ADVISORY COUNCIL ON ALZHEIMER'S RESEARCH, CARE, and SERVICES
Monday, May 3, 2021
Printer Friendly Version in PDF Format (31 PDF pages)
Federal Agency Updates
Alzheimer's Disease and Related Dementias Research Update
Richard J. Hodes, M.D.
Director, NIA
Growth Trends in NIA's AD/ADRD Research Portfolio Fiscal Years 2008-2019
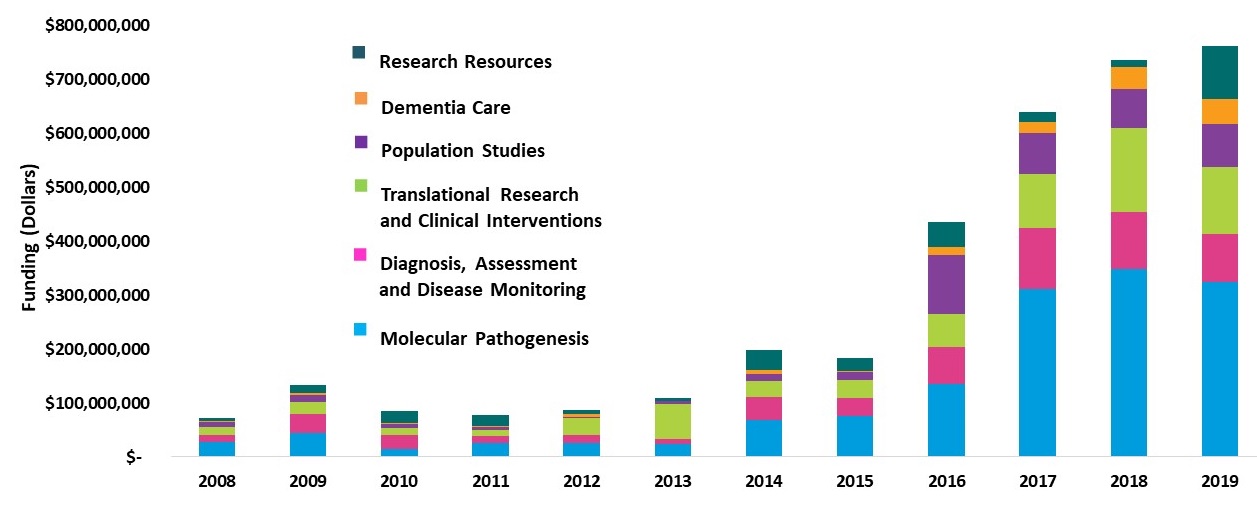 |
Trends in NIA's AD/ADRD Research Portfolio Fiscal Years 2008-2019
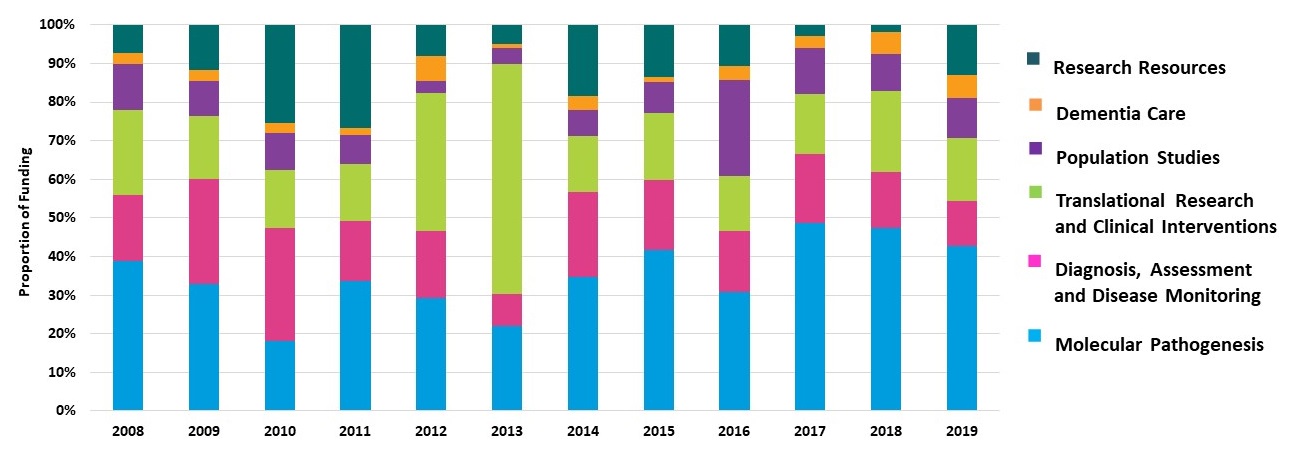 |
Recruitment of a Growing AD/ADRD Research Workforce
- ∼1/3 of NIA's Alzheimer's disease and related dementias awardees from Fiscal Year 2015-2020 were either new or early-stage investigators.
- ∼1/4 of NIA's Alzheimer's disease and related dementias awardees were new to the field.
NIA's Alzheimer's Translational Research Program
NIA's Alzheimer's Translational Research Program provides a pipeline of funding opportunities for academic institutions and biotech companies. These programs support the discovery and development of new drugs targeting key aspects of the disease process.
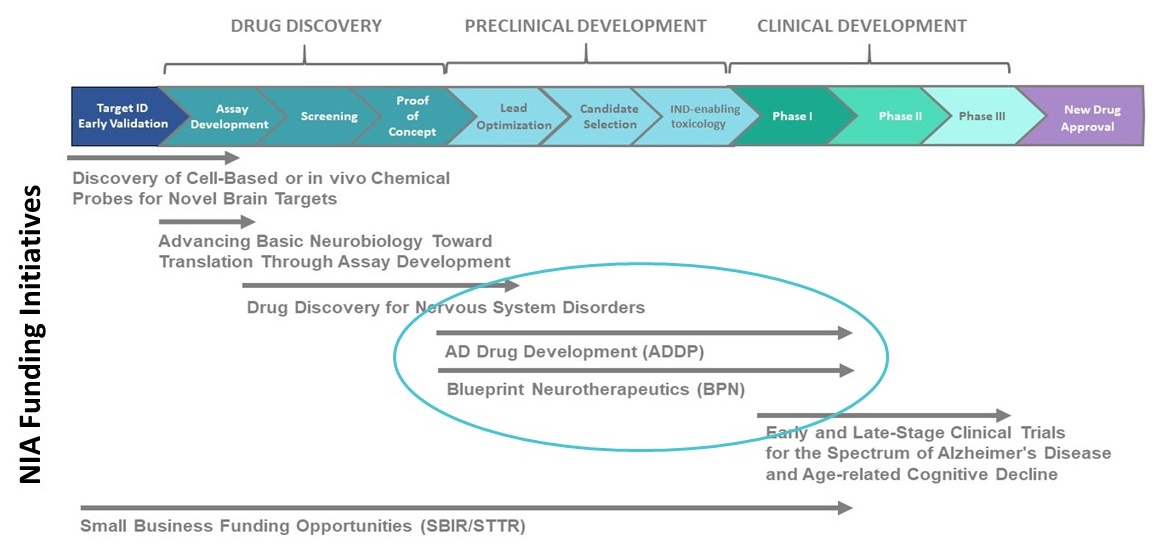 |
ADDP and BPN Candidates in Pre-clinical Drug Development are Diverse
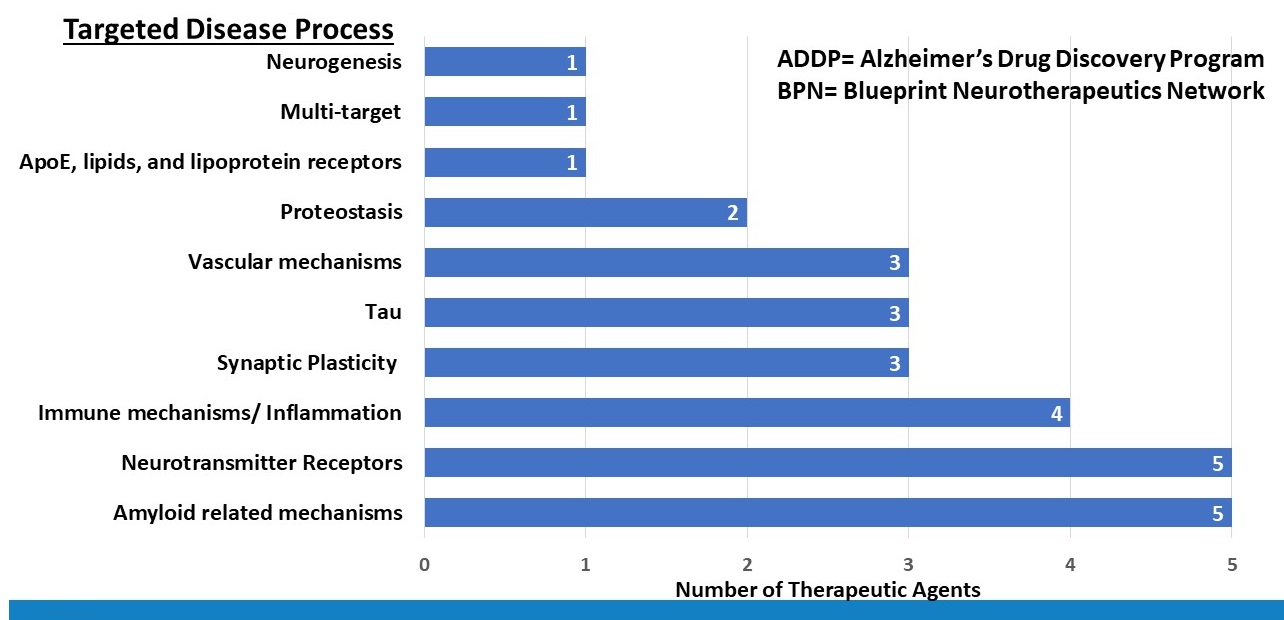 |
Accelerating Medicines Partnership for Alzheimer's Disease (AMP-AD)
- AMP-AD
- Genomic, proteomic, metabolomic data from human brain and peripheral fluid samples
- Computational modeling/network biology
- Experimental validation in cell-based and animal models
- Drug Discovery
- AMP-AD is an NIA-led, public-private partnership among government (NIA, NINDS,FDA), pharmaceutical industry and non-profit foundations
- Uses big data approaches to better understand the complex nature of AD to identify new targets for treatment.
- AMP-AD Accomplishments
- Centralized data resources for sharing data, analytical results and target nominations, over 2500 AD and control cases
- Rich, high quality data and network models of disease pathways and targets
- Datasets being widely used: ∼3000 users to date - 60% academia, 40% biotech/pharma
- Over 500 novel candidate targets identified
AMP AD 2.0: Enabling a Precision Medicine Approach to Target and Biomarker Discovery
- Expand multi-omic profiling in samples (brain, CSF, blood) from diverse cohorts (African American and Latino American)
- Generate longitudinal immunologic profiling data across diverse cohorts (Caucasian, African American and Latino American)
- Expand the existing sn/sc molecular profiling efforts to multiple brain regions, and in samples from diverse cohorts
- AMP AD 2.0 Partners
- National Institute on Aging
- National Institute of Neurological Disorders and Stroke
- Food and Drug Administration
- Foundation for the National Institutes of Health
- Alzheimer's Association
- GlaxoSmithKline
- Eisai
- Takeda Pharmaceutical Company
- Gates Ventures
- Launched: March 2021
- AMP AD 2.0 brings together 8 research teams from over 20 academic institutions and 9 public-private partner organizations.
- open data and methods
- mechanistic insights
- molecular disease subtypes
- new targets and biomarkers
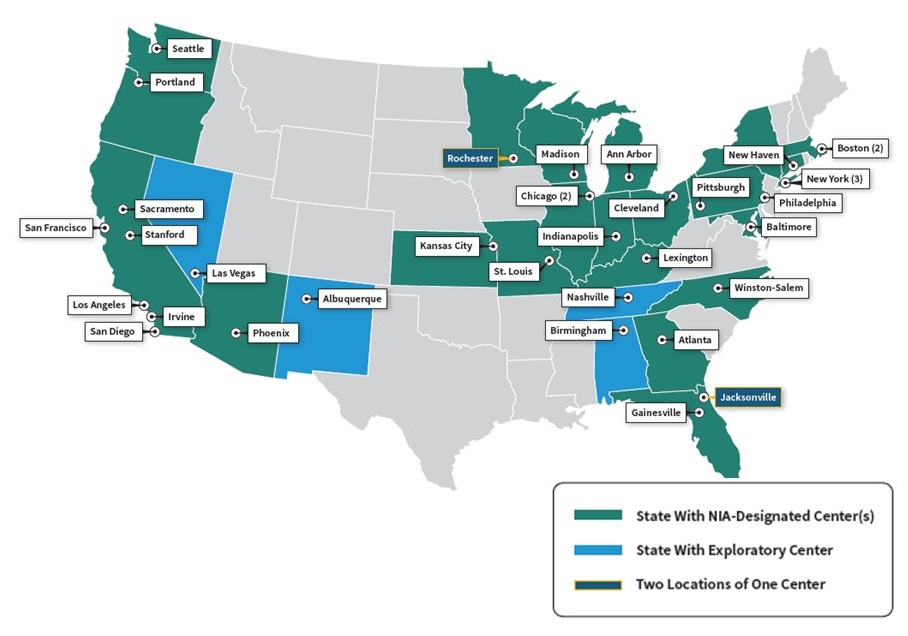
Enhancing the Alzheimer's Disease Research Centers Network
- 31 ADRCs
- 4 new exploratory centers
- Clinical, neuropathology, imaging, biomarkers, genetics, pathogenesis, therapeutics
- Provide diagnosis and education services for patients and families
 |
Active NIA AD/ADRD and Related Intervention and Prevention Trials (~270)
| 45 Early-Stage Clinical Drug Development (Phase I and Phase II Clinical Trials) | 11 Late-Stage Clinical Drug Development (Phase II/III and Phase III Clinical Trials) | 123 Non-Pharmacological Interventions | 7 Clinical Therapy Development for Neuropsychiatric Symptoms of AD/ADRD | 84 Care and Caregiver Interventions |
|---|---|---|---|---|
| Amyloid (11) Receptors (2) Sleep (1) Growth factors and Hormones (2) Inflammation (3) Metabolism and Bioenergetics (2) Multi-target (2) Neurogenesis (2) Oxidative stress (3) Synaptic Plasticity/Neuroprotection (6) Vasculature (2) Other (9) | Amyloid (7) Synaptic Plasticity/Neuroprotection (2) Tau (1) Vasculature (1) | Assistive Tech/Device (13) Cognitive Training (18) Combination Therapy (11) Decision-Supportive (5) Diagnostic Tools (8) Diet/Supplements (7) Exercise (27) Light Therapy (1) Neurostimulation (14) Sleep (6) Other (13) | Pharmacological (5) Non-Pharmacological (2) | Improving Care for PWD (43) Improving Family or Informal Caregivers (41) |
| https://www.nia.nih.gov/research/ongoing-AD-trials | ||||
Clinical Trials Pivot During Pandemic to Keep Studies Going
- Examples of Shifts to Clinical Trial Protocols in response to COVID-19
- Exercise trials are now using virtual coaching and fitness training
- Trials with intravenous infusions of medication are moving to in-home infusions
- Trials have shifted to remote monitoring and screening of participants
- A number of these flexibilities will remain even after trial sites re-open as they present greater options and reduce barriers for participants
IMbedded Pragmatic Alzheimer's disease (AD) and AD-Related Dementias (AD/ADRD) Clinical Trials (IMPACT) Collaboratory
- NIA IMPACT Collaboratory
- Support Design & Conduct of ePCTs
- Build Investigator Capacity
- COVID-19 Response
- Catalyze Stakeholder Collaboration
- Develop & Disseminate Knowledge
- IMPACT supported pilot trials include:
- Working to improve AD/ADRD care management across interdisciplinary teams
- Designing mobile apps to help adult day service centers prevent minor health issues from escalating to medical emergencies
- Empowering bedside nurses in emergency departments to improve detection of dementia in patients
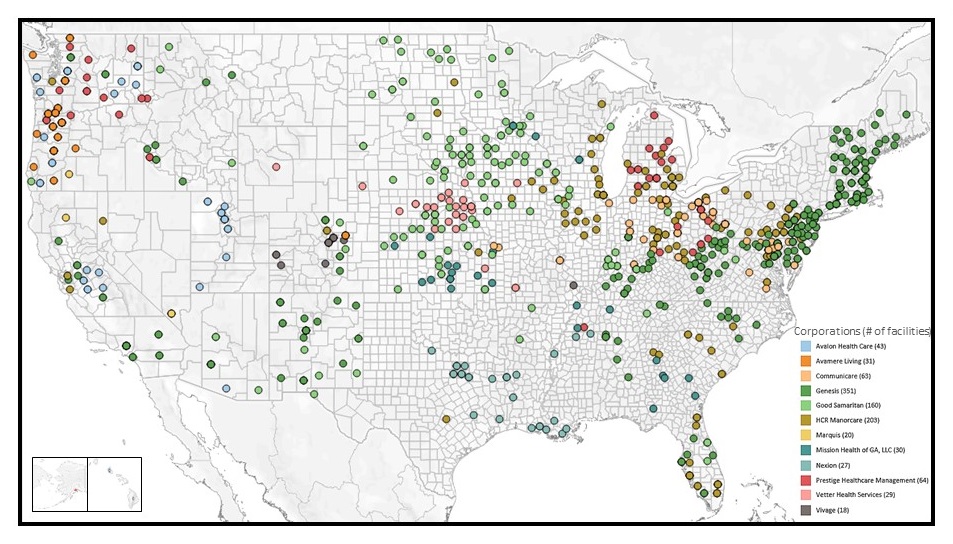
NIA IMPACT Collaboratory Data Sharing Cooperative: Geography of Facilities
 |
AHRQ-National Academies Study on Dementia Care & Caregiving
- Two parts:
- Agency for Healthcare Research & Quality (AHRQ)
Conduct a rigorous systematic review of the evidence on care and caregiving interventions for people living with dementia (PLWD) and their caregivers -- released in July 2020 - National Academies of Sciences, Engineering, and Medicine (NASEM)
Establish an expert committee to develop a report outlining a set of recommendations on the readiness of these interventions for broad dissemination and implementation, and research gaps in the field -- released in February 2021
- Agency for Healthcare Research & Quality (AHRQ)
National Academies Recommendations on Dementia Care & Caregiving
- Key Messages from NASEM Synthesis:
- Most caregiving interventions were not assessed in the study because they had small sample sizes, were pilots, or had high risk of bias
- Two types of interventions demonstrated low-strength evidence of benefit in clinical trials:
- Collaborative Care
- Resources for Enhancing Alzheimer's Caregiver Health (REACH) II
- The report recommends broader implementation of these programs in real-world settings that allow for continual monitoring, evaluation, and quality improvement
- https://www.ncbi.nlm.nih.gov/books/NBK567818/
2021 Alzheimer's Disease Research Summit
- Path to Precision Medicine for Treatment and Prevention
- April 19-22, 2021 Virtual Event
- #ADSummit2021
- Archived presentations found at: https://www.nia.nih.gov/2021-alzheimers-summit
Introducing the new Alzheimers.gov!
- Your destination for dementia information, resoures, and research
- Visit www.Alzheimers.gov
Ways to Stay Informed and Connected
- Search all active NIA funding opportunities: https://www.nia.nih.gov/research/funding
- Review the latest approved concepts: https://www.nia.nih.gov/approved-concepts
- Subscribe to our blog and stay up to date on the latest NIA news: https://www.nia.nih.gov/research/blog
NIA -- The Leader in Aging Research
Long-Term Services and Supports Federal Update
Administration for Community Living -- FUNDING OPPORTUNITY ANNOUNCEMENT
- Alzheimer's Disease Programs Initiative - Grants to States and Communities
- HHS-2021-ACL-AOA-ADPI-0083
- Estimated Total Funding: $11,053,915
- Expected Number of Awards: 14
- Award Ceiling: $1,000,000 Per Project Period
- Award Floor: $550,000 Per Project Period
- Applications due: MAY 10, 2021
- NAPA Goals 2, 3, 4
Administration for Community Living -- UPCOMING FUNDING OPPORTUNITY ANNOUNCEMENT
- Alzheimer's Disease Programs Initiative - Dementia Capability in Indian Country
HHS-2021-ACL-AOA-ADPI-0087 - Alzheimer's Disease Programs Initiative - Grants to States and Communities - Round Two
HHS-2021-ACL-AOA-ADPI-0083 - NAPA Goals 2, 3, 4
Administration for Community Living -- National Alzheimer's and Dementia Resource Center
- 2021 WEBINAR Series
- January 13, 2021- Reaching Socially Isolated People Living with Dementia with Carol Manning and Mike Splaine- 727 participants
- February 9, 2021- Exploring Male Caregiving for People Living with Dementia with Molly Perdue and Bob O'Brien, male caregiver- 505 participants
- March 11, 2021- Complex Care Management for People Living with Dementia with Sarah Dulaney and Lori Resnick- 586 participants
- April 27, 2021- Community Health Workers: A key support for people living with dementia and their caregivers with Constantina Mizis and Alicia Nielson -- 346 participants
- Recordings and presentation PowerPoints are posted on the NADRC webpage
- UPCOMING WEBINARS https://nadrc.acl.gov/
- Person Centered Goal Discovery for People Living With Dementia, Tuesday, May 18, 2021 1:00-2:00 PM ET. Supports for people living with dementia and their families are most effective when they are person-centered and the result of a thoughtful planning process that considers what is important to them. When guiding providers on how to deliver high quality support, it is important to use a clear and engaging approach to discover and address people's priorities as dementia progresses. This webinar will present foundational person-centered principles, and provide examples of how to plan for people who are living with dementia in the community and other settings.
- June 23, 2021- Elder Abuse and Dementia with Dr. Laura Mosqueda and Charlie Sabatino
- August 24, 2021- Building and Maintaining Relationships after a Dementia Diagnosis (Matt Estrade and Care Partner)
- NAPA Goals 2, 3, 4
Administration for Community Living -- ACL Brain Health Webpage
- New Content -- Focus on Resources for Community Based Organizations
Administration for Community Living -- Issue Brief
- Cataloguing ACL-Funded Support of Brain Health in States and Communities Over Many Years
- Brain Health Services or Activities Include:
- Psychoeducational programs for family caregivers of persons with dementia.
- Educational series covering basics of brain function, types of dementia, and living with dementia.
- Community presentations covering topics on healthy lifestyle choices and signs of cognitive impairment.
- Sources: https://acl.gov/sites/default/files/common/Iyengar_Long_BrainHealth_IssueBrief_ACLAlzheimersDiseasePrograms_0.pdf.
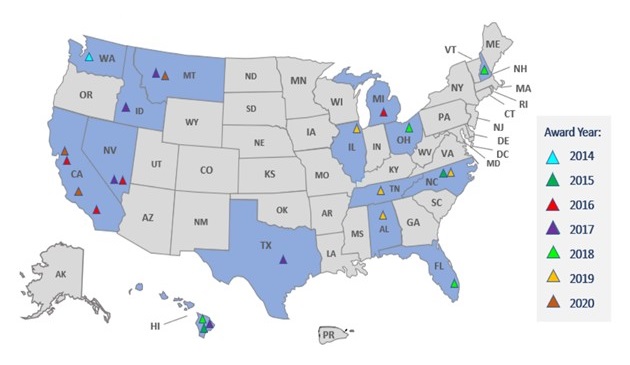
| Figure 1: ACL has supported brain health services or activities from 23 unique organizations (shaded triangles) spanning 15 states (shaded in dark blue) between the years 2014-2020. |
|---|
 |
Administration for Community Living -- Minority Organizations Technical Assistance & Resource Center (TARC) Program
- ACL funds five national organizations that support diverse older adult populations:
- Technical Assistance call -- MHP Salud: COVID-19, Social Isolation, and Hispanic Adults with Alzheimer's and other Dementia February 17, 2021.
- Blog -- MHP Salud: How to Raise Awareness About Dementia in Hispanic/Latino Older Adults, (available in English and Spanish) January 2021.
- NAPA Goals 2, 3, 4
Questions? Thank you!
Erin Long, MSW
Team Lead
Alzheimer's Disease Programs Initiative (ADPI)
Office of Supportive and Caregiver Services
Administration on Aging
Administration for Community Living
Erin.Long@acl.hhs.gov
202-795-7389
CMS Posts Information on Medicare Cognitive Assessment and Care Plan Services
- In March 2021 CMS updated the web page for beneficiaries that describes covered items, devices, and services to include a description of the Cognitive Assessment and Care Plan Service (CPT code 99483) available to beneficiaries with under Medicare Part B, distinct from the cognitive screening in the Annual Wellness Visit
- The new page links to resources including the Medicare depression screen, Alzheimer's.gov, CDC and NIH resources, ACL's Eldercare locator, and VA benefits
- CMS also plans to include a reference in the 2022 "Medicare & You" handbook
- The new page can be found at: https://www.medicare.gov/coverage/cognitive-assessment-care-plan-services
- (Strategy 2.B)
Indian Health Service Updates
Dr. Bruce Finke, MD
Indian Health Service
- $5,000,000 appropriated to the IHS in FY 2021 to address Alzheimer's Disease.
- IHS is currently highly engaged in Tribal Consultation and Urban Confer with Tribal and Urban Indian Program Leadership on the allocation of these resources.
- Consultation initiated with Dear Tribal Leader Letter on March 31, 2021. https://www.ihs.gov/newsroom/triballeaderletters/2021letters/
- April - May: Learning Sessions, Tribal Consultation and Urban Confer Sessions, Area Listening Session - recorded and available. https://www.ihs.gov/dccs/consultationandconfer/
- To submit comments and for the schedule of all sessions: https://www.ihs.gov/dccs/consultationandconf
- Approach aligned with the National Plan to Address Alzheimer's Disease https://aspe.hhs.gov/report/national-plan-address-alzheimers-disease-2020-update
- Partnership with Tribes and Urban Indian Health programs.
- Integrated with other federal efforts.
- Opportunities for partnership and collaboration with non-federal partners.
- Conclusion of Tribal Consultation / Urban Confer by June, plan for action by July.
- This is the first ever specific appropriations for ADRD in the IHS budget.
ASPE Updates
New Reports on People with Dementia
- The Impact of COVID-19 on Medicare Beneficiaries with Dementia
- The Risk and Costs of Severe Cognitive Impairment at Older Ages: Literature Review and Projection Analyses
- https://aspe.hhs.gov/basic-report/risk-and-costs-severe-cognitive-impairment-older-ages-literature-review-and-projection-analyses
- Issue Brief: https://aspe.hhs.gov/basic-report/risk-and-costs-severe-cognitive-impairment-older-ages-key-findings-our-literature-review-and-projection-analyses-research-brief
Risk & Costs of Cognitive Impairment
- Uses the Dynamic Simulation of Income Model (DYNASIM) to project:
- The risk and costs of severe cognitive impairment at older ages
- Severe cognitive impairment (SCI): "Cognitive impairment that is severe enough to limit function, usually defined as social or occupational function. In its severe forms, a person with dementia/SCI may not be able to recognize people, use language, or execute purposeful movements."
- (Finding consistent with presentations by Judy Dey and Melissa Favreault in October 2019)
Distribution of Projected Costs of Paid LTSS and Value of Unpaid Care for Those Who Ever Experience SCI: DYNASIM 1955-1959 Birth Cohort
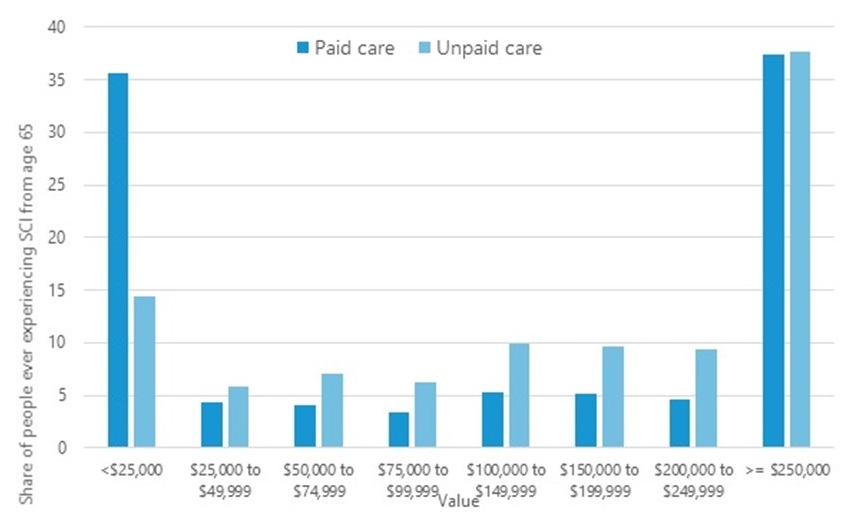 |
| The value of informal care --close to $243,5000 ($161,000 in present value terms)--just exceeds the formal costs--$281,000 ($164,000 in present value terms). |
New Reports on Risks & Financing of LTSS
- Long-Term Services and Supports for Older Americans: Risks and Financing, 2020 Research Brief
- Projections of Risk of Needing Long-Term Services and Supports at Ages 65 and Older
New Reports on LTSS and Economic Hardship
- Economic Hardship and Medicaid Enrollment in Later Life: Assessing the Impact of Disability, Health, and Marital Status Shocks
- Extended LTSS Utilization Makes Older Adults More Reliant on Medicaid: Issue Brief
- Risk of Economic Hardship Among Older Adults: Issue Brief
CDC Updates
Lisa C. McGuire, PhD
COVID-19 Information for Older Adults
- Older adults and people of any age who have serious underlying medical conditions are at higher risk for developing more serious complications from COVID-19 illness.
- COVID-19 Guidance for Older Adults (cdc.gov) https://www.cdc.gov/aging/covid19-guidance.html
COVID-19 Guidance for Older Adults: ADULT DAY SERVICE CENTERs (ADSCs)
- Guidance for ADSC Participants and Caregivers https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/adult-day-service-centers.html
- Ways to protect yourself and other people
- What to do if you're sick or have been exposed to COVID-19
- What to do if you're worried, stressed, or sad
- Additional resources
- Guidance for ADSC Administrators and Staff https://www.cdc.gov/coronavirus/2019-ncov/community/adult-day-care-service-centers.html
- Promotion of vaccines and mitigation behaviors
- Strategies for healthy operations
- Maintenance of healthy environments
- Mental health resources
COVID-19 Cases Among Nursing Home Residents and Staff Mirror Community Spread
- https://www.cdc.gov/aging/covid19-guidance.html
- COVID-19 infection rates increased during June, July, and November 2020 among nursing home residents and staff.
- Trends in reported COVID-19 cases among nursing home residents and staff members resembled trends in COVID-19 incidence in surrounding communities. Morbidity and Mortality Weekly Report, January 9, 2021. https://www.cdc.gov/mmwr/volumes/70/wr/mm7002e2.htm
COVID-19 Information for Older Adults Mental Health and Social Isolation Among Older Adults
- How Right Now https://howrightnow.org/
- HowRightNow.org provides inspiration, tips, and resources to help manage and reduce any feelings of stress, loneliness, worry, or sadness, you might be experiencing.
- Social Isolation and COVID-19 https://podcasts.apple.com/us/podcast/ep38-cdc-highlights-social-isolation-loneliness-among/id1508046828?i=1000501704755
- Dr. Craig Thomas discusses growing concerns of social isolation and loneliness among older adults before and during the COVID-19 pandemic.
COVID-19 Information for Older Adults
- Maintaining Physical and Mental Well-Being of Older Adults and Their Caregivers During Public Health Emergencies https://www.norc.org/Research/Projects/Pages/maintaining-physical-and-mental-well-being-of-older-adults-and-their-caregivers-during-public-health-emergencies.aspx
- Highlights racial and ethnic groups, persons with disability, rural populations, tribal populations, LGBT individuals, populations with limited English proficiency, and groups that have been economically/socially marginalized.
- Key needs and concerns of older adults and caregivers during COVID-19 and needed assistance for older adults and caregivers
- Older adults' and caregivers' information-seeking behaviors and resource preferences
- Interventions and strategies focused on social isolation, deferral of medical care, management of chronic conditions, elder abuse and neglect, and deconditioning
- The National Foundation of the Centers for Disease Control and Prevention funded this study conducted by NORC at the University of Chicago, with partners TMN Corp and Burness
Resources: The National Association of Chronic Disease Directors (NACDD) Rack Cards
- https://www.cdc.gov/aging/partnership/nacdd-partner-resources/index.html
- The NACDD partnered with CDC developed a series of visual information products to provide information about reducing risk for cognitive decline. The cards focus on areas such as healthy diet, adequate physical activity, and blood pressure management.
Resources: Healthy Heart, Healthy Brain Editable Messaging
- The Healthy Brain Initiative Road Map Series calls on public health to educate the public about steps to take to reduce risk of cognitive decline. https://www.cdc.gov/aging/healthybrain/roadmap.htm
- The new editable templates provide ways people can work with their health care providers and independently to protect cognitive and heart health. https://astho.org/Healthy-Aging/Healthy-Heart-Healthy-Brain/
Resources: Managing Hypertension to Protect Heart and Brain Health
- https://www.alz.org/media/Documents/executive-summary-healthy-brain-initiative-protecting-heart-and-brain.pdf
- Hypertension is a major risk factor for numerous health and chronic conditions, including cognitive impairment. The public health community can help reduce the risk of cognitive decline in populations by preventing and managing high blood pressure.
Resources: Community Health Workers (CHW) Guide
- The new Community Health Workers (CHW): A Resource for Healthy Aging and Addressing Dementia guide features how CHW can promote healthy aging and address dementia challenges. https://www.alz.org/media/Documents/community-health-workers-a-resource-for-healthy-aging-and-addressing-dementia.pdf
Risk Reduction Goal Updates
Lisa C. McGuire, PhD
Risk Reduction Goal
- Rationale
- Advances in science of risk factors: Science continues to evolve in the prevention, identification, treatment, and management of Alzheimer's disease and related dementias (ADRD). The most current scientific information will be utilized to drive the development of this goal.
- Preclinical stages of the disease: The preclinical states of ADRD are ripe for primary prevention. Intervening during this portion of the life course will not only reduce or delay the onset on ADRD but also will help to maintain the health, well-being, and independence of older adults by improving the health of the nation.
- Heterogeneity of pathologies that cause dementias: Dementia symptomology is caused by multiple pathologies and combinations of pathologies. The intervention of risk factors known to prevent or delay onset of ADRD, regardless of pathology, is the key.
- Objective
- To develop a national goal to reduce the burden of risk factors in order to prevent or delay onset of Alzheimer's Disease and related dementias (ADRD)
Steering Committee
- Matthew Baumgart, Vice President of Health Policy at Alzheimer's Association
- Lisa C. McGuire, Ph.D., Lead, Alzheimer's Disease Program, National Center for Chronic Disease Prevention and Health Promotion, Division of Population Health
- Kelly O'Brien, Executive Director of the Brain Health Ecosystem project at UsAgainstAlzheimer's
Process and Timeline
- Process
- Steering Committee Initial Meetings:
- Establish Subcommittee Members
- Develop initial recommendations
- Form Workgroups (subset of risk factors)
- Science, clinical intervention, and public health action
- Technical guidance for national risk reduction goal
- Subcommittees revise recommendations
- Steering Committee reviews and revises recommendations
- Recommendations sent to broader review and then revised
- Recommendations presented to NAPA Advisory Council
- Steering Committee Initial Meetings:
- How to get involved as a reviewer:
SUBCOMMITTEE MEMBERSHIP
- Steering and Subcommittee Advisor: Alan Levey, NAPA co-chair
- Expertise Categories:
- Public Health
- Clinical Care
- Innovation Industry
- Research
- Selection Criteria/Consideration: Will include two members who fall into one of the categories above to serve as subcommittee members.
- Multi-disciplinary, action-oriented, and diverse candidates
- Those with a history of working within the field (e.g., clinician, academic)
- Range of experiences and representation from fields of aging, cardiovascular disease, and other primary risk factor areas of AD/ADRD
Subcommittee Members
- Jewel Mullen, Dell Medical School
- Joshua Chodosh, New York University Langone Health
- Laurie Whitsel, American Heart Association, American Stroke Association
- Caraline Coats, Humana
- Joe Chung, Kinto Care, Redstar Ventures
- Marilyn Albert, Johns Hopkins Medicine
- Karthik Sivashanker, American Medical Association
- Rebecca Gottesman Johns Hopkins University
Workgroup Leaders
| Group A | Group B | Group C | Group D | ||||
|---|---|---|---|---|---|---|---|
| Joshua Chodosh | Jewell Mullen | Laurie Whitsel | Caraline Coats | Marilyn Albert | Joe Chung | Rebecca Gottesman | Karthik Sivashanker |
| Obesity Diet Sleep Traumatic Brain Injury | Physical Activity Smoking/Tobacco Alcohol | Social Isolation Depression Hearing Loss Cognitive Activity | Hypertension Hyperlipidemia Diabetes | ||||
Risk & Protective Factors Selected
- Alcohol
- Cognitive Activity
- Depression
- Diabetes
- Diet
- Hearing Loss
- Hyperlipidemia
- Hypertension
- Obesity
- Physical Activity
- Sleep
- Smoking/Tobacco
- Social Isolation
- Traumatic Brain Injury
Workgroup Members
| Group A: Joshua Chodosh and Jewell Mullen | Group B: Laurie Whitsel and Caraline Coats | Group C: Marilyn Albert and Joe Chung | Group D: Rebecca Gottesman and Karthik Sivashanker |
|---|---|---|---|
| Obesity, Diet, Sleep, Traumatic Brain Injury | Physical Activity, Tobacco Use, Alcohol | Social Isolation, Depression, Hearing Loss and Cognitive Activity | Hypertension, Hyperlipidimia, Diabetes |
| Jeannette Beasley Jennifer Martin Temitayo Oyegbile-Chidi Kristine Yaffe Alice Lichtenstein | Ross Arena Aruni Bhatnagar Cedric Bryant Jordan Endicott Hadiya Green Gerraro James Galloway Mark Stoutenberg Monica Rivera-Mindt Amy Bantham Melinda Kelley Monica Cornelius Alison Moore John Omura/Kathy Watson | Carla Perissinotto Ipsit Vahia Meryl Butters Frank Lin Michael Marsiske | Hugo Aparicio Heather Hodge Walter Kernan David Hoffman Ann Marie Navar Laura Baker Denard Cummings Judy Hannan |
Framing Questions
- To what extent does the potential dementia risk and protective factor have strong evidence from a population-based perspective?
- To what extent is the potential dementia risk and protective factor ripe for public health action (i.e., Does it have promising [strong evidence-based] community-level interventions)?
- To what extent would the potential dementia risk and protective factor, if addressed by the public health community, have strong public health impact (including in diverse communities)?
- Does the potential dementia risk and protective factor have interventions or actions that individual health care providers/teams can recommend to their individual patients?
- Can the potential dementia risk and protective factor be addressed through policy and payment (systemic) changes, either at the federal, state, or private sector level?
Timeline
| March |
|
|---|---|
| April |
|
| May |
|
| June |
|
| July |
|
| August |
|
Want to be a reviewer?
Email: aging@cdc.gov
