National Institute of Neurological Disorders and Stroke - September 2020
Printer Friendly Version in PDF Format (21 PDF pages)
Table of Contents
Comments and questions, or alerts to broken links, should be sent to napa@hhs.gov.
Background
On March 14-15, 2019, the National Institute of Neurological Disorders and Stroke (NINDS) hosted the 3rd Alzheimer’s Disease-Related Dementias (ADRD) Research Summit, one of a series of triennial conferences (also including the Alzheimer’s Disease Research Summit and the Dementia Care Summits) designed to respond to the National Plan to Address Alzheimer’s Disease [https://aspe.hhs.gov/national-plans-address-alzheimers-disease]. Committees of research experts and ADRD stakeholders developed the focus areas for the Summit, organized around seven broad topics, including multiple etiology dementias (MED), health disparities (HD), Lewy body dementias (LBD), frontotemporal lobar degeneration (FTLD), vascular contributions to cognitive impairment and dementia (VCID), emerging topics, and nomenclature. Informed by pre-Summit review of the research landscape, iterative Committee discussions over the six months leading up to the Summit, a public request for information (RFI) with 78 responses, and direct public input at the Summit itself, the seven Committees developed milestones that represent national ADRD research priorities. These milestones will inform future NIH AD/ADRD bypass budgets and, as Congressionally appropriated funds become available, corresponding research activities. In addition, the milestones guide the AD/ADRD research community generally, and as such, their broad implementation and execution by various federal, national and international stakeholders will be vital to their impact.
All milestones in this report represent very important research goals. For a research milestone to be included, it must be among the top priorities in its respective field. Each topic committee had the option of proposing up to eight recommendations, with the priorities ranked from #1 to #4. Committees also estimated timeline, i.e. the number of years needed to complete or fully implement each milestone. Neither the timelines nor the order of topics reflect prioritization.
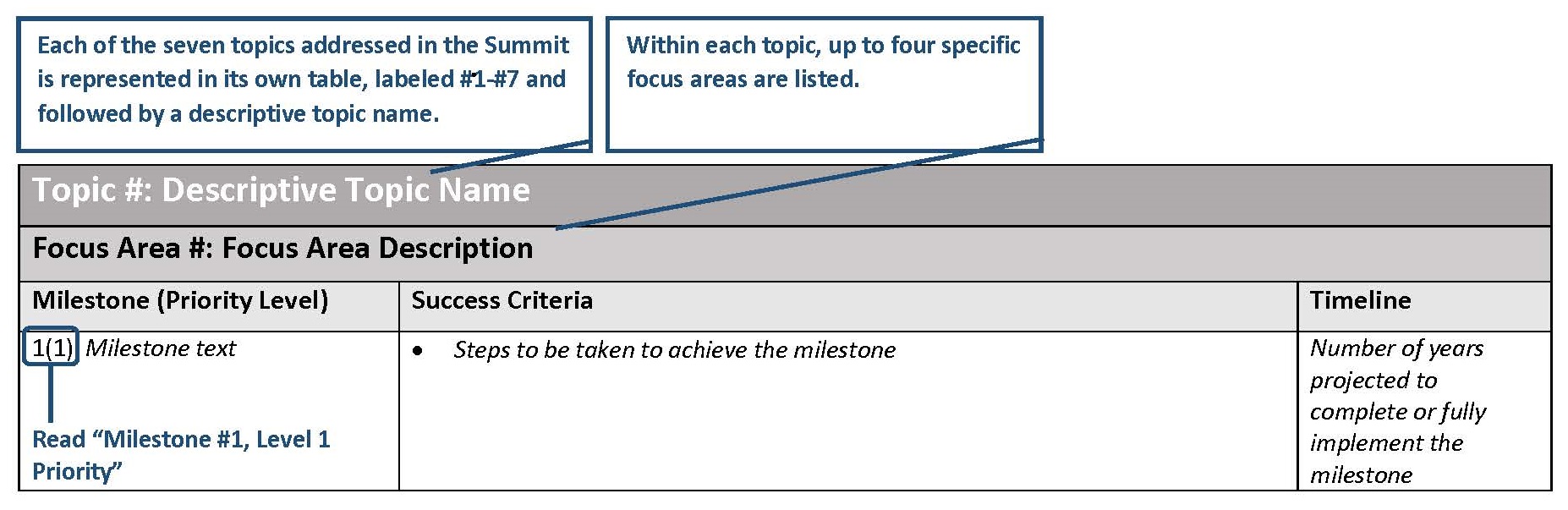
Research Milestone Key
 |
| Topic 1: Multiple Etiology Dementias (MED) |
| Focus Area 1: Improving Detection and Diagnostic Skills in the Community |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 1(1) Detect whether cognitive impairment is objectively present when a patient, care partner or clinician reports cognitive, behavioral or functional changes. |
- Support at least 2 research programs to develop and validate assessment paradigms that address the unmet need to detect cognitive impairment and dementia in large and diverse populations seen in primary care practice and other every day clinical care settings, including in non-English speaking settings.
- Evaluate the success of the resulting paradigms for:
- Coverage of both general and diverse populations in the U.S., including via engagement of local communities in the development of community and culturally informed paradigms.
- The extent to which validated paradigms are reimbursable, time efficient, with easily interpreted results, and useful in diverse primary care settings.
- The extent to which the paradigms include provider guidance for appropriate follow-up, including specialty referrals and caregiver support.
- The extent to which the paradigms have training materials suitable for both physicians and non-physician medical personnel who administer them.
- The extent to which the paradigms are readily accessible and to which there is uptake by both primary caregivers and researchers.
|
3-7 years |
| 2(3) Improve differential diagnosis of symptomatic cognitive impairment. |
- Initiate research to achieve improved and increased differential diagnosis of cognitive impairment and dementia disorders, including using biomarkers when possible, by medical specialists who serve the general public (e.g., but not limited to, neurologists, geriatricians, neuropsychologists and geriatric psychiatrists). This research is to be designed for caregiver buy in and impact in urban, suburban and rural environments, including in populations with health disparities. In addition to differential diagnosis across AD/ADRD, the research must also focus on differential diagnosis of rapidly progressive dementias as well as potentially treatable cognitive impairment and dementia. The research outcomes will include concise and implementable recommendations for medical follow-up based on the results of differential diagnoses.
- Make available effective cognitive impairment and dementia differential diagnosis paradigms to providers, including in community-based settings.
- Conduct an evaluation to determine the economic impact of earlier differential diagnosis on patients and healthcare systems.
|
5-10 years |
| Focus Area 2: Advancing Basic and Clinical Research in MED |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 3(1) Advance basic and clinical research on common mechanisms of multi-etiology cognitive impairment and dementia. |
- Initiate at least one new funding opportunity announcement that promotes understanding of interactions among different pathologies of dementia, e.g. beta-amyloidosis, 3R/4R tauopathy, 4R tauopathy, TDP-43, alpha-synucleinopathy, arteriolosclerosis and other cerebrovascular diseases, as well as other neurodegenerative processes such as TBI or prion diseases.
- Support research in general and diverse U.S. populations focused on identifying molecular pathways that, during aging, either accelerate cognitive decline or protect against cognitive decline and non-cognitive but related symptoms such as gait impairment or physical frailty. This research should be designed to further develop and optimize biomarkers of age-related cognitive impairment and dementia for clinical trials, clinical research, and toward general clinical use.
- Develop research nomenclature for dementia that can clearly and accurately communicate the emerging scientific understanding of multiple etiology dementias both in research and in clinical practice.
|
3-7 years |
| Focus Area 3: Increasing the Dementia Capable Workforce |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 4(2) Increase education and training of health professionals and researchers focused on cognitive impairment and dementia. |
- Establish new programs with equal missions of research and training (MD, PhD, and other professionals) of individuals in the full spectrum of basic through clinical research in AD/ADRD, including in health disparities of AD/ADRD, and who in the future plan to be: (i) basic, basic disease-related, or clinical researchers, (ii) clinicians who lead clinical research or who support clinical research (e.g. by supporting enrollment in clinical trials), (iii) and clinicians who may not be directly involved in research, but who seek AD/ADRD education to be effective for their constituents. For these training programs it is important to include diverse trainees from varied research backgrounds; quantitative research is encouraged and should be reflected in the education of trainees when appropriate (e.g., statistics, bioinformatics, physics).
- Increase training in grant review relevant to cognitive impairment and dementia, including for trainees representative of the diverse U.S. population.
- Support a scholarship program that supports later stage in training health science professionals (MD, PhD, and other relevant health professionals) to attend AD/ADRD Summits.
- Evaluate the impact on the AD/ADRD workforce of training initiatives designed to recruit and retain graduate students, postdoctoral fellows, and/or early-stage investigators in cognitive impairment and dementia research, including from groups underrepresented in the biomedical workforce.
|
5-10 years |
| Focus Area 4: Intervention Studies to Mitigate Reversible Causes of Dementia |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 5(2) Conduct intervention studies to mitigate reversible causes of cognitive dysfunction in persons with or at-risk for cognitive impairment and dementia of aging where etiology may be uncertain or where multiple etiologies appear likely. |
- Establish a national research consortium to perform applied research to test, refine and implement, in primary care and other everyday care settings, effective paradigms for determining whether reversible causes of cognitive dysfunction are present in an individual, and if so, how to facilitate successful intervention.
- Develop and disseminate best practices for primary care and other everyday care settings for determining whether reversible causes of cognitive dysfunction are present in an individual.
|
3-7 years |
| Focus Area 5: Research to Implement Effective Dementia Care |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 6(3) Bridge the science-practice gap by conducting implementation research with the goal of better and more widespread implementation of proven dementia care programs that effectively support persons with dementia and their caregivers. |
- Hold a workshop and write a white paper on proven dementia care programs that effectively support persons with dementia and their caregivers, as well as barriers to national implementation of these programs.
- Establish a national consortium tasked with applied research to determine how to implement, widely across the country in everyday care settings, proven care programs that effectively support persons with dementia and their caregivers, including in diverse populations and populations with health disparities.
|
|
| Topic 2: Health Disparities in AD/ADRD |
| Focus Area 1: Assessment |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 1(1) Generate and/or improve cognitive assessment tools for populations facing AD/ADRD health disparities. |
- Initiate at least 2 research programs to develop and validate assessment tools to detect early cognitive changes that occur in AD/ADRD in diverse U.S. populations that face health disparities in cognitive impairment and dementia, including non-English-speaking people and people with varying levels of education.
- Evaluate the success of the resulting assessment tools for:
- Extent of engagement of local communities in the development of community- , diversity- , and culturally-informed tools.
- Incorporation of existing best practices in assessment tool design and contributions to improving best practices for assessing cognitive impairment and dementia in populations facing AD/ADRD health disparities.
- Validation across different cultural groups that face AD/ADRD health disparities.
- Training materials for assessment tool(s) use; guidance regarding which tool(s) to be used for cognitive assessment of any individual.
- Guidance for individual follow-up depending on assessment results, including specialty referrals and caregiver support when appropriate.
- Develop normative references to facilitate harmonized comparisons among cognitive assessments obtained using different tools across diverse populations.
|
1-3 years |
| 2(1) Increase availability and utilization of harmonized culturally- and linguistically valid assessment tools within ongoing and newly generated studies of AD/ADRD and cognitive health intervention trials. |
- Use validated assessment tools designed to detect early cognitive changes in new studies and clinical trials for AD/ADRD that include diverse patient populations experiencing health disparities.
- Facilitate effective health disparities AD/ADRD research by generating a dedicated resource for housing and disseminating a validated minimal core of culturally and linguistically appropriate best practices, paradigms, and tools for assessing cognitive impairment and dementia in diverse populations.
|
1-3 years |
| Focus Area 2: Resolve AD/ADRD Health Disparities by Discovering Culturally Appropriate Pathways to Effective Prevention and Treatments |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 3(2) Test early mechanistic pathways of multiple etiologies that may account for AD/ADRD health disparities and scientifically move forward potential opportunities for precision medicine. |
- Initiate or leverage a longitudinal community-based cohort study of incident cognitive impairment in populations that experience heath disparities burdens in dementia. This research is to be designed to assess the interactions of social, environmental, and molecular mechanisms. Biospecimen and clinical data collection should be embedded in the study design to facilitate wide sharing for research. Studies should incorporate cutting-edge imaging, fluid-based and other biomarkers, autopsy (when possible), and other biospecimens for mechanism-oriented research.
- Support at least two studies investigating whether changes in risk factors for cognitive impairment and dementia occur over the life course in diverse populations. Identify critical periods of life and critical lifestyle and other parameters with respect to cognitive impairment and dementia prevention.
- Support at least two studies investigating whether the prevalence and interaction of AD/ADRD risk factors (e.g., genetic, vascular, behavioral, environmental, or social risks), and their impact on outcomes, differs across populations, including populations that experience health disparities in dementia. Use this information to estimate the highest impact intervention targets (i.e., population burden associated with each risk factor) toward the development of population-specific informed precision medicine for dementia.
- Facilitate data and biospecimen availability for future research.
|
3-7 years |
| 4(2) Implement culturally tailored multimodal intervention trials and drug therapy trials to reduce AD/ADRD burden in populations facing disparities. |
- Initiate one new clinical trial targeting AD/ADRD specifically in a population that experiences health disparities in dementia.
|
3-7 years |
| Focus Area 3: Monitoring Changes in AD/ADRD Disparities |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 5(3) Clarify the epidemiology of health disparities in AD/ADRD prevalence and incidence by documenting and monitoring trends in health disparities in AD/ADRD prevalence and incidence over time. |
- Initiate at least one study that assesses the full spectrum of AD/ADRD (i.e. AD, FTLD, VCID, LBD and mixed dementias) prevalence, incidence, trends over time, and long-term outcomes in diverse populations (e.g. racial/ethnic, socioeconomic, geographic) and in populations that experience health disparities. To ensure valid estimates this study is to be embedded within large scale community-based health surveillance systems, potentially including primary care, and must control for confounding factors including but not limited to stigma and under diagnosis. The study must collect participant data across the full range of social determinants of health including race/ethnicity, nativity, primary language, income and wealth, educational background, gender identity and sexual orientation, and geographic location in relevant research programs. Genomic sex and ancestry should be documented separately from self-identified gender and race/ethnicity, as these are different variables.
- Develop and release a report on the results of this study.
|
Ongoing |
| 6(3) Increase policy-relevant research on disparities in access to care, awareness and stigma, and costs of care for persons living with AD/ADRD and their families and caregivers. |
- To inform future health care planning and implementation, initiate at least two new research studies in populations that experience health disparities in dementia that focus on factors that impact access to AD/ADRD care including awareness, stigma, and cost. Determine the potential economic impact, both on healthcare systems and on patients, of earlier diagnosis and care in populations experiencing health disparities.
|
Ongoing |
| Focus Area 4: A Diverse and Inclusive AD/ADRD Workforce |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 7(4) Improve and increase training, including for individuals who are members of under-represented populations, and of different career levels of scholars who conduct health disparities research in AD/ADRD. |
- Develop an initiative to train AD/ADRD scientists, including from groups that are underrepresented in the biomedical workforce (e.g. but not limited to, racial/ethnic groups, individuals with disabilities, sexual and gender minorities, and individuals from disadvantaged backgrounds), in health disparities research. This initiative should offer opportunities for health disparities training to scientists at all career stages from undergraduate through established senior levels. In addition to subject matter-based training, this initiative should also emphasize training in community engagement methods, mentorship in health disparities research, and training in grant review processes used to evaluate AD/ADRD health disparities research applications.
- Establish, as a part of this initiative, evaluation criteria and timelines to determine the trajectory and impact, with built in opportunities to adjust to improve outcomes.
|
3-5 years |
| 8(4) Leverage ongoing initiatives on the science of inclusion to facilitate community engagement, understanding, recruitment, and long-term retention of populations experiencing AD/ADRD health disparities. |
- Support research to develop new community-based strategies (e.g., specific language and outreach mechanisms/venues) to recruit, retain, and engage study participants from diverse populations, based the way each population understands and recognizes AD/ADRD.
- Develop, disseminate and implement a national strategy for AD/ADRD clinical research to increase engagement and retention of diverse AD/ADRD patients and caregivers in clinical research. Refine this strategy on an iterative basis to reflect emerging best practices in participant engagement.
- Track subject recruitment in clinical trials by gender, race/ethnicity, socioeconomic status, geographic region, and other key features that inform the understanding of health disparities and outcomes across diverse populations.
|
3-5 years |
| Topic 3: Dementia Nomenclature |
| Focus Area 1: Dementia Nomenclature Working Groups |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 1(1) Form research, clinical practice and public stakeholder dementia nomenclature working groups. |
- Establish high level working groups of stakeholders to begin discussions that will lead to draft recommendations to develop, refine and clarify dementia nomenclature for research (Research Working Group), clinical practice (Clinical Practice Working Group) and the public (Public Stakeholders Working Group).
- Convene the first in person meeting of each working group within 6 months of its establishment. Each working group is to report its recommendations to the NAPA Council within 1 year of establishment.
|
1-2 years |
| Focus Area 2: Integration and Interoperability of Dementia Nomenclature |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 2(1) Integrate and refine recommendations from the Research, Clinical Practice, and Public Stakeholder Working Groups into standardized, acceptable and accurate nomenclature that works across the spectrum of stakeholders. |
- Integrate dementia nomenclature recommendations from the three working groups to develop an approach to cognitive impairment and dementia nomenclature that uses unambiguous terms that are identical when possible, but at least interoperable with mutually agreed upon meaning, across research, clinical practice, and public stakeholders.
- Increase everyone’s, including the general public’s, understanding of cognitive impairment including dementia diagnoses by using plain and clear language when discussing dementia, without sacrificing accuracy of terminology.
|
2-4 years |
| Topic 4: Lewy Body Dementias (LBD) |
| Focus Area 1: Clinical Science |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 1(1) Prepare for and initiate clinical trials that aim to alleviate or slow the course of LBD symptoms, and delay or prevent the onset of disease. |
- Establish a clinical trial network with the personnel and infrastructure that can carry out LBD clinical trials.
- Fund research for the development and validation of clinical tools to track cognitive changes and other symptoms that occur in LBD.
- Initiate one or more clinical trials that test prospective therapies based on pharmaceutical approaches, gene therapy, regenerative medicine, surgical interventions, or non-pharmacological approaches to prevent or alter LBD processes.
|
1-7 years |
| 2(2) Longitudinal antemortem LBD characterization. |
- Evaluate existing LBD biomarker efforts, including systematic assessments of their impact on identifying pre-dementia LBD and prodromal or at-risk for LBD patients.
- Informed by this evaluation, initiate at least one new study designed to identify pre-dementia LBD and prodromal or at-risk for LBD patients.
- Ensure studies collect and share standardized clinical and neuropsychological data.
|
3-5 years |
| 3(3) Neuroimaging characterization of LBD. |
- Evaluate existing efforts to develop novel imaging markers of LBD, including the feasibility of implementing current candidate imaging markers in clinical trials.
- Informed by this evaluation: (i) standardize neuroimaging to facilitate multicenter studies; and (ii) begin at least one new study to validate available and proposed imaging tools for the prodromal diagnosis, diagnosis, and progression of LBD.
- Support neuroimaging research to develop improved approaches for differential diagnoses of LBD compared to other dementing illnesses in longitudinally followed cohorts ultimately confirmed by autopsy.
|
3-6 years |
| 4(4) Neuropathologic characterization of LBD and use of LBD pathology cohorts. |
- Evaluate existing LBD post-mortem clinical data and tissue resources, as well as existing best practices in neuropathologic evaluation, for their impact and effectiveness in advancing the field.
- Based on this evaluation, determine the best course of action to enhance the acquisition and distribution of post-mortem LBD tissue, including central and peripheral tissue, and corresponding clinical data.
- Develop best practices for patient outreach and education to emphasize the importance of brain donation, as well as the means for providing logistical support and resources to facilitate brain donations.
|
2-7 years |
| Focus Area 2: Basic Science |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 5(1) Biomarker development. |
- Evaluate existing LBD biomarker efforts, including systematic assessments of their impact on identifying pre-dementia LBD and prodromal or at-risk for LBD patients.
- Informed by this evaluation, develop new biomarkers for LBD in both model systems and in humans, and in a diversity of tissues including in the periphery.
- Validate at least one biomarker in multi-site large scale trials.
- Incorporate at least one new LBD biomarker into studies designed to diagnose prodromal LBD.
|
3-7 years |
| 6(2) Genetic, epigenetic, and environmental characterization. |
- Identify novel common and rare genetic variants, epigenetic and other genomic changes, gene expression changes, and environmental influences on genomic and genetic factors that impact the risk for and clinical features of LBD.
|
3-7 years |
| 7(3) Understanding the molecular, cellular and pathophysiology of a-synuclein in the context of non-motor brain areas. |
- Develop in vitro and in vivo models that fit the known molecular pathology of LBD.
- Establish research focused on understanding α-synuclein biology in disease and non-disease states.
- Support research on how aging influences α-synuclein biology, pathology and cognitive impairment and dementia.
|
2-4 years |
| 8(4) Identify mechanisms by which Lewy body diseases may spread between and affect different brain regions and how Lewy bodies interact with other pathologies. |
- Support research investigating α-synuclein spreading and α-synuclein interactions with β-amyloid, tau, TDP-43 and other proteins and pathologies associated with dementia.
|
5-7 years |
| Topic 5: Vascular Contributions to Cognitive Impairment and Dementia (VCID) |
| Focus Area 1: Basic Mechanisms and Experimental Models |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 1(1) Develop next-generation experimental models and translational imaging methods for VCID. |
- Establish new small vessel VCID animal models suited for VCID and mixed dementias of aging research that: (i) reproduce small vessel disease and other key pathogenic processes thought to result in human cognitive impairment, (ii) are easily applicable to research on both VCID and AD dementia, (iii) address vascular contributions to damage of both white matter and gray matter; and (iv) include genetic and acquired conditions that cause and or are associated with VCID.
- Support development of in vitro, including cell-based, models to study specific molecular mechanisms of VCID that are not feasible in animal models.
- Support the development of new imaging and other tools that can be used to better understand VCID etiology at the behavioral (e.g. memory-based tasks), systems (e.g. hypoperfusion, chronic blood brain barrier breakdown), network (e.g. white matter lesions and network connectivity), pathology, cellular (e.g. variations and vulnerability in the vascular tree, neurovascular unit damage), synaptic (e.g. electrical activity), and molecular (e.g. chronic inflammation) levels.
|
3-5 years |
| 2(3) Foster basic science research on neurovascular unit function and how it is impacted by the following: aging, cardiovascular disease, AD pathology and genetics. |
- Support at least two new basic science research projects designed to understand the role of the perivascular (CSF) and paravascular (lymphatic) clearance pathways in normal brain health and whether these systems help protect the brain from VCID and other AD/ADRD dementias, as well as their progression.
- Support new research to determine: (i) how the neurovascular unit contributes to neurovascular coupling, i.e. regulation of brain blood flow across different vascular zones (arterioles, capillaries and venules), (ii) how normal neurovascular unit function is impacted by VCID risk factors (e.g. cardiovascular and cerebrovascular disease, metabolic factors, lifestyle factors, hypertension, TBI, AD/ADRD pathology and genetics, and aging), and by other AD/ADRD disorders, (iii) the relationship between blood brain barrier function and neurovascular unit function in health and in AD/ADRD.
|
3-5 years |
| 3(4) Encourage basic science research to determine the impact of cardiovascular/ cerebrovascular risk factors and genes on dementia-related brain changes, neurodegeneration and myelin biology. |
- Support new basic research that provides rigorous and novel insight into how cerebrovascular disease (small vessel) or cardiovascular and cerebrovascular risk factors (e.g. hypertension, diabetes mellitus/metabolic syndrome, dyslipidemia, genetic risk factors) impact the onset and progression of AD/ADRD-related disease processes including in different brain vascular zones (arterioles, capillaries, venules), neurodegeneration, myelin and neural circuits in gray matter and white matter.
|
5-7 years |
| Focus Area 2: Human-Based Studies |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 4(1) Develop, validate and longitudinally track: (i) cognitive, physical, or other functional assessment components that indicate the presence of VCID, (ii) VCID biomarkers, including when VCID is accompanied by pathological AD. |
- Support human-based research to identify imaging and fluid-based biomarkers that, together with appropriate cognitive and clinical measures (examples to consider, but not limited to locomotion, heart function, metabolic measures, and kidney function), indicate the presence (or absence), type, severity, and progression of small vessel VCID in at least two human cohorts.
- Establish a direct link from in vivo VCID biomarker measures (including appropriate cognitive and clinical measures) to ex vivo imaging to histopathology.
|
3-5 years |
| 5(2) Test for efficacy across the spectrum of VCID severity: (i) interventions proven to reduce cardio- and cerebrovascular risk, (ii) established care models. |
- Initiate at least one VCID clinical trial to test whether intervention(s) known to reduce cardiovascular and cerebrovascular risk also decrease VCID burden; this trial should use leading edge VCID biomarkers and cognitive measures, obtain VCID-relevant clinical data including on comorbidities, and when appropriate include others measures of neurologic function (e.g. walking test) that are relevant to cognitive outcomes.
- Support research to determine whether the best existing models for delivering care to persons with AD/ADRD and supporting their caregivers are effective in persons affected by VCID
|
3-5 years |
| 6(4) Determine interrelationships among cerebro- and cardiovascular disease, VCID risk factors, aging, resilience, genetics, amyloid, tau, and neurodegeneration including along the life course. |
- Complete one or more comprehensive studies of the independent associations between biomarkers of VCID and biomarkers of Αβ- and tau-related pathology/neurodegeneration in a human cohort.
- Initiate at least one intervention study to identify the effects of modifying VCID risk factors on biomarkers of Αβ- and tau-related pathology and neurodegeneration.
|
3-5 years |
| Focus Area 3: Translational Studies |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 7(2) Use data and other resources from large-scale clinical research and trials to test hypothesized mechanisms of human VCID in basic science models. |
- Support: (i) VCID studies in model systems that leverage data, resources, and results from, and also test hypotheses from, human VCID clinical research and clinical trials, (ii) the development of improved VCID model systems by leveraging data, resources, and results from human VCID clinical research and clinical trials.
|
3-5 years |
| 8(3) Incorporate VCID findings from basic science into the design of clinical research and trials targeting VCID-relevant cognitive impairment and dementia. |
- Support VCID clinical research and trials informed by basic VCID science research outcomes, e.g. biomarkers that emerge from basic studies, and basic mechanisms such as (but not limited to) glymphatic and intramural periarterial drainage (IPAD).
|
5-7 years |
| |
|
|
| |
|
|
| Topic 6: Frontotemporal Lobar Degeneration (FTD) |
| Focus Area 1: Science of Pathogenesis and Toxicity |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 1(1) Clarify unique and converging cellular mechanisms related to tau pathogenesis, C9orf72 hexanucleotide repeat expansions, GRN mutations, and other targets and pathways contributing to FTD neurodegeneration. |
- Support research that investigates mechanism of tau-driven neurotoxicity and its relationship to the formation and spreading of tau pathological inclusions in a prion-like manner.
- Support research to identify the predominant mechanism(s) of C9orf72 hexanucleotide repeat expansion pathogenesis in FTD.
- Support research examining the role of GRN mutations in FTD pathogenesis.
|
2-10 years |
| 2(2) Determine the mechanism of TDP-43 and FUS pathogenesis and toxicity. |
- Support research to identify the predominant mechanism(s) of TDP-43 and FUS-related pathogenesis and neurodegeneration in FTD.
- Determine if and how aggregated TDP-43 spreads.
|
3-10 years |
| 3(3) Develop data and resource infrastructures to support management and collaborative analysis of diverse clinical, imaging, genetic, molecular, and biomarker data and resources from FTD basic science and clinical studies. |
- Support one or more platform(s) that allow the research community to visualize and access clinical data, biospecimens, neuropathological and genetic resources.
|
1-3 years |
| 4(4) Develop better FTD in vivo and cell-based model systems. |
- Generate validated and standardized in vivo animal and cell-based models of TDP-43, FUS, GRN haploinsufficiency, and C9orf72 expansion disease, which recapitulate key biochemical, anatomical, neuropathological and functional aspects of FTD.
- Develop and validate in vivo functional assays and neuropathological endpoints for mammalian models that are aligned with the anatomical sites affected in FTD.
|
1-3 years |
| Focus Area 2: Clinical Science |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 5(1) Develop FTD biomarkers for diagnosis, prediction, and disease monitoring. |
- Develop, test, and pathologically confirm novel blood/CSF biomarkers or PET ligands for FTLD-tau, -TDP or -FUS during life, including in populations that experience health disparities.
- Develop, test and validate sensitive, systems-level biomarkers using wearable technologies that monitor FTD progression, including in populations that experience health disparities.
|
2-7 years |
| 6(2) Advance FTD clinical trial design and execute new prevention and treatment studies. |
- Support a research registry for FTD clinical studies that includes underserved and minority group representation reflecting population demographics.
- Develop standardized protocols for prevention studies of familial FTD syndromes or clinical trials in symptomatic, sporadic, and familial FTD.
- Support clinical trials of novel FTD therapeutics.
|
1-4 years |
| 7(3) Expand efforts to genotype patients with FTD, identify new risk factor genes and epigenetic modifiers. |
- Support efforts to genotype and perform genomic analyses on patients across the full spectrum of FTLD.
|
1-5 years |
| 8(4) Understand phenotypic heterogeneity and natural history including in populations that experience health disparities. |
- Support natural history studies of preclinical inherited FTD, by following individuals from health to disease.
- Support natural history studies of sporadic FTLD, starting from early symptomatic FTD and prioritizing clinical syndromes for which the clinico-pathological correlation is high.
|
>10 years |
| Topic 7: Emerging Scientific Topics |
| Focus Area 1: TDP-43 Pathology in Common Dementias |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 1(1) Develop biomarker and risk profiles to establish in-vivo diagnostic criteria for TDP-43 pathology in persons without cognitive symptoms and in persons with amnestic syndromes, e.g. amnestic MCI and AD clinical syndrome. |
- Support research to develop biomarker and/or risk profiles designed to facilitate clinical trial stratification for TDP-43 proteinopathy in common dementias.
|
5-7 years |
| 2(2) Determine underlying pathobiologic and molecular mechanisms of cellular TDP-43 displacement, post-translational modifications such as phosphorylation, and pathology in pre-symptomatic and manifest common dementias. |
- Support at least one new initiative that fosters rigorous research focused on advancing knowledge on the systems, cellular and/or molecular mechanisms and consequences of TDP-43 proteinopathy in common dementias using in vivo models, cellular models and/or human biospecimens.
|
3-5 years |
| 3(3) Examine the pathologic phenotype(s) of TDP-43 pathology in asymptomatic persons and those with common dementias. |
- Complete and report the results of one or more rigorously conducted studies that characterize TDP-43 dependent pathologic phenotypes in elderly individuals both with and without a clinical diagnosis of a common dementia to better understand the significance of TDP-43 pathology.
|
5-7 years |
| 4(4) Build new animal models to advance knowledge about TDP-43 pathology in common dementias, capitalizing on lessons learned from animal models in FTLD/ALS, AD and other diseases. |
- Develop, validate and make available to the broader scientific community two or more novel animal models that exhibit TDP-43 pathology aligned with the affected anatomical sites and functional changes that occur in common dementias such as the clinical syndrome of Alzheimer’s disease.
|
3-7 years |
| Focus Area 2: TBI and AD/ADRD Risk |
| Milestone (Priority Level) |
Success Criteria |
Timeline |
| 5(1) Encourage crosstalk and interdisciplinary collaboration between TBI and dementia researchers. |
- Convene a working group of stakeholders from the TBI and AD/ADRD communities, including trainees, to evaluate the extent to which current knowledge in AD/ADRD can be applied to the study of dementia after TBI. This working group can assess the level of data harmonization across existing TBI and dementia clinical cohort studies to facilitate current and future comparisons and data sharing.
- Support the addition of interdisciplinary retrospective assessment for TBI history and prospective TBI occurrence in existing AD/ADRD cohort studies.
- Support the addition of AD/ADRD assessments to existing TBI cohort studies.
|
1-3 years |
| 6(2) Establish infrastructure to study TBI as a risk factor for AD/ADRD. |
- Support further development of national TBI research sharing infrastructure and best practices for clinical data (including cognitive outcomes), biospecimens (including autopsy), pathology, and imaging to promote broad cross-disciplinary discovery and validation of acute and chronic TBI-related risk stratification models of dementia. Ensure that data and samples represent the diversity of the population and the range and variability of TBI causing events and history.
- Develop and/or identify collections of tissue and biofluid samples from existing or newly developed longitudinal case-control or cohort studies in which samples are collected using standardized protocols and in which the samples are linked to clinical data that include TBI and AD/ADRD cases.
|
1-5 years |
| 7(3) Promote basic and clinical research examining the development and progression of TBI AD/ADRD neuropathologies and associated clinical symptoms. |
- Develop one or more new TBI-related dementia animal models (informed by neuropathologic and epidemiological studies of chronic TBI) and new translatable functional outcome measures that model both the static and progressive decline of cognitive, motor, and neurovascular function in humans.
- Support basic research to identify mechanisms (e.g., immune response, neurovascular changes, blood-brain barrier breakdown) that initiate and maintain progressive neuropathological processes after TBI in human and animal models.
- Support studies to identify potential neuropathological substrates of dementia in TBI. Characterize the relative burden of individual pathologies in relation to injury severity and mechanism and to identify similarities, differences, and common mechanisms of pathological protein isoforms with AD/ADRD.
|
2-10 years |
| 8(4) Characterize the clinical phenotype of progressive dementia associated with TBI and develop non-invasive diagnostic approaches. |
- Develop and validate a provisional clinical definition of TBI-related dementia(s) that distinguishes chronic static TBI-related symptoms from a progressive neurodegenerative disease.
- Establish diverse longitudinal prospective studies of individuals with TBI and unexposed controls with harmonized multimodal clinical evaluations including autopsy endpoints to identify clinical and pathophysiological features for differential prognoses of AD/ADRD risk following TBI. Consideration of the role of common TBI comorbidities, such as PTSD, pain, and mood dysregulation, should be included in the clinical phenotyping.
- Support the discovery and validation of chronic TBI biomarkers. These biomarkers should non-invasively identify the development of TBI-AD/ADRD pathologies and track their progression over time in relation to dementia.
|
2-10 years |

