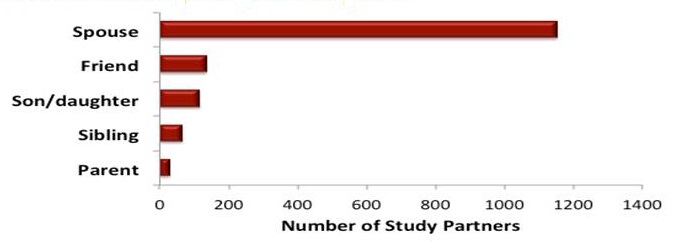 |
| Caregiver Status |
|---|
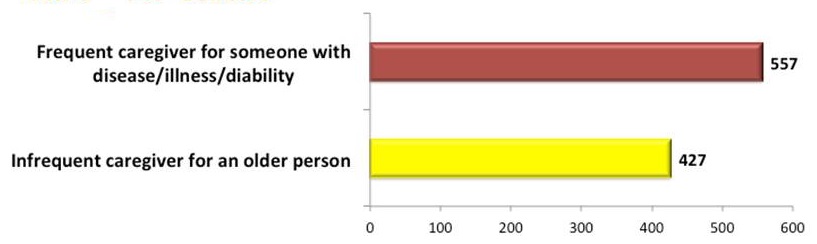 |
Characteristics of BHR Study Partners: Report of health/cognitive status
| About the participant | |
|---|---|
| Report of participant status | |
| Concern about participant memory | 9.5% |
| Participant cognitive change | 15.6% |
| Participant IADL change | 10.5% |
| Participant MCI | 2.0% |
| Participant AD | 0.9% |
| About the study partner him/herself | |
| Self-report health | |
| Memory concern | 34.7% |
| Positive stress screening question | 6.3% |
| Positive mood screening question | 15.4% |
Online study partner-reported data provides valid information about participant function
- Study partner-reported variables are significantly associated with:
- self-reported subjective memory complaints
- Self-reported recent changes in memory and daily functioning
- Online memory test scores of the participant
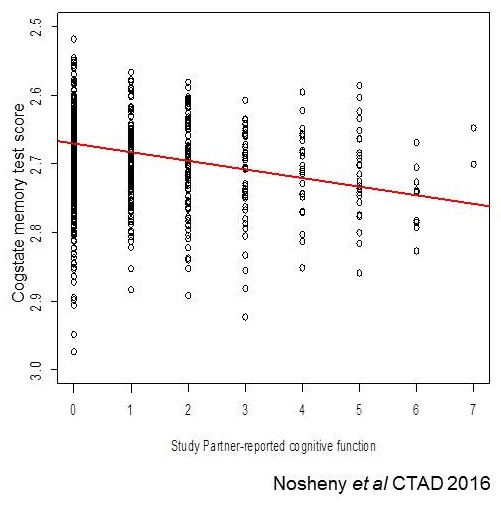 |
Summary & Future Directions
- Overall Goal of the AD-PCPRN is to facilitate Alzheimer's clinical research
- To date
- Over 30,000 participants, 1800 study partners, and 800 caregivers are enrolled in AD-PCPRN through the Brain Health Registry
- Over 700 participants have been enrolled in clinical research studies
- Engagement efforts are reaching many more, with a focus on increasing diversity
- Major challenges are successful enrollment in to treatment trials and recruitment of minorities
- In the future, the AD-PCPRN will aim to
- Increase enrollment and engagement of participants
- Optimize referral to studies by analysis of success rates
- Conduct research to advance recruitment science
Alzheimer's Patient & Caregiver-Powered Research Network
- Partners:
- Mayo Clinic
- Univesity of Florida
- USAgainst Alzheimer's
- BrainHealth Registry
- Leadership:
- Ronald C. Petersen, MD, PhD, Mayo Clinic
- Alex Alexander, MBA, MPA, Mayo Clinic
- Meryl Comer, Geoffrey Beene Foundation
- Glenn Smith, PhD, University of Florida
- Michael W. Weiner, MD, UCSF
- Governance Board:
- all investigators +
- Debra Lappin, FaegreBD Consulting
- Rachel Nosheny, UCSF, Brain Health Registry
- Sally Sacher, Us Against Alzheimer's
- George Vradenburg, Global Alzheimer's Platform Foundation
February 3, 2017 -- Advisory Council Meeting #23
The meeting was held on Friday, February 3, 2017, in Washington, DC. The theme of this Advisory Council meeting was clinical trials for Alzheimer's disease and related dementias and recruitment challenges. Additional afternoon presentations included updates on progress towards a Care and Services Summit, federal workgroup updates, and preparation for the Advisory Council's 2017 Recommendations, due in April 2017. Material available from this meeting is listed below and is also available at https://aspe.hhs.gov/advisory-council-alzheimers-research-care-and-services-meetings#Feb2017.
Comments and questions, or alerts to broken links, should be sent to napa@hhs.gov.
General Information
-
Agenda -- [HTML Version] [PDF Version]
-
Meeting Announcement -- [HTML Version] [PDF Version]
-
Meeting Summary -- [HTML Version] [PDF Version]
-
Public Comments -- [HTML Version]
Handouts
-
Panelist Bios -- [HTML Version] [PDF Version]
Presentation Slides
-
2017 Recommendations -- [HTML Version] [PDF Version]
-
Alzheimer's Association TrialMatch -- [HTML Version] [PDF Version]
-
Clinical Services Subcommittee Federal Update -- [HTML Version] [PDF Version]
-
Clinical Studies Recruitment Strategy -- [HTML Version] [PDF Version]
-
Development and Use of Registries to Accelerate Enrollment into Alzhiemer's Trials -- [HTML Version] [PDF Version]
-
Long-Term Services and Supports Committee Update -- [HTML Version] [PDF Version]
-
National Recruitment Strategy -- [HTML Version] [PDF Version]
-
NINDS Update -- [HTML Version] [PDF Version]
-
Randomized Controlled Trials for Alzheimer's Disease -- [HTML Version] [PDF Version]
-
Strategies to Facilitate Recruitment and Screening for Alzheimer's Clinical Trials -- [HTML Version] [PDF Version]
-
Transforming AD Therapy Development -- [HTML Version] [PDF Version]
-
Update National Research Summit on Care, Services and Supports for Persons with Dementia and Their Caregivers -- [HTML Version] [PDF Version]
