Friday, July 28, 2017
Printer Friendly Version in PDF Format (8 PDF pages)
Development of the FY19 NIH Bypass Budget for Alzheimer's Disease and Related Dementias
Richard J. Hodes, M.D.
Director
National Institute on Aging
Growth of AD and ADRD Research Spending at NIH
| Research/Disease Areas (Dollars in millions and rounded) |
FY 2015 Actual |
FY 2016 Actual |
FY 2017 Estimate (Enacted) |
|---|---|---|---|
| Alzheimer's Disease | $589 | $929 | $1,348 |
| Alzheimer's Disease including Alzheimer's Disease Related Dementias (AD/ADRD) | $631 | $986 | $1,414 |
| Alzheimer's Disease Related Dementias (ADRD) | $120 | $175 | $234 |
| Frontotemporal Dementia (FTD) | $36 | $65 | $68 |
| Lewy Body Dementia | $15 | $22 | $23 |
| Vascular Cognitive Impairment/ Dementia | $72 | $89 | $93 |
Bypass Budget Language
SEC. 230. Hereafter, for each fiscal year through fiscal year 2025, the Director of the National Institutes of Health shall prepare and submit directly to the President for review and transmittal to Congress, after reasonable opportunity for comment, but without change, by the Secretary of Health and Human Services and the Advisory Council on Alzheimer's Research, Care, and Services, an annual budget estimate (including an estimate of the number and type of personnel needs for the Institutes) for the initiatives of the National Institutes of Health pursuant to the National Alzheimer's Plan, as required under section 2(d)(2) of Public Law 111-375.
NIH Meetings Provided the Basis for Priorities, Milestones, and Budget Estimates
- 2012 Alzheimer's Disease Research Summit
- 2013 meeting on Alzheimer's Disease-Related Dementias: Research Challenges and Opportunities
- 2013 meeting on Advancing Treatment for Alzheimer's Disease in Individuals with Down Syndrome
- 2015 Alzheimer's Disease Research Summit
- 2016 meeting on Alzheimer's Disease-Related Dementias
Recommendations used to create milestones for developing the Alzheimer's Disease (and related dementias) Bypass Budget
Combined External and Internal Input -- FY19 AD Bypass Budget
- Input at 2012-2017 meetings:
- Academic research community
- Industry
- Non-governmental organizations
- Other Input:
- 2017 Request for Information
- Academic research community
- Development of comprehensive milestones (NIH staff) -- based on recommendations and milestones from meetings
- Trans-NIH (including NINDS) staff discussion; milestones edited to ensure comprehensive inclusion of priorities for FY19
- NIH staff "price" the milestones
- Final budget estimate for FY19
Trans-NIH Input for FY19 ADBB
- Nine Institutes and Centers provided feedback that contributed to the development of this year's professional judgment budget:
- NIA
- NINDS
- NIMH
- NINR
- NICHD
- NIEHS
- NIDDK
- NIDCR
- NHLBI
Using CADRO as a Framework
- The eight CADRO (Common Alzheimer's Disease Research Ontology) categories provide the overarching framework for the FY19 AD Bypass Budget and narrative.
- CADRO provides the framework for IADRP (the International Alzheimer's Disease Research Portfolio) and will allow tracking of implementation in the budget areas in future years.
FY19 Professional Judgment Budget
| Area of Research | Amount (dollars in thousands) |
|---|---|
| A. Molecular Pathogenesis and Physiology of Alzheimer's Disease | $110,000 |
| B. Diagnosis, Assessment and Disease Monitoring | $70,100 |
| C. Translational Research and Clinical Interventions | $191,000 |
| D. Epidemiology | $58,025 |
| E. Care and Caregiver Support | $32,800 |
| F. Research Resources | $76,375 |
| H. Alzheimer's Disease-Related Dementias | $55,000 |
| Staff Needs, Support, and Misc. | $3,800 |
| Total Additional Resources Needed | $597,100 |
Distribution of Additional Budget Funding Request Across Research Areas, FY19(Total: $597,100,000)
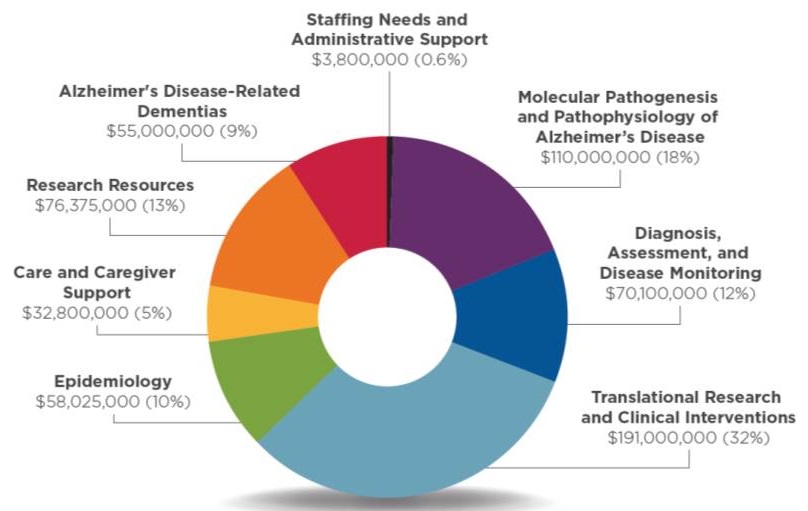 |
FY19 Professional Judgment Budget Baseline
| Baseline Estimate, President's Budget, Fiscal Year 2018 (dollars in thousands) Alzheimer's Disease including Alzheimer's Disease Related Dementias (AD+ADRD) |
$837,000 |
|---|
FY19 Professional Judgment Budget Considerations
| Resources needed for new AD/ADRD research (dollars in thousands) |
$597,100 |
|---|---|
| Difference between FY18 President's Budget Request and the FY17 Omnibus Appropriation (dollars in thousands) |
$577,000 |
| TOTAL ADDITIONAL RESOURCES NEEDED (dollars in thousands) |
$1,174,100 |
| TOTAL RESOURCES NEEDED (dollars in thousands) |
$2,011,100 |
Implementation Tracking
- IADRP: http://iadrp.nia.nih.gov/
- Will continue to offer detailed tracking of initiatives and awards with respect to our research milestones, under the CADRO categories
- Web-based tool for tracking funding initiatives and activities: https://www.nia.nih.gov/alzheimers/milestones
- Developed by the NIH and other AD funding organizations
- Aimed at addressing the research milestones associated with NAPA
Other Federal Updates
ADRD Non-Governmental Organizations (NGO) Roundtable Meeting
June 16, 2017
Hosted by NINDS and NIA
- To share updates about research activities, funding-related information, and progress on the ADRD Research Milestones
- To discuss opportunities for NIH and NGOs to work together toward preventing or effectively treating AD/ADRD by 2025
- Participants: NINDS and NIA Leadership and staff, and 9 NGOs, including
- Alzheimer's Association
- American Heart Association & American Stroke Association
- Association for Frontotemporal Degeneration
- The Bluefield Project to Cure Frontotemporal Dementia
- BrightFocus Foundation
- CurePSP
- Lewy Body Dementia Association
- The Michael J. Fox Foundation for Parkinson's Research
- Rainwater Charitable Foundation
2017 VHA-NIA Collaboration/Partnership Alzheimer's Disease and Related Disorders
- Goals:
- Foster new networks between VA- and NIA- investigators.
- Leverage each Institution's state-of-the-art instrumentation, data information, clinical resources, and Centers of Excellence to synergistically move the field forward.
- Timelines (in process):
- Request for Information (RFI)- NOT-AG-16-083 (9-received)
- Potential AD and ADRD research topics to address.
- Barriers & challenges that could prevent coordination and collaboration.
- How to leverage resources
- Meeting between VA point of contact (Dr. Block) and NIA (Drs. Opanashuk and Liggins) points of contact (2-29-17).
- Development of Work Groups (April 15, 2017) - ~10 people/group
- Basic Disease Mechanisms Work Group
- Genetic and Population Studies Work Group
- Diagnoses and Disease monitoring Work Group
- Translation and Clinical Research Work Group
- Care and Caregiving Research Work Group
- Request for Information (RFI)- NOT-AG-16-083 (9-received)
July 28, 2017 -- Advisory Council Meeting #25
The meeting was held on Friday, January 26, 2018, in Washington, DC. The Research Subcommittee took charge of this meeting's theme, focusing on the process from targets to treatments. The Council heard speakers on the preclinical pipeline, the clinical trial pipeline, and the industry perspective. The meeting also included discussion of a driver diagram to guide the Council's future work, updates and a report from the October Care Summit, and federal workgroup updates. Material available from this meeting is listed below and is also available at https://aspe.hhs.gov/advisory-council-alzheimers-research-care-and-services-meetings#Jan2018.
Comments and questions, or alerts to broken links, should be sent to napa@hhs.gov.
General Information
-
Agenda -- [HTML Version] [PDF Version]
-
Meeting Announcement -- [HTML Version] [PDF Version]
-
Meeting Summary -- [HTML Version] [PDF Version]
-
Public Comments -- [HTML Version]
Presentation Slides
-
2017 National Plan Update and Non-Federal Recommendations -- [HTML Version] [PDF Version]
-
Clinical Services Subcommittee Federal Update -- [HTML Version] [PDF Version]
-
CommunityRx for Community-Residing People with Dementia and Their Caregivers -- [HTML Version] [PDF Version]
-
Dementia Caregiving in the U.S. -- [HTML Version] [PDF Version]
-
Development of the FY 19 NIH Bypass Budget for Alzheimer's Disease and Related Dementias -- [HTML Version] [PDF Version]
-
Implications of a Biologically Based Definition of Alzheimer's Disease -- [HTML Version] [PDF Version]
-
Increasing Opportunities for Choice and Control for Persons with Dementia -- [HTML Version] [PDF Version]
-
Interventions to Prevent or Slow Cognitive Decline, MCI and Dementia in Individuals without Dementia -- [HTML Version] [PDF Version]
-
Living Alone With Dementia -- [HTML Version] [PDF Version]
-
Long-Term Services and Supports Committee Update -- [HTML Version] [PDF Version]
-
Managing Chronic Conditions in People Living with Dementia -- [HTML Version] [PDF Version]
-
Preventing Cognitive Decline and Dementia -- [HTML Version] [PDF Version]
-
Research Summit on Dementia Care -- [HTML Version] [PDF Version]
-
The Many Challenges of Alzheimer's Disease -- [HTML Version] [PDF Version]
