Once a drug comes off patent, the U.S. has been highly successful in reducing prices and improving the value received by promoting generic drug introduction and competition. The U.S. usage of generic drugs is greater than other developed countries and the U.S. pays less for generic drugs than other developed countries.
While overall, the U.S. generally pays more for drugs than other developed countries, this differential varies substantially:
However, a bright spot for the U.S. has been generic drugs.
- The U.S. both pays less for generics and uses more of them compared to other OECD nations. About 84% of all prescription volume in the U.S. is generics compared to 35% in other OECD nations. The U.S. pays about $0.84 on the dollar for generic drugs compared to other OECD nations.
- A recent ASPE brief found that generic competition works to reduce the nation’s drug spending both when a generic drug initially comes onto the market and then over time as more generic manufacturers enter the marketplace.
(Medicare Part D: Competition and Generic Drug Prices, 2007-2018)- Once a drug goes off patent protection and generic drug competition begins, prices decline by an average of 20% in markets with three generic competitors. Even after that initial price decline, prices for generic drugs relative to the pre-generic entry price generally continue to decline with more competitors entering the market. In markets of 10 or more competitors, prices decline by 70% in two years and by 80% in three years after first generic entry. The amount of decline in price from the original brand price varies by the competitiveness of the generic market, markets that have more competitors result in larger price declines.
- This finding illustrates the importance of FDA generic approvals, both of the first generic drug and then subsequent competitors as well. In recent years, the FDA has approved a record number of generic drugs.
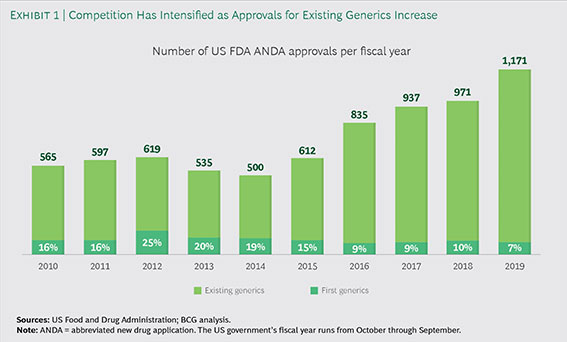
Number of US FDA ANDA Approvals Per Fiscal Year
The record number of generic approvals translates directly into lower drug spending via greater competition and lower prices. This translates into lower U.S. healthcare spending. In fact, if generics were used when available, an ASPE brief shows that Medicare and its beneficiaries would save an additional $2.8 billion annually.
Resources:
