The Risk and Costs of Severe Cognitive Impairment at Older Ages: Literature Review and Projection Analyses
Melissa M. Favreault and Richard W. Johnson
Urban Institute
January 2021
Link to Printer Friendly Version in PDF Format (85 PDF pages)
ABSTRACT: Projections of future care needs and costs are difficult because the older population is changing in ways that will likely shape the course of cognitive impairment. This study uses the Dynamic Simulation of Income Model (DYNASIM) to project the risk and costs of severe cognitive impairment at older ages over the coming decades. We project large differences in the chances of ever experiencing severe cognitive impairment for different groups, for example, African Americans, Hispanics, and women. Our estimates of the prevalence and duration of severe cognitive impairment fall within the bounds of prior literature. Lastly, those lower in the income distribution can expect to use most or all of their wealth on care should they become impaired.
This report was prepared under contract #HHSP233201600024I between HHS's ASPE/BHDAP and the Urban Institute. For additional information about this subject, you can visit the BHDAP home page at https://aspe.hhs.gov/bhdap or contact the ASPE Project Officers, at HHS/ASPE/BHDAP, Room 424E, H.H. Humphrey Building, 200 Independence Avenue, S.W., Washington, D.C., 20201; Judith.Dey@hhs.gov, Lauren.Anderson@hhs.gov, Helen.Lamont@hhs.gov.
DISCLAIMER: The opinions and views expressed in this report are those of the authors. They do not reflect the views of the Department of Health and Human Services, the contractor or any other funding organization. This report was completed and submitted in May 2020.
TABLE OF CONTENTS
- Assessment and Measurement
- Incidence and Prevalence Studies
- Longitudinal Studies: Lifetime Impairment Risk, Life Expectancy with Cognitive Impairment, Relationship between Cognitive Impairment and Mortality
- Studies of Cross-Sectional and Lifetime Costs of Cognitive Impairment
- Projection Studies
- Community-Based Studies
- Health and Disability Models
- Spending for Health Care and Long-Term Services and Supports
- Valuing Unpaid Family Care
- Baseline Validation
- Risks
- Costs for Paid Services and the Value of Unpaid Care
CONCLUSIONS AND POLICY IMPLICATIONS
LIST OF BOXES
- BOX 1. Selected Terms Related to Cognitive Impairment
LIST OF FIGURES
- FIGURE 1: Alternative Estimates of the Age-Specific Prevalence of CI at Older Ages
- FIGURE 2: Alternative Projections of the Number of Cognitively Impaired Adults in the US Using Different Measures of SCI, 2010-2060
- FIGURE 3: Comparison of Alternative Estimates of the Age-Specific Prevalence of CI at Older Ages Using 5-Year Age Bands with DYNASIM Projections: Prevalence of Dementia Rises Markedly with Age
- FIGURE 4: Comparison of Alternative Estimates of the Age-Specific Prevalence of CI at Older Ages Using 10-Year Age Bands with DYNASIM Projections: Prevalence of Dementia Rises Markedly with Age
- FIGURE 5: Alternative Projections of Number of Cognitively Impaired Adults in the US Using Different Measures of SCI at Older Ages, Including DYNASIM Projections, 2010-2060
LIST OF TABLES
- TABLE 1: Selected Studies of the Cross-Sectional Prevalence of CI: US and Other Wealthy Countries
- TABLE 2: Selected Studies of the Cross-Sectional Incidence of CI: US and Other Wealthy Countries
- TABLE 3: Selected Studies of the Long-Range Likelihood of CI, Remaining Life Expectancy with CI, Duration of CI, and Other Longitudinal Metrics
- TABLE 4: Selected Studies of the Relationship between CI and Mortality Rates in the US
- TABLE 5: Selected Studies of the Costs of CI in the US
- TABLE 6: Selected Studies of the Prevalence and Intensity of Caregiving for People with CI in the US
- TABLE 7: Estimates from Selected Studies of the Cost of CI in the US: Using Consistent Wage-Indexed Dollars
- TABLE 8: Selected Studies Based on Long-Range Models of Cognitive Status
- TABLE 9: Selected Studies of the Effects of Future Interventions on CI in the US
- TABLE 10: Miscellaneous Community-Based Studies Related to CI and Performance in the US
- TABLE 11: Historical Cross-Sectional Prevalence of SCI at Age 65 and Older, by Sex and Age: DYNASIM 2013-2017 and NHATS 2015
- TABLE 12: Projected Cross-Sectional Age-Specific Prevalence of SCI at Ages 65 and Older: DYNASIM 2013-2055 in Selected Years, by Age
- TABLE 13: Probability of Ever Experiencing SCI and Projected Life Expectancy with SCI from Age 65: By Birth Cohort, Gender, Education, and Race and Ethnicity
- TABLE 14: Average and Distribution of Projected Years with SCI from Age 65, for Those Who Become Severely Impaired: By Birth Cohort
- TABLE 15A: Average Projected Cost of SCI from Age 65 Onward for Paid and Unpaid LTSS (Sum): DYNASIM 1955-1959 Birth Cohorts, by Gender, Education, and Lifetime Earnings Quintile--UNCONDITIONAL
- TABLE 15B: Average Projected Cost of SCI from Age 65 Onward for Paid and Unpaid LTSS (PDV): DYNASIM 1955-1959 Birth Cohorts, by Gender, Education, and Lifetime Earnings Quintile--UNCONDITIONAL
- TABLE 16A: Average Projected LTSS Costs from Age 65 Onward for Those with SCI (Sum): DYNASIM 1955-1959 Birth Cohorts, by Gender, Education, and Lifetime Earnings Quintile
- TABLE 16B: Average Projected LTSS Costs from Age 65 Onward for Those with SCI (PDV): DYNASIM 1955-1959 Birth Cohorts, by Gender, Education, and Lifetime Earnings Quintile
- TABLE 17A: Distribution of Projected Costs of LTSS for Those Who Ever Experience SCI (Sum): DYNASIM 1955-1959 Birth Cohorts, by Gender
- TABLE 17B: Distribution of Projected Costs of LTSS for Those Who Ever Experience SCI (PDV): DYNASIM 1955-1959 Birth Cohorts, by Gender
- TABLE 18: Projected Total LTSS Spending from Age 65 among People with Significant Disabilities and Paid Care Use, by whether Ever SCI: DYNASIM 1955-1959 Birth Cohorts, by Gender
ACKNOWLEDGMENTS
This report was funded by the Office of the Assistant Secretary for Planning and Evaluation (ASPE) at the U.S. Department of Health and Human Services (contract number HHSP233201600024I, order number HHSP23337001T). We are grateful to them and to all our funders, who make it possible for Urban to advance its mission.
The views expressed are those of the authors and should not be attributed to the Urban Institute, its trustees, or its funders. Funders do not determine research findings or the insights and recommendations of Urban experts. Further information on the Urban Institute's funding principles is available at https://www.urban.org/aboutus/our-funding/funding-principles.
Brenda Spillman of Urban's Health Policy Center provided helpful comments on an earlier draft and estimates from the National Health and Aging Trends Study for Table 11. Judith Dey, Helen Lamont, and William Marton of ASPE also provided helpful comments on earlier drafts.
ACRONYMS
The following acronyms are mentioned in this report.
| AD | Alzheimer's Disease |
|---|---|
| ADAMS | Aging, Demographics, And Memory Study |
| ADL | Activity of Daily Living |
| ADRD | Alzheimer's Disease and Related Dementias |
| AHEAD | Asset and Health Dynamics of the Oldest Old |
| APOE | Apolipoprotein E |
| BMI | Body Mass Index |
| BPC | Bipartisan Policy Center |
| CI | Cognitive Impairment |
| CIND | Cognitive Impairment No Dementia |
| CMS | Centers for Medicare & Medicaid Services |
| DLB | Dementia with Lewy Body |
| DNA | Deoxyribonucleic Acid |
| DYNASIM | Dynamic Simulation of Income |
| DYNASIM4 | Dynamic Simulation of Income Model 4 |
| ELSA | English Longitudinal Study of Aging |
| FEM | Future Elderly Model |
| GAO | Government Accountability Office |
| HIPAA | Health Insurance Portability and Accountability Act |
| HRS | Health and Retirement Study |
| HS | High School |
| IADL | Instrumental Activity of Daily Living |
| LTCI | Long-Term Care Insurance |
| LTSS | Long-Term Services and Supports |
| MCBS | Medicare Current Beneficiary Survey |
| MCI | Mild Cognitive Impairment |
| MMSE | Mini-Mental State Examination |
| NHATS | National Health and Aging Trends Study |
| NLTCS | National Long-Term Care Survey |
| PDV | Present Discounted Value |
| SCI | Severe Cognitive Impairment |
| SES | Socioeconomic Status |
| TICS | Telephone Interview of Cognitive Status |
ABSTRACT
The possibility of becoming severely cognitively impaired is among the most consequential risks facing older adults and their families. In addition to the emotional and physical toll associated with dementia, the financial consequences can be overwhelming, as many patients require expensive paid care. Projections of future care needs and costs are difficult because the older population is changing in ways that will likely shape the course of cognitive impairment. This study uses the Dynamic Simulation of Income Model (DYNASIM) to project the risk and costs of severe cognitive impairment (SCI) at older ages over the coming decades. Using multiple data sources and econometric techniques, DYNASIM simulates the future population and its characteristics, projecting financial resources, disability status, medical conditions, cognitive status, and use of long-term services and supports. Unlike some past research, this study shows how SCI and associated costs vary across the population. We project large differences in the chances of ever experiencing SCI for different groups. Those with less than a high school education, for example, are about three-fifths to three-quarters more likely to ever become severely cognitively impaired in late-life than their counterparts with even a high school diploma, despite not living as long. African Americans and Hispanics are also more likely to become impaired than non-Hispanic Whites. Women are more likely to become impaired than men, in large part due to their longer life expectancy. Our estimates of the prevalence and duration of SCI fall within the bounds of prior literature, sometimes higher and sometimes lower. Because the typical spell of SCI lasts about four years, cost and care burdens can be quite significant for families of those who become impaired. Those lower in the income distribution can expect to use most or all of their wealth on care should they become impaired.
INTRODUCTION
The possibility of developing cognitive impairment (CI) is among the most consequential risks older adults and their families face. In addition to the emotional and physical toll associated with dementia, the financial consequences can be overwhelming. As cognitive functioning declines, help with personal care and everyday activities often becomes necessary. Assistance is usually provided by unpaid family members and friends. Paid help, often provided in nursing homes and other residential settings, frequently becomes necessary when patients need round-the-clock care. Most care costs are initially paid out-of-pocket, which can create substantial financial burdens. Once patients with severe cognitive impairment (SCI) exhaust their financial resources they can qualify for Medicaid. As the population ages in coming decades, Medicaid spending on older adults with SCI will likely increase, posing significant financial risks to federal and state governments.
A growing literature examines the incidence, costs, and correlates of SCI at older ages. However, relatively little is known about how SCI risks and costs will likely evolve over the coming decades. Projections of future care needs and costs are difficult because the older population is changing in ways that could alter the course of cognitive impairment. For example, the older population is becoming better educated and more racially and ethnically diverse. At the same time, some health risks--like obesity (Hales et al. 2017) and diabetes (Selvin and Ali 2017)--have grown among older adults and could affect the future trajectory of cognitive impairment.[1, 2] Care delivery options are changing, as residential care and care at home are slowly replacing nursing home care. While long-term productivity growth is raising incomes, the gains have not been shared evenly across the population, and these changes could affect future Medicaid spending.
This study uses the Dynamic Simulation of Income Model (DYNASIM), the Urban Institute's dynamic microsimulation model, to project the risk and costs of moderate and SCI among older adults over the coming decades. Using multiple data sources and econometric techniques, DYNASIM simulates the future population and its characteristics, projecting financial resources, disability status, medical conditions, cognitive status, and use of long-term services and supports (LTSS). Unlike some past research, this study shows how SCI and associated costs vary across the population, with a focus on differences by socioeconomic status (SES). We show how projected experiences with cognitive impairment vary across three birth cohorts and compare outcomes by sex, education, race/ethnicity, and income within each cohort.
Projections, especially over the long term, require many assumptions and are inherently uncertain. Before delving into the projections, we review existing estimates of SCI, including recent incidence and prevalence, projections of future prevalence, duration of impairment spells, and associated care costs. This extensive review enables us to ground the DYNASIM projections and verify their reasonableness and validity. This grounding is important because debates about how to finance LTSS for people living with SCI require a common understanding of assumptions, methods and estimates, so that we can fully understand the tradeoffs between options. Documenting that our projections fall within the mainstream of the social science literature can offer reassurance that they are not biased in any way that would unduly favor any specific policy approach.
MOTIVATION
Our aging population is likely to bring increased attention to long-standing challenges in LTSS delivery and financing in the United States. Several expert groups have tried recently to devise policy solutions to several well-documented problems (BPC 2017; Commission on Long-Term Care 2013; LeadingAge 2016; Long-Term Care Financing Collaborative 2016; U.S. House of Representatives 2016), including the following:
-
Rising health and long-term care costs could increasingly strain public budgets (Congressional Budget Office 2013).
-
Private LTSS insurance is not affordable for many mid-life and older adults, as many carriers exit the market and those remaining increase premiums (Cohen 2016; Schmitz and Giese 2019; Ujvari 2018).
-
Family caregivers provide much of the care to those with severe disabilities, including SCI (Rainville, Skufca and Mehegan 2016; Wolff et al. 2016). Care activities can impose significant financial costs on caregivers, including direct outlays and lost wages as caregivers are forced to curtail paid employment to meet their care responsibilities. In addition, caregiving often takes an emotional toll, raising stress, increasing health problems, and leading to burnout, especially among caregivers providing more extensive assistance, like help with health care.
Some dementia patients in later stages of the disease have complex needs that make them expensive to serve. Facilities serving late-stage dementia patients may require higher staff-to-resident ratios to address their residents' more significant personal care and supervision needs. For example, some dementia patients have behavioral challenges like becoming agitated or physically aggressive in the late stages of the disease.[3] Also, some patients may wander or try to leave their homes or care facilities, requiring extra security to ensure their safety.[4]
Another motivation for our paper is to help policymakers in the states and Federal Government prepare for the coming decades, when the large baby boom cohorts will reach the ages when severe care needs are common.
KEY DEFINITIONS
Box 1 presents concise definitions of key terms surrounding cognitive impairment. We draw the definitions from various sources, including Alzheimer's Association (2016, Table 1), Hugo and Ganguli (2014), and National Academy of Sciences (2017). Throughout this report, we generally discuss cognitive impairment and differentiate between mild and severe cases. Our data generally do not allow us to differentiate between different types of dementia (such as Alzheimer's disease, vascular dementia, and Lewy body dementia), and some people experience multiple types of dementia.[5] When describing estimates from the literature, we generally use the authors' terms and characterizations.
| BOX 1. Selected Terms Related to Cognitive Impairment |
|---|
|
| SOURCE: Definitions derived and adapted from Alzheimer's Association (2016), Centers for Disease Control and Prevention (2011), Hugo and Ganguli (2014), and National Academy of Sciences (2017). Other terms for specific types of dementia--like vascular dementia, frontotemporal dementia, or dementia with Lewy Bodies (DLB)--are described in some of these sources. |
PREVIOUS LITERATURE
In this section, we present detailed tables that describe previous estimates of the historical prevalence and incidence of SCI and projections of future prevalence and lifetime risks and costs from the literature. These tables provide important grounding for the DYNASIM projections.
The tables describe various aspects of cognitive impairment. We cover prevalence, incidence, longitudinal measures of experience (including relative mortality/longevity experience), longitudinal measures of costs (including prevalence and intensity of unpaid care), projections of future experience under baseline assumptions, projections of the potential effects of interventions (including interventions that delay dementia onset or that reduce co-morbidities), and related sensitivity tests, and community-based studies.
Our review covers research mostly from the social sciences, not the medical literature, because we focus on distributional rather than clinical issues. Most studies we discuss describe cognitive impairment in later life, and the vast majority focus on the United States experience. We emphasize recent studies but include some research from the late 1990s for context. Studies that cover multiple outcomes may appear in more than one table.
We structure the tables to facilitate comparisons of disparate studies. They identify the population studied (age range, geographic coverage, inclusion of institutional residents[6]), estimation period, and indicators used to determine cognitive status. We try to use comparable measures in the tables. For example, we express most rates as percentages, even if authors used different metrics in their study, such as reporting incidence per 1,000 people.
Assessment and Measurement
Assessing cognitive status in nationally representative household survey data and even in clinical environments poses many challenges. Some studies use diagnosis data, sometimes self-reported by respondents and sometimes reported by proxy respondents. Others rely on payment codes from matched medical benefit records to classify cognitive status, which may include physician diagnosis information or indicate use of prescription drugs associated with cognitive impairment. Others administer cognitive tests to respondents, sometimes conducted over the telephone. Still others use physician evaluation or medical tests (such as tests of blood or spinal fluid for amyloid plaques). Some studies take images of participants' brains while they are alive and perform autopsies of their brains after death.
Given these different measures and the complexities associated with each, as well as differing diagnostic criteria, estimates of cognitive impairment vary widely (Erkinjuntti et al. 1997). For example, some studies document significant underdiagnosis of dementia (Amjad et al. 2018). Even claims data provide imperfect indicators of dementia (Taylor et al. 2009). Informant reports may differ systematically across groups (Potter et al. 2009). The literature highlights the importance of including proxy reports for those who are either unable to respond to a survey or to take a cognitive test and, in longitudinal surveys, accounting for those who die between waves of data collection because cognitive impairment often occurs near the time of death.
Researchers using the same tests and similar samples sometimes make somewhat different choices about how to interpret scores or use different cutoffs to designate various impairment levels. They often do this to capture subtleties, like differences in populations of interest. For example, several studies described in the literature review tables use data from the Health and Retirement Study (HRS), including the older cohorts in the Asset and Health Dynamics of the Oldest Old (AHEAD) sample that is now part of the HRS. These authors rely on scores from the Telephone Interview for Cognitive Status (TICS) and sometimes also the Aging, Demographics, and Memory Study (ADAMS). As the literature review tables show, authors sometimes use modestly different cutoff values from the TICS, often in response to the age range of the population of interest and respondents' experience with the diagnostic tests. For example, Crimmins et al. (2018), Langa et al. (2016), and Zissimopoulos, Crimmins, and St. Clair (2014) consider adults ages 65 and older and use a cutoff score of 6 to identify SCI. Garcia et al. (2017) include adults ages 50 and older in their sample and also use 6 as the cutoff score for SCI. Langa et al. (2008) study adults ages 70 and older use 7 as the cutoff. Langa et al. (2008) and Lièvre, Alley, and Crimmins (2008) also consider adults ages 70 and older but use 8 as the initial cutoff score; Lièvre, Alley, and Crimmins use a cutoff score of 9 in subsequent waves to account for potential learning effects for respondents who have taken the test more than once.
Consistent with Lièvre, Alley, and Crimmins (2008), other researchers who use longitudinal data to measure cognitive status increasingly use a two-wave criterion for assessing cognitive status to account for the possibility that respondents may learn from participating in the test in earlier waves. These authors include Freedman et al. (2018), who use the National Health and Aging Trends Study (NHATS), and Zissimopoulos et al. (2018) who use HRS.
Some other researchers use relative measures of cognitive impairment that allow threshold scores to vary across the population, such as among people with different levels of education. Ahmadi-Abhari et al. (2017), for example, define impairment as having a cognitive performance that is 1.5 standard deviations below the mean for their broad education group.
Other researchers, including Hudomiet, Hurd, and Rohwedder (2018), use a probabilistic, multi-factor approach to determine whether a study participant is cognitively impaired. They argue that using multiple indicators--such as combining reports of limitations with instrumental activities of daily living (IADLs)--is better than using a single report or the result of a single test. As they note, these approaches are sensitive to assumptions about how multiple factors are incorporated into a composite score.
Incidence and Prevalence Studies
The first two tables provide United States estimates of prevalence (Table 1) and incidence (Table 2) of cognitive impairment and limitation and related outcomes, like Alzheimer's disease. Several of the studies in Table 1--including Choi et al. (2018); Crimmins et al. (2018); Freedman et al. (2018); Hudomiet, Hurd, and Rohwedder (2018); Langa et al. (2008, 2016); Li et al. (2017); Rocca et al. (2011); Stallard and Yashin (2016)--explore whether the prevalence of dementia has declined recently on an age-adjusted basis. Figure 1 presents some of this information in graphical form, but direct comparisons are difficult because studies often compare people in different age ranges.
All the studies examined find that both the prevalence and incidence of cognitive impairment increase rapidly with age.[7] Estimated prevalence rates usually climb from a few percent in one's early 60s to anywhere from 30 to 45 percent in one's 90s, depending on the time period, precise age groupings, and impairment definition. Most studies find higher rates of impairment for women than men, but some exceptions exist on an age-adjusted basis for particular age groups, especially younger ages when men's rates sometimes exceed women's. Studies that report differentials by education generally find much higher rates of impairment for those with less education. Those reporting differences by race and ethnicity usually find higher age-adjusted impairment rates for African Americans and Hispanics than non-Hispanic Whites;[8] the few studies that consider Asians and Pacific Islanders find that they have lower than average age-adjusted impairment rates. A growing literature explores the complexities of these differences by race and ethnicity, including how they vary by nativity and neighborhood (Weden et al. 2017).
Most of the studies attempting to compare prevalence and incidence over time find evidence of meaningful decline at older ages in recent years (Freedman et al. 2018; Hudomiet, Hurd and Rohwedder 2018; Langa et al. 2008; Langa et al. 2016; Li et al. 2017; Rocca et al. 2011; Stallard and Yashin 2016). Exceptions include Choi et al. (2018), who focus on younger ages (55 and 69) and aggregate severe and moderate impairment, and several community-based--rather than nationally representative--studies, which we describe in a later table. A recent comparative study (Wu et al. 2017) that reviews consistent population-based prevalence and incidence studies for eight countries (France, Japan, the Netherlands, Nigeria, Sweden, Spain, the United Kingdom and the United States) finds evidence of stable or declining prevalence and incidence in all countries except Japan, where some studies suggest a reverse pattern. However, results are inconclusive due to differences in study designs.
Longitudinal Studies: Lifetime Impairment Risk, Life Expectancy with Cognitive Impairment, Relationship between Cognitive Impairment and Mortality
Table 3 describes projections of lifetime or cumulative old-age risk of cognitive impairment, including the risk of ever being impaired, and conditional or unconditional durations of impairment,[9] as available. It also includes other longitudinal measures, like average age at impairment onset. The studies vary in terms of birth cohorts and the starting ages of the samples examined and whether they include people in nursing homes, so direct comparisons are difficult. Brodaty, Seeher, and Gibson (2012) point out the importance of making consistent comparisons that account for selectivity of samples.
Most studies that restrict their samples to adults who survive to age 65 estimate remaining lifetime risks of cognitive impairment in the 15-35 percent range, in most cases with significantly higher risk for women than men. Because most authors estimate that no more than a third of people who survive into later life can expect to experience SCI, unconditional durations--the average time impaired including both those who are never impaired and those who become impaired--of cognitive impairment are much lower than conditional durations--average time impaired for those who are ever impaired. Unconditional estimates of dementia duration from age 65 cluster around 1-3 years, whereas conditional durations from age 65 range from 4-6 years in most studies. There may be important differences between mean and median experiences, but the literature is not entirely conclusive. These estimates are sensitive to definitions and the estimation time period. People in earlier cohorts have shorter total life expectancy, and survival differences between those with and without SCI may be shifting (Dufouil et al. 2018; Langa et al. 2008).
Few of these studies show within-group distributions, which would provide useful information for understanding relative risk. Again, where reported, differences between education groups are often dramatic, with less-educated adults experiencing much larger shares of late-life with moderate or SCI than their better-educated counterparts. Crimmins et al. (2018), for example, report that in 2010 a 65-year-old woman without a high school education could expect to spend more than half (53.8 percent) of her remaining life cognitively impaired (21.8 percent mildly impaired and 32.0 percent severely impaired), compared with only about 20 percent for her college-educated counterpart (12.7 percent mildly impaired and 7.0 percent severely impaired). Garcia et al. (2017) find large differences by race and gender in life expectancy from age 50 with dementia and CIND and in the share of remaining life expectancy impaired/unimpaired for people in the community. For example, non-Hispanic White men can expect to spend 1.1 years from age 50 with dementia and 17 percent of remaining life at least mildly cognitively impaired. Foreign-born Hispanic women can expect to spend 6.0 years with dementia and 49 percent of remaining life at least mildly cognitively impaired.
Implicit in many of these projections of long-range experience with cognitive impairment are mortality differentials between those who are cognitively impaired and those who are not. Table 4 takes a closer look at some of the studies in this area. Davis et al. (2018), James et al. (2014), and Wilson et al. (2009) find much higher mortality among those with cognitive impairment than those with good cognition. Estimates vary by age, and those with lower levels of cognitive impairment have lower mortality differentials than those with more severe impairments. Racial differentials in survival among those severely cognitively impaired may be important. Both Helzer et al. (2008) and Mayeda et al. (2017) find that non-Hispanic Whites have shorter survival times than others who are cognitively impaired. There are also large differences based on age at onset or diagnosis of dementia. Some estimates, such as Brookmeyer et al. (2002), Ganguli et al. (2005), Johnson, Brookmeyer, and Ziegler-Graham (2007), and Mayeda et al. (2017), report that those who become impaired early in life can expect to live much longer than those who become impaired later, with important implications for the burdens their family caregivers face.
Studies of Cross-Sectional and Lifetime Costs of Cognitive Impairment
Table 5 reports studies that attempt to estimate the costs of dementia in the United States, including valuations of the unpaid care provided by family caregivers.[10] Table 6 provides additional information about the prevalence and intensity of care provided to people with dementia. Table 7 converts the cost estimates from Table 5 into consistent 2020 wage-indexed dollars to facilitate comparisons across studies, which use different time periods that range from the early 1990s to 2017. In these papers, most authors attempt to distinguish private costs, including the value of uncompensated care provided by family caregivers and public costs paid by Medicaid and Medicare. When valuing uncompensated family care, authors use a variety of estimates, usually centered on the replacement cost of care, proxied by the average wages paid to agency home health aides or other hands-on care workers.[11]
Important differences across these studies include whether and how they distinguish between costs for medical care and costs for LTSS and how they address the cost of co-morbidities. Some estimates include the total costs incurred by patients with dementia, whereas others focus on the added costs of dementia for people who also receive care for other conditions and impairments. Langa et al. (2010) report these additional costs; Hurd et al. (2013) report both adjusted and unadjusted estimates to allow readers to compare. Answers to both questions are important. For policymakers interested in how much dementia contributes to public expenses and how changes in dementia onset and prevalence might affect public spending, the measure of additional costs from dementia may be most useful. For households planning for retirement, it may be more helpful to know the total financial risk associated with dementia--and to have as much information as possible about out-of-pocket health care spending risk in retirement more broadly.
The methods these studies use--especially those studies that focus on net costs--can be complex. For example, several studies match people with dementia to otherwise similar people without dementia to try to determine the added costs that can be attributed to the disease; others simulate the additional effects of dementia on spending by using coefficients for dementia's added effects from regression equations used to project spending.
Incomplete and low-quality data also pose challenges for research in this area. Some studies use rich data from payment records, which are likely to be more accurate than self-reported cost data. One important limitation of many of these data is that they often cover only Medicare beneficiaries who are enrolled in fee-for-service plans, excluding those enrolled in Medicare Advantage plans, who now comprise about a third of aged Medicare beneficiaries (Jacobson, Damico and Neuman 2018). People enrolled in Medicare Advantage plans may have different economic and health characteristics than those enrolled in traditional Medicare with Medigap supplemental insurance. Literature suggests that people with higher health care costs and needs are more likely to enroll in traditional Medicare and to switch from Medicare Advantage to traditional Medicare when their health worsens (Neuman and Jacobson 2018; Rahman et al. 2015).
Another important limitation is that payment records may not capture significant amounts of home care, given that families pay for it in private transactions, often to non-medical providers (Newquist, DeLiema and Wilber 2015; Seavey and Marquand 2011). Such care is understudied and many important government statistics exclude it, potentially biasing estimates of the share of care financed by various payers and understating the role of families' out-of-pocket payments (Newquist, DeLiema and Wilber 2015).
Findings from these analyses of the costs associated with cognitive impairment are mixed. Most researchers conclude that dementia raises Medicaid and out-of-pocket spending more than Medicare spending. However, estimates differ depending on how they account for co-morbidities and compositional differences between those who do and do not develop SCI. Cost impacts vary as the disease progresses, with excess Medicare costs more prevalent early in a dementia spell and excess Medicaid costs more prevalent later (see, for example, Coe et al. 2018).
Projection Studies
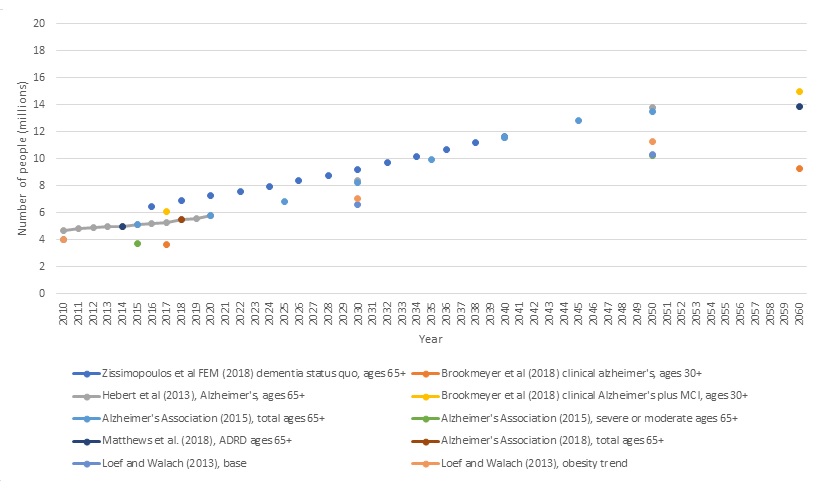
Table 8 describes different forecasting approaches that modelers have taken to project lifetime or longitudinal risk and costs of dementia. Figure 2 compares selected projections from six of the studies we describe. (Because three studies report more than one outcome, the figure shows ten projection series.) Estimates of the number of people with SCI vary widely, and these differences cannot be explained by the use of different definitions. In addition, there is no consensus about how the future is likely to evolve. Differences in outcome variables (e.g., Alzheimer's disease, which is a subset of dementia, compared to SCI) partially explain some of the variation across projection studies.
These studies generally use different methods. Some apply prevalence or incidence rates to population projections that often come from federal agencies. The studies use different state spaces, with some considering dementia jointly with cardiovascular disease, others considering disease progression in a narrow way, and others looking more narrowly at recent age-sex prevalence rates, perhaps considering an additional characteristic.
In analyses applied to adults ages 50 and older in England, Ahmadi-Abhari et al. (2017) model cognitive impairment jointly with cardiovascular disease. They use the English Longitudinal Study of Ageing (ELSA), which includes cognitive tests, to estimate transitions between various states, including disease-free, cardiovascular disease without impairment, cardiovascular disease with functional impairment, cardiovascular disease with functional and cognitive impairment, no cardiovascular disease with functional impairment, no cardiovascular disease with functional and cognitive impairment, and death.
Brookmeyer, Abdalla, Kawas, and Corrada (2018) apply incidence rates for various transitions to Census population projections. Their model is grounded in clinical factors and focuses on the progression of disease through clinical stages, including amyloidosis, amyloidosis with neurodegeneration, mild cognitive impairment (MCI), and early clinical or clinical dementia. An important aspect of this study is that the authors present a range for every estimate because precise measurement is impossible. For example, they estimate that in 2017, approximately 3.65 million people in the United States had clinical Alzheimer's disease, with a lower bound estimate of 1.70 million and an upper bound estimate of 7.62 million. In 2060, they project 9.30 million people will have Alzheimer's, with a lower bound estimate of 4.58 million and an upper bound estimate of 17.82 million.
A model developed by the Lewin group and used by the Alzheimer's Association (2015, especially the appendix; 2018) largely relies on secondary data, earlier estimates from high-quality studies. Several of the studies on which it relies are becoming outdated, such as Sloane et al. (2002). The version of the model used in the 2018 paper uses data from the 2011 Medicare Current Beneficiary Survey (MCBS) for projecting costs among those with Alzheimer's disease.[12]
The University of Southern California's Future Elderly Model (FEM) (Goldman et al. 2015; Zissimopoulos, Crimmins, and St. Clair 2014; Zissimopoulos et al. 2018) projects limitations in activities of daily living (ADLs) and cognitive status using HRS data. It thus has significant similarities to the DYNASIM projections, which are also based on HRS data, although some important differences exist.[13]
Matthews et al. (2019) use Medicare payment records for beneficiaries ages 65 and older with fee-for-service claims in 2014. They use diagnosis codes to identify whether people have Alzheimer's disease and related dementias (ADRD). They then construct prevalence rates by five-year age groups, sex, and race-ethnicity, which they apply to Census projections, assuming no decline in future rates. The payment data are incredibly rich, representing over 28 million older United States adults. However, the data exclude more than one-third of Medicare beneficiaries who participate in Medicare Advantage rather than fee-for-service. This raises concerns about the data's selectivity by health status.
Table 9 describes the projected effects of various interventions, such as hypothetical interventions that would delay the onset of cognitive impairment, reduce dementia incidence, or reduce the prevalence of co-morbidities (such as diabetes) as well as sensitivity analyses. Most of the studies focus on positive interventions, including efforts at prevention. The study by Zissimopoulos et al. (2018), however, underscores the interplay between survival and dementia risk. For example, they conclude that reducing diabetes by 50 percent or eliminating hypertension would increase the number of people with dementia because more people would survive to older ages, when dementia rates are high, under both scenarios; the increase in the at-risk population would more than offset the reduction in co-morbidities. This is inconsistent with projections by Barnes and Yaffe (2011), which rely on simpler methods to estimate the effects of a diabetes reduction on cognitive impairment. Yang et al. (2012) consider the potential effects of increased obesity; they find that the life expectancy losses do not offset increased dementia prevalence, boosting care costs. Given the complexity of these types of analyses because of the competing risks older adults face, this literature deserves further monitoring.
Community-Based Studies
Table 10, our final literature review table, describes several analyses of data from community-based studies. (We also reference some of these studies in earlier tables when they report other metrics, such as lifetime risk, or are used to estimate projection-model inputs, such as incidence rates.) These studies do not generate nationally representative estimates, but they often use better methods than studies using national data to verify the presence of impairment or disease. For example, some studies make use of expert review panels that examine every case to determine if a participant's disease or impairments meet certain clinical criteria. Some even examine participants' brains after they die (Bennett et al. 2013). Community-based studies also sometimes use longer follow-on periods than other studies. For example, the Massachusetts-based Framingham Heart Study enables researchers to look at changes in dementia from as far back as the late 1970s, whereas the nationally representative HRS survey began only in the early 1990s.[14]
Several of these studies find little or no evidence of decline in incidence and prevalence of cognitive impairment in recent decades (Hall et al. 2009; Hebert et al. 2010; Rajan et al. 2019; Weuve et al. 2018), contrary to several of the nationally representative studies.
PROJECTION METHODS
DYNASIM starts with a nationally representative population based on the 2004 and 2008 panels of the Survey of Income and Program Participation. The model "ages" the population year-by-year, simulating demographic and economic events using transition probabilities and rule-based algorithms. The model's aging rules include socioeconomic differences--such as by education, lifetime earnings, marital status, and race and ethnicity--when projecting health and mortality, using rich longitudinal data. DYNASIM projections capture compositional change in the population for the next seven decades. Many outcomes are calibrated to the intermediate assumptions of the 2019 Social Security Trustees Report (Board of Trustees, Federal Old-Age and Survivors Insurance and Disability Insurance Trust Funds 2019), typically on an age-sex basis, with the underlying behavioral equations driving differences by other characteristics, such as education, within age-sex groups. Because we rely on historic data and 2019 trustees report assumptions, the projections do not account for the 2020 novel coronavirus outbreak's on-going demographic and economic effects.
Health and Disability Models
DYNASIM's underlying model of health status and disability from age 51 onward considers the progression of health change and disability, using a structure that recognizes that these processes are dynamic; people develop limitations, but sometimes recover. Disability onset can be either sudden or gradual. How rapidly disability progresses can have important implications for the ability to work or care for oneself and thus meet financial needs. The model, estimated using HRS data, integrates occurrence and duration dependence in health and disability outcomes.[15] The model projects self-reported health status, limitations with ADLs and IADLs, counts of chronic conditions (with stroke modeled separately from other conditions given its close association with cognitive decline[16]), and cognitive status. Projections from the historical period are calibrated to estimates from the MCBS and NHATS.
Our approach for forecasting cognitive impairment in DYNASIM is grounded in estimates that rely on HRS data through 2014.[17] We use data from the TICS when it is available. Our models of cognitive status transition include information about those who cannot take the cognitive test or who die, integrating proxy and exit interviews, as this information significantly affects estimates of cross-sectional prevalence and lifetime dementia risk.[18] We model cognitive status using transitions between three discrete states: good cognition, CIND, and dementia. We use a score of 7 as our cutoff threshold for respondents who took the test regardless of age; as noted above, some other researchers use higher cutoff values, some use lower values, and some use a two-wave criterion. To identify CIND, we use scores from 8 to 13. In addition to projecting a cognitive function category, we project continuous values for the level of cognition, as proxied by the TICS score, so that model users can develop estimates for policies that modify eligibility thresholds for benefits in subtle ways.
We integrate this empirical model into DYNASIM. The model generates forecasts of cognitive status using compositional change, such as population aging and the increased educational attainment of older adults, as drivers of change. We assume that future disability rates will depend on relative age (i.e., proximity to death) rather than absolute age, so that healthy life expectancy will also increase as life expectancy rises. Historical SES differentials in disability are assumed to persist, but not grow. We calibrate mortality to intermediate assumptions of the Social Security trustees report. Mortality differentials by SES are expected to continue growing for the next 15 years and then persist at that level indefinitely.
The final model development stage is to calibrate the micro-dynamic equations for cognitive impairment to match age-specific NHATS data from 2015 to projections for the year 2015. This typically entails searching for sources of discrepancies between the estimates and reconciling them to maintain as much consistency as practical. We also calibrate the relationship between cognitive impairment and mortality to adjust for censoring in some of the cases we used for estimating cognitive impairment.
The model projects cognitive and functional status every year from 2007 through 2090. Starting our projections in the past enables us to test how well the model performs relative to historic estimates, serving as an important validity test for the econometric specification.
Because of this long projection horizon and the annual projections, we can readily and easily use the model to consider both cross-sectional outcomes (such as the number of people impaired, incidence and prevalence rates, and annual costs) and longitudinal outcomes (such as the share of older adults ever impaired, the unconditional and conditional duration of impairment, the average age at impairment, lifetime costs, and so forth).
Spending for Health Care and Long-Term Services and Supports
Our health spending models, which include projections of both acute care and LTSS, account for the close relationship between health status, disability, technological change, and health care spending. They also capture how income and out-of-pocket cost burdens affect health care spending.[19] Those with more income tend to be healthier, which reduces their spending. Because they are also less burdened by health care costs, they are more likely to purchase higher-quality or discretionary services and less likely to skip necessary ones. The model generates Part B, Medigap, and Medicare Advantage premiums endogenously, depending on the distribution of the population's health and disability status, income, and services use. In modeling insurance choices and premiums, those with greater health problems are more likely to opt for more comprehensive coverage. This phenomenon, known as adverse selection, raises costs and drives many of those in better health to seek lower-cost alternatives.
In our models of LTSS utilization, families choose whether to seek paid care. Decisions about different types of paid care--home care, nursing home, and residential care--are made jointly. For families who choose to purchase paid care, prices are set based on the state-specific median for each service type (Genworth 2019), with some small adjustments based on income, presence of long-term care insurance (LTCI), and severity of disability.[20] We model eligibility for Medicaid using program rules for all 50 states. Medicaid participation is higher among those with greater economic and disability needs. We base prices for Medicaid-covered services on recent published data (Hansen Hunter and Company 2018). Medicaid is the payer of last resort under current law, so we also check whether people have other forms of coverage, like private LTCI. If a person holds an unlapsed plan, we assume that the insurance pays up to the plan's daily maximum for as long as the person is eligible. (People are assigned plans based on their availability at the time of purchase). People are also assigned to U.S. Department of Veterans Affairs care using simple models from MCBS and care under the Older Americans Act from simple look-up tables based on published reports. Consistent with our brief on LTSS more broadly (Favreault and Dey 2020), we do not include any post-acute care to people with LTSS needs that Medicare covers in these analyses.
Valuing Unpaid Family Care
When families provide care, they forgo leisure and may also miss out on the opportunity to work more hours; some may need to leave work completely. Policymakers may have an interest in the overall contribution caregivers make in providing LTSS. We therefore provide estimates of the value of LTSS provided by unpaid family caregivers. The current analyses assume that an hour of unpaid care has a value equal to the state-specific median hourly wage for home care workers; this is sometimes referred to as a replacement cost framework. We are not at this stage modeling unpaid care for people with SCI explicitly; instead, we compare the share of the year one is severely disabled with the number of days and hours of paid services they receive to get a sense of the potential use of unpaid care. We then calibrate the estimates based on disability level and reports from the HRS. In forthcoming work, we will develop more sophisticated projections of unpaid family care.
PROJECTION RESULTS
We next describe the DYNASIM projections, illustrating how the model can be used to evaluate both outcomes at different points in time and also accumulated over one's retirement years. We start with projections of the share of people living with SCI at several points. We then describe how long people can expect to live with SCI and also their expected care costs and the value of unpaid care their families and friends provide. In describing care costs, we express values two ways: as the sum of expected costs expressed in current dollars (i.e., real costs as of 2020) and then as the present discounted value (PDV) of real costs. The present value takes into account the interest that funds set aside to pay for future care could accrue. We use two measures because sums are more intuitive to some readers, but the academic literature more commonly uses present values, facilitating comparisons with prior estimates. As we discuss the projections, we compare results to the most similar studies in the literature.
Baseline Validation
Validation analyses of the historical period are important to build confidence in our future projections. We begin, then, by comparing DYNASIM projections to related studies. We first show how the historical prevalence estimates projected in DYNASIM match NHATS data. DYNASIM's projected age-sex-specific prevalence of SCI at ages 65 and older for 2013-2017 generally mirror the corresponding 2015 estimates from NHATS (Table 11).[21] The prevalence of SCI climbs rapidly with age. Age-specific proportions for men and women are broadly similar, with men's prevalences possibly a bit higher at younger ages and women's a bit higher at older ages. Overall, women have a higher prevalence of SCI at ages 65 and older than men, primarily because women are more likely to survive to older ages when rates of SCI are highest. The ratios of the age-specific estimates from DYNASIM and NHATS are generally close to one, indicating reasonably close correspondence, with a few groups that may deserve additional attention, such as women ages 70-74.[22]
Figure 3 and Figure 4 likewise compares DYNASIM projections to related studies. When we compare DYNASIM's historic (2013-2017) prevalence projections with those from studies that use five-year age bands (Figure 3), DYNASIM's projections generally fall on the high end, except at ages 90 and older. When we compare DYNASIM's prevalence projections with those from the literature that use ten-year age bands (Figure 4), DYNASIM's projections generally fall on the lower end. Importantly, although DYNASIM's projections are lower than several estimates from other literature at older ages, they are higher than the NHATS estimates. Our conclusion from these three sets of comparisons is that DYNASIM's projections fall within range of the best literature from the literature, but that we should continue to monitor the literature given the wide range of existing estimates.
Table 12 shows the DYNASIM projections over a longer time horizon--2013-2055--and reports both the age-specific prevalences of SCI and the projected total number of people with dementia. Over time, the age-specific prevalences of SCI generally decline modestly. Prevalence is projected to continue to increase steadily with age in all years reported.[23] Importantly, even though some of the age-specific prevalences fall, the projected rates are typically less than 15 percent lower than the NHATS estimates we report in Table 11. The largest reductions are roughly in line with the declines over the 20 years from 1984 to 2004 reported by Stallard and Yashin (2016). The reductions in age-specific prevalence of SCI very closely track the education distribution of the population--as the share of people without a high school diploma declines and the share with college increases, the rates of SCI fall.
Even as age-specific prevalences fall, the numbers of people impaired increase steadily as the population ages, especially as the large post-war baby boom cohorts reach ages at which age-specific SCI rates are highest. Moreover, the number impaired increases as exposure to the risk of cognitive impairment rises with increased life expectancy. For example, the Social Security Trustees project in their 2019 annual report that a man reaching age 65 in 2018 could expect to live another 18.91 years, and a man turning 65 20 years later could expect to live another 20.23 years. The corresponding estimates for women are 21.43 years and 22.61 years. For recent years (2013-2017), the model projects that roughly 3.8 million people ages 65 and older were severely cognitively impaired. We project that this number will rise to 4.3 million older adults in 2025--an increase of about 500,000 people--5.9 million people in 2035, nearly 7.0 million people in 2045, and 7.2 million people in 2055.
Figure 5 places these DYNASIM projections, as well as our projections for moderate cognitive impairment--in the context of the earlier literature. It repeats the projections shown in Figure 2 and adds confidence intervals for earlier projection studies that report them, such as Brookmeyer et al. (2018). The figure reveals that the DYNASIM projections tend to fall within a reasonable range of other published projections. The DYNASIM projections are higher than all of Brookmeyer et al.'s lower bound projections and roughly track those of the Alzheimer's Association (2015) and Hebert et al. (2013) in earlier years. They are lower than some others, such as Zissimopoulos et al. (2018), and the later-year projections of Hebert et al. (2013). The differences between these projections deserve further examination, but they are likely largely driven by the fact that the DYNASIM projections take careful account of changing socioeconomic variables. Increases over time in the educational attainment of older adults substantially reduce projected rates of dementia in DYNASIM.
Importantly, Figure 5 underscores that the number of people living with moderate cognitive impairment will also grow markedly in coming decades. We estimate that about 3.8 million people are living moderate cognitive impairment in recent years and project that this will more than double--to over 7.5 million--in 2060. DYNASIM's combined projection of people with moderate or severe SCI in 2060 is very close to Brookmeyer et al.'s intermediate projection (2018).
Risks
Mirroring the Table 3 findings from our literature review, we next describe projections of the likelihood of ever having dementia and the remaining life expectancy from age 65 with cognitive impairment (Table 13). We report the mean unconditional remaining life expectancy from age 65 with dementia, which includes people who never develop dementia. We show how these outcomes vary by birth cohort, gender, education, and race and ethnicity.
DYNASIM projects that about a third of all people who survive to age 65 will eventually become severely cognitively impaired. The estimate is a bit lower (31 percent) for the earliest cohort--those who just turned 65 or are about to turn 65--and a bit higher (33 percent) for those who will reach age 65 in about 20-30 years. Women are more likely than men to ever become severely impaired, those without a high school diploma are significantly more likely to be impaired than those who attended college, and those with only a high school diploma are somewhat more likely to be impaired that those who attended college but much less likely than high school dropouts. DYNASIM projects that non-Hispanic Blacks and Hispanics are much more likely to ever become severely cognitively impaired than non-Hispanic Whites; in the 1973-1977 birth cohort, more than two-fifths of non-Hispanic Blacks and Hispanics are projected to become impaired, compared with a quarter of non-Hispanic Whites. Compared with earlier studies, DYNASIM's overall estimate of shares severely impaired at some point from age 65 exceeds the projected shares in Chêne et al. (2015), Murtaugh, Spillman, and Wang (2011) and Yang and Levey (2015) and falls just below the projected shares in Zissimopoulos et al. (2018).
These projected differences in the chances of ever becoming severely impaired are reflected in the projections of unconditional remaining life expectancy with SCI at age 65. Within the entire older population, people can expect to live about 1.5 years with SCI in the oldest cohorts and about 1.7 years in the youngest. Women in the oldest cohorts reaching age 65 over the next few years can expect to live 1.7 years with SCI, compared with 1.4 years for men. In the youngest cohorts, the corresponding projections are 2.0 and 1.5 years, respectively. Differences by education are again stark, with those in the oldest cohort who lack a high school diploma expected to live about 2.7-3.3 years with SCI, compared with 1.5-1.6 years for those with only a high school diploma who never attended college. Racial and ethnic differences in expected life with SCI from age 65 onward are also very large, close to 2.5 years of remaining life expectancy impaired for non-Hispanic Blacks and Hispanics, compared to closer to 1.2 years among non-Hispanic Whites.
Placing these estimates in the context of our literature review, we see that DYNASIM's projected unconditional duration with SCI from age 65 from DYNASIM exceeds those reported in Stallard (2011) and Stallard and Yashin (2016) and falls short of the duration reported in Zissimopoulos et al. (2018).
Table 14 reports the mean and median conditional remaining life expectancy impaired, which is the amount of time that those who are impaired can expect to live with the disease. The mean duration is about five years, but the median is closer to 3.5-4 years. The DYNASIM mean is close to the estimates reported by Murtaugh, Spillman, and Wang (2011) and Zissimopoulos et al. (2018), but lower than reported in Ganguli et al. (2005).[24] The difference between the mean and median values reflects the skewness of the distribution, with a relatively small number of older adults experiencing very long dementia spells.[25] Table 14 presents this distribution of years with SCI. We see about 41-42 percent have durations of at least five years in the three cohorts. Conditional on experiencing SCI, differences across subgroups are modest (not reported). This is consistent with some prior literature. Ganguli et al. (2005), for example, report that men and women have similar conditional durations. However, some other literature suggestions important differences in conditional duration of cognitive impairment for women and men.
Costs for Paid Services and the Value of Unpaid Care
Tables 15A-B and Tables 16A-B present our projections of the LTSS costs (not including medical spending) that those with dementia incur and describes who pays these costs, focusing on those now entering retirement. The first cost tables (Table 15A and Table 15B) presents the unconditional costs for all those who survive until age 65, first as a sum and then in present value terms. The second (Table 16A and Table 16B) present the conditional costs for those who survive until age 65 and become severely cognitively impaired at some subsequent age, first as a sum and then expressed as a PDV.
Average paid LTSS costs are about $86,000 expressed as a sum (Table 15A) and $50,000 expressed as a present value (Table 15B). Consistent with prior literature, we find that unpaid care from families and friends plays a critical role--comparable in scope to paid care. By our estimate, care partners to people with SCI provide care that, when valued at replacement cost, is similar to paid care: about $74,400 as a sum (Table 15A) or $49,300 in present value terms (Table 15B). Both women's paid costs and the value of their unpaid care exceed men's, but the gap is much larger for paid care. Costs are higher for those with less education and lower incomes, and these groups also receive more unpaid care. Families and the Medicaid program shoulder most of the LTSS costs of paid care for people with dementia. Families pay larger shares of the total for those with more education and more income, while Medicaid pays larger shares for those with less education and lower incomes.
Conditional on having dementia, the paid costs more than triple--to about $282,000 when expressed as a sum (Table 16A) or $164,000 as a PDV (Table 16B). The value of unpaid care that care partners provide--close to $243,500 as a sum or $161,300 in present value--again mirrors the paid costs. Families' additional out-of-pocket expenses average $157,500 ($89,400 as a present value)--less for those in lower-income quintiles, where people with SCI are more likely to qualify for Medicaid, and more in higher-income quintiles.
Women's total paid LTSS costs during periods of SCI are higher than men's. However, men receive roughly the same amount of unpaid care as women--largely because men are more likely than women to receive spousal care. Given that on average wives are a few years younger than their husbands and women outlive men, more women survive and are healthy enough to provide care to a spouse with severe disabilities.
Placing our estimates in the context of the prior literature, those who compare paid care and unpaid care often find a roughly equal split (Hurd et al. 2013)--consistent with our findings.[26] Our unpaid care estimate is comparable to Jutkowitz et al.'s base case estimate, which values lifetime family care at about $158,000 (2017), compared to our present value estimate of $161,000.
Because extreme values can distort averages, it is helpful to examine full distributions. Table 17A and Table 17B show the cost distribution for paid services and the distribution of the value of unpaid family care for those with SCI in the 1955-1959 birth cohorts. We see that costs are skewed, with a significant right tail for both paid costs and the value of unpaid care. Over one in three (35.9 percent) of those ever experiencing SCI will incur paid costs of more than $250,000 in real terms (or 24 percent in present value terms). When we consider the value of family care, similarly close to 37 percent will need the equivalent sum of at least $250,000 in care when valued at replacement cost (or 20 percent in present value terms).
Table 18 provides context for how costs for people with dementia who use paid care differ from costs for people with other significant disabilities who use paid care.[27] DYNASIM projects that costs for a person with dementia in the 1955-1959 birth cohorts who uses paid care should be about 27-29 percent higher than costs for a person with disabilities other than dementia, depending on measure. Broyles et al. (2020) provide additional information on how the late-life trajectories of people with and without cognitive impairment vary.
CONCLUSIONS AND POLICY IMPLICATIONS
As our population ages, families and governments can expect to face increased demands for services from older adults with SCI. Although recent trends suggest that the age-specific prevalence of SCI may be declining modestly (Freedman et al. 2018; Hudomiet, Hurd and Rohwedder 2018; Langa et al. 2008; Langa et al. 2016; Li et al. 2017; Rocca et al. 2011; Stallard and Yashin 2016), increases in the number of older adults at the oldest ages, when cognitive impairment risks are especially high, are likely to raise the number of people with SCI who need care. Simply because the population is aging policymakers should prepare for an increase in the number of people with significant, often hard-to-serve LTSS needs. Our best projection is that between 2015 and 2055 the population ages 65 and older with SCI will nearly double, from about 3.9 million adults to 7.3 million; if we include those with moderate cognitive impairment, the growth is from 7.5 million to 14.7 million adults.
On an individual level, the risk of becoming severely cognitively impaired at older ages is significant. Estimates from the literature vary significantly. Our best estimate is that about one in three of today's working-age adults who survive to age 65 will become severely cognitively impaired before they die, and 40 percent of those who do become impaired will need care for five or more years. Those with extended spells will face heavy care burdens. People who develop dementia at early ages are especially vulnerable.
Importantly for public policy, those who are most likely to become impaired and who experience the longest impairment spells tend to have limited education and thus low lifetime earnings. African Americans and Hispanics are also at especially high risk, and they also have relatively low lifetime earnings (Favreault 2018). The higher risk among those with the lowest lifetime earnings is likely to limit the ability of prefunding and private market solutions to address the country's LTSS financing challenges. Public solutions, whether through changes to Medicaid or broader social insurance will continue to be examined by policymakers. At the same time, states are beginning to move forward with new programs and reforms. Innovative state initiatives, including the enactment of legislation in Washington State to create a new public insurance program for a front-end long-term care benefit and new public support for caregivers in Hawaii, are two examples.
FIGURES AND TABLES
| FIGURE 1. Alternative Estimates of the Age-Specific Prevalence of CI at Older Ages |
|---|
 |
| SOURCE: Brookmeyer et al. (2011), Freedman et al. (2018), Herbert et al. (2013), Hudomiet, Hurd, and Rohwedder (2018), Hurd et al. (2013), Li et al. (2017), Matthews et al. (2019), Prince et al. (2015), Rajan et al. (2019), Spillman estimates (see Table 11), Stallard and Yashin (2016), and Zissimopoulos et al. (2018). NOTES: Studies vary widely in definitions, populations, age groups, and time periods reported, and data and methods used. See Table 1 for details. |
| FIGURE 2. Alternative Projections of the Number of Cognitively Impaired Adults in the US Using Diferent Measures of SCI, 2010-2060 |
|---|
 |
| SOURCE: Alzheimer's Association (2015), Brookmeyer et al. (2018), Herbert et al. (2013), Loef and Walach (2013), Matthews et al. (2019), and Zissimopoulos et al. (2018). NOTES: Studies vary widely in definitions, populations, time periods reported, and data and methods used. See Table 8 for details. |
| FIGURE 3. Comparison of Alternative Estimates of the Age-Specific Prevalence of CI at Older Ages Using 5-Year Age Bands with DYNASIM Projections: Prevalence of Dementia Rises Markedly with Age |
|---|
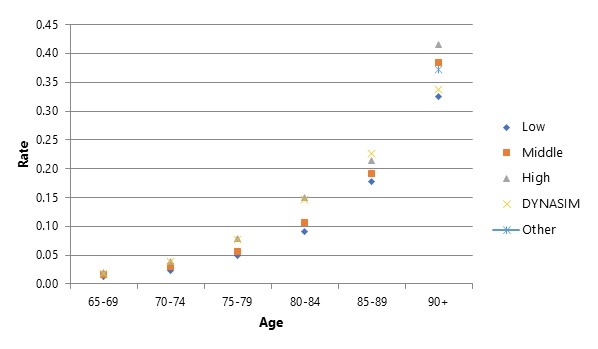 |
| SOURCE: Authors' Calculations from DYNASIM and Brookmeyer et al. (2011), Hurd et al. (2013), Li et al. (2017), Prince et al. (2015), and Stallard and Yashin (2016). NOTES: As in Table 11, DYNASIM projections cover the period 2013-2017. Because the age-specific estimates from the studies do not follow a consistent order, we rank them from highest to lowest within each age band rather than labeling each study. Studies vary widely in definitions, populations, and time periods reported, and data and methods used. See Table 1 for details. |
| FIGURE 4. Comparison of Alternative Estimates of the Age-Specific Prevalence of CI at Older Ages Using 10-Year Age Bands with DYNASIM Projections: Prevalence of Dementia Rises Markedly with Age |
|---|
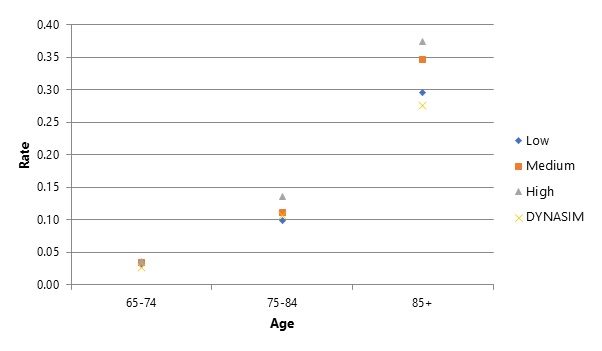 |
| SOURCE: Authors' Calculations from DYNASIM and Brookmeyer et al. (2011), Hurd et al. (2013), Li et al. (2017), Prince et al. (2015), and Stallard and Yashin (2016). NOTES: As in Table 11, DYNASIM projections cover the period 2013-2017. Because the age-specific estimates from the studies do not follow a consistent order, we rank them from highest to lowest within each age band rather than labeling each study. Studies vary widely in definitions, populations, and time periods reported, and data and methods used. See Table 1 for details. |
| FIGURE 5. Alternative Projections of Number of Cognitively Impaired Adults in the US Using Different Measures of SCI at Older Ages, Including DYNASIM Projections, 2010-2060 |
|---|
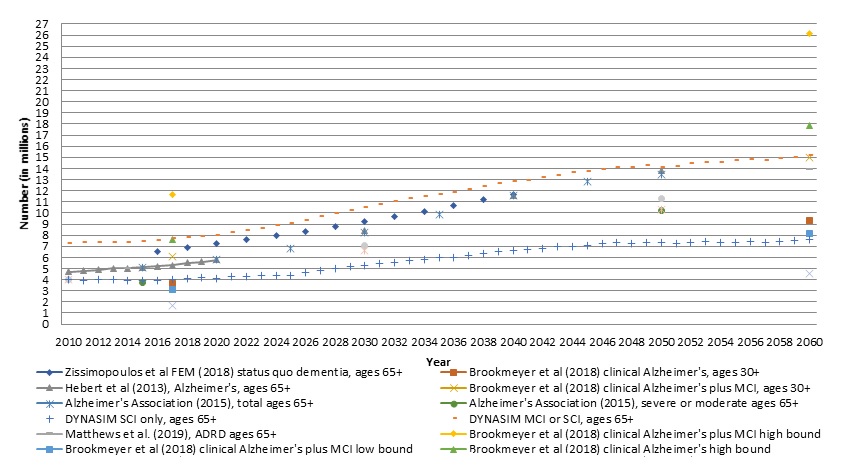 |
| SOURCE: Alzheimer's Association (2015), Brookmeyer et al. (2018), Herbert et al. (2013), Loef and Walach (2013), Matthews et al. (2019), and Zissimopoulos et al. (2018). NOTES: Studies vary widely in definitions, populations, time periods reported, and data and methods used. See Table 6 for details. |
| TABLE 1. Selected Studies of the Cross-Sectional Prevalence of CI: US and Other Wealthy Countries | |||
|---|---|---|---|
| Study | Data, Sample Definitions, Timing, Ages | Age-Specific Prevalence or Trend, with Gender Differences | Other Group-Level Differences |
| Brookmeyer et al. (2011) | Focuses on ADAMS, based on the HRS, 2000 and 2002 (N=856), but compares to other sources, including Chicago Health and Aging Project, East Boston Senior Health Project, and other published studies. Choice of disease definition may account for prevalence differences across studies. Differences in study designs limit ability to make inferences. |
| Overall prevalence is higher for women than men, but gender differences vary across age groups. |
| Choi et al. (2018) | HRS, 1998 and 2014 (N=76,972 person-years); focus on younger people (ages 55-69), using 3 age bands: 55-59, 60-64, and 65-69. Defines cognitive limitation as TICS score less than 12. Dementia prevalence is too small to measure at those ages. Models control for age, gender, previous testing. Main analyses exclude proxies for respondents not living in the community; sensitivity analyses consider these exclusions' importance. Examines 3 race/ethnic groups (non-Hispanic White, non-Hispanic Black, and Hispanic), education, income, and wealth quartiles. | Finds no evidence of cognitive limitation prevalence decline (11.3% in 1998 versus 11.2% in 2014) or narrowing of SES differentials in this age range. | Impairment rates are as much as 10 times) for lower-SES groups (income, education, wealth) than higher-SES groups. Rates are about 4 times higher for non-Hispanic Blacks than non-Hispanic Whites. |
| Crimmins et al. (2018) | HRS, 2000 (N=10,374) and 2010 (N=9,995), ages 65+, classifies respondents as CIND or dementia based on cognitive tests and additional indicators (cognitive score, neuropsychological assessment, dementia diagnosis) for a sample subset. TICS cognitive score cutoffs are 0-6 for dementia and 7-11 for CIND. Includes both community and nursing home residents and respondent and proxy interviews (where cognitive status depends on proxy's assessment, including IADL limitations evaluation). | Dementia prevalence rates (%) in 2010 for not HS graduate, HS graduate, some college, and college:
| |
| Freedman et al. (2018) | NHATS 2011 and 2015 (Medicare population), ages 70 and older. N=27,547 person-year observations--annual sample ranges from 3,748 to 6,454 (sample includes refresher cohorts). Focus on probable dementia. Uses criterion of probable dementia in 2 waves to reduce chances of learning effects. Uses uniform cutoff. Tests for sensitivity of including nursing homes residents in the sample. Considers 3 age groups (70-79, 80-89, and 90+), plus gender, and race/ethnicity. | Probable dementia prevalence (%) in 2015: 4.9 at ages 70-79; 16.1 at ages 80-89; 30.6 at ages 90+. Finds dementia decline of 1.4-2.6% per year between 2011 and 2015. Declines may be larger for women, those without vascular risk factors. | Prevalence much higher for African Americans, Hispanics, those with vascular risk factors, and less-educated adults. |
| Gardner, Valcour, and Yaffe (2013) | Analyzes multiple studies from Canada, Finland, Sweden, and the U.S. published between 1999 and 2011. | Reports prevalence estimates: 18%-38% at ages 85+; 28%-44% at ages 90+. | All studies that report gender differentials find higher prevalence for women than men. |
| Hudomiet, Hurd, and Rohwedder (2018) | HRS, 2000 and 2012 (N is about 10,000), ages 65+. Emphasizes the importance of exit interview data, including both community dwellers and those in nursing homes and both self and proxy respondents. Uses HRS cognition measures (score on TICS), self-reported health and disability measures (IADL limitations) and past diagnoses, and ADAMS sample (N=857) for determining probability. Recommends probabilistic rather than "cutoff" approach and modeling selection into proxy interviews, but approach could be sensitive to underlying assumptions. | Prevalence at ages 65+ declines from 12% in 2000 to 10.5% in 2012. Best age-specific estimate for 2012 (adjusting for proxies, mortality): 3.47% at 65-74 (men: 2.71, women: 4.12); 11.15% at 75-84 (men: 8.97%, women: 12.78%); 37.39% at 85+ (men: 28.11%, women: 42.18%). | Larger percentage reduction from 2000 to 2012 for men and younger people; gender gap quite large at oldest ages. |
| Hurd et al. (2013) | HRS 2000-2008 (N=29,493 person-year observations) linked to Medicare fee-for-service records (about 70% of sample); uses models based on ADAMS, TICS, and proxy reports to assign cognitive status. | Overall dementia prevalence (%): 71+, 14.7; 71-74, 2.8; 75-79, 4.9; 80-84, 13.0; 85-89, 20.3; 90+: 38.5. | |
| Langa et al. (2008) | HRS 1993 (N=7,406) and 2002 (N=7,104), ages 70+ living in the community (excludes nursing home residents). TICS cutoff scores are 7 for SCI and 10 for MCI. | SCI at ages 70+ fell from 12.2% in 1993 to 8.7% in 2002. | Education protects against SCI. |
| Langa et al. (2016) | HRS 2000 (N=10,546) and 2012 (N=10,511), ages 65+; respondents with TICS scores of 0-6 (out of 27) were classified as having SCI, 7-11 as having CIND. For respondents with proxies, uses 11-point scale from proxy's assessment of respondent's memory ranging from excellent to poor (score, 0-4) and IADL limitations (taking medication, preparing hot meals, using phones, grocery shopping; score, 0-5), plus interviewer's assessment of whether respondent had difficulty completing interview because of cognitive limitation (score of 0-2 indicating none, some, and prevents completion). Scores from 6-11 indicate SCI and those 3-5 indicate CIND. | Dementia rate at 65+ fell from 11.6% in 2000 to 8.6% in 2012 (age-sex adjusted); on an age-specific, standardized (2000) basis: 65-74: 5.4% to 3.2%; 75-84: 13.5% to 9.9%; 85+: 34.3% to 29.6%. | Documents large difference by gender, race, and education; associations with net worth and BMI. |
| Li et al. (2017) | NHATS 2011 (N=7,609) and 2015 (N=7,499), ages 65+. Defines probable dementia using reported diagnosis of dementia (self or proxy); a low score (at least 1.5 standard deviations below the mean) on a cognition test in at least two domains; for proxy respondents, reporting a score of 2 or higher in Ascertain Dementia 8-item Informant Questionnaire (AD8) interview. Classification validated against 2010 ADAMS diagnosis information. Identifies a sampled person as having SCI if he/she was classified as having probable dementia. |
| Shows gender and education differentials, though not on an age-adjusted basis. Documents differences in ever-diagnosis rates by sample |
| Matthews et al. (2019) | Medicare beneficiaries ages 65+ with fee-for-service spending in 2014. N=28,027,071 (roughly 60% of the 65+ population). However, the sample may not be randomly selected on the basis of health because about 35% of Medicare beneficiaries participate in Medicare Advantage. Uses diagnosis codes.1 | 2014 prevalence: 10.9% overall; 12.2% for women; 8.6% for men; 3.6% ages 65-74; 13.6% age 75-84; 34.6% ages 85+. |
|
| Plassman et al. (2007) | ADAMS 2002, ages 71+ (N=856). Comprehensive, independent in-home assessment, including detailed functional and medical histories, current medications, neuropsychiatric symptoms, impairment measures, examination, and DNA samples. Had 56% participation rate among non-deceased HRS respondents selected for participation. | Prevalence for all dementia/ AD/vascular (%):
| Regression analyses present odds ratios by race (African American or White), sex, and education, indicating higher rates for African Americans, less-educated people, and women. |
| Prince et al. (2015) | Meta-analysis based on 10 studies for men and women combined and 6 studies with separate estimates for men and women. Poisson random effects models. Because the authors use more studies for computing overall dementia prevalence than for computing gender-specific prevalence rates, the totals for all people are not consistent with the weighted average of the men's and women's rates. The totals for all are lower. | Prevalence for dementia:
| None reported. |
| Rocca et al. (2011) | Three community-based studies (Chicago Aging Study 1997-2008; African Americans in Indianapolis 1992 and 2001; Rochester, MN from 1975-1994) and a national study (HRS 1993 and 2002, ages 70+). | Varies by study; the HRS study suggests prevalence decline from 12.2% in 1993 to 8.7% in 2002. | Education and net worth is associated with lower prevalence of CI in HRS estimates. |
| Stallard and Yashin (2016) | Compares NLTCS 1984 and 2004 (N=49,258 for cumulative sample over 6 waves; N ranges from 16,000 to 21,000 in annual cross-sections, with 6,000-7,500 in more detailed interviews). Focuses on SCI that meets HIPAA criteria,2 disaggregates community and institutional populations. | HIPAA prevalence in 2004 (table 2.16): 65-69: 1.22%; 70-74: 2.26%; 75-79: 4.93%; 80-84: 9.07%; 85-89: 17.70%; 90-94: 26.69%; 95+: 44.67%. Documents large decline from 1984-2004. Tests sensitivity to test score. | Age-adjusted SCI rates were lower for men than women and men's rates fell faster (tables 2.17 and 2.18). |
| Suthers, Kim, and Crimmins (2003) | AHEAD (1994-2000, 7,143 in community plus 387 institutionalized respondents, with at least 2 interviews). Adjustments made for CI in institutions. Uses TICS cognition score of 8 or less to classify as cognitively impaired. | Prevalence ages 70+: 9.5%. | Women: 10.5%, men: 7.9%. Age-specific rates are higher for women at older ages and higher for men at younger ages. |
| U.S. GAO (1998) | Meta-analysis based on 18 prevalence studies (mostly outside the U.S.). | In the U.S., at least 1.9 million ages 65+ impaired in 1995, closer to 2.1 million after accounting for missing data, with a range from 700,000 to 3.2 million; projects at least 2.9 million in 2015. SCI (and total CI) prevalence rates by age (%): 65-69: 0.6 (1.1); 70-74: 1.3 (2.2); 75-79: 2.5 (4.6); 80-84: 5.1 (9.2); 85-89: 10.0 (17.8); 90-94: 18.7 (31.5); 95+: 34.9 (52.5). | Gender differentials reported. |
NOTE:
| |||
| TABLE 2. Selected Studies of the Cross-Sectional Incidence of CI: US and Other Wealthy Countries | |||
|---|---|---|---|
| Study | Data, Sample Definitions, Timing, Ages | Age-Specific Incidence and Gender Differences | Other Group-Level Differences |
| Brookmeyer, Gray, and Kawas (1998) | Combine information from multiple early epidemiological studies (Baltimore, Boston, Framingham and Rochester) published 1984-1993 to determine incidence rates. Shows bounds. Characterizes outcomes as AD. | Age 60: 0.08% annually; age 65: 0.17%; age 70: 0.35%; age 75: 0.71%; age 80: 1.44%; age 85: 2.92%; age 90: 5.95%; age 95: 12.1%. | |
| Corrada et al. (2010) | Ages 90 and older (N=330) in a California retirement community, non-demented at baseline in January 2003, followed until December 2007. Mostly well-educated and upper class. Evaluate using full in-person examination where possible. Included MMSE. | Overall incidence assuming dementia-free at age 90:18.2%; ages 90-94: 12.7%; 95-99: 21.2%; 100+: 40.7%. | Gender differences are minimal; lower risk with graduate school education for women but not men. |
| Fishman (2017) | ADAMS (856 HRS baseline participants with 308 dementia cases from sample of 1,770, with 227 deaths and 687 refusals), 2001-2009; 456 followed. Comprehensive in-home assessment, including functional and medical histories, current medications, examination, neuropsychiatric symptoms, impairment measures, and DNA samples. Participating clinicians were not aware of cognitive test scores. Estimates dementia onset rates for those dementia-free at age 70 in 1920 birth cohort; projects for 1940 cohort. Uses multiple decrement life table techniques. | Smoothed single-year-of-age incidence rates among those dementia-free at age 70: age 70: 1.0%; 75: 1.6%; 80: 2.4%; 85: 3.7%; 90: 5.6%; 95: 8.6%; 100: 13.0%. | |
| Freedman et al. (2018) | NHATS 2011 and 2015 (Medicare population), ages 70+. N=27,547 person-year observations; yearly sample sizes range from 3,748 to 6,454 (includes refresher cohorts). Focuses on probable dementia. See full description in Table 1. | Incidence rate for 2014 among those 70+ without dementia is 3.4%; also 3.4% for women. | Non-Hispanic Black: 3.7%; non-Hispanic White: 2.5%; no cardiovascular risk: 1.5%. |
| Gilsanz et al. (2019) | California health plan participants (Kaiser Permanente of Northern California) ages 90+ with no prior dementia diagnosis on January 1, 2010 (N=2,350). Follows through September 2015. Uses diagnosis information from inpatient and outpatient medical records to identify dementia.1 Age adjusts when computing rates. | Overall annual age-adjusted incidence rate at ages 90+: 10.5%. Suggests that racial differentials persist even at the oldest ages. | Ages 90+ incidence: 12.15% for African Americans; 10.58% for Latinos; 9.69% for Whites; 8.99% for Asians. |
| Katz et al. (2012) | Einstein Aging Study, Bronx County, New York, ages 70+, non-institutionalized, ambulatory, English speaking, without visual or auditory impairments that prevent testing; 1993 with up to 16 years of follow-up (to 2009), with an average of 3.9 follow-up years. N=1,944. (Table 2 reports 130 cases of impairment.) Uses CMS enrollment data (for sample drawn through 2006) and voter rolls (after 2006) to select the sample of people who are screened for enrollment. Measures functional status and uses neurological exam, neuropsychological testing, and physical measures; diagnosis assigned based on case conference. Differentiates between dementia, AD and MCI. | Incidence rate of dementia/AD by age (%):
|
|
| Plassman et al. (2011) | ADAMS (N=1,770). Respondents are ages 70+ who completed the 2000 or 2002 wave of HRS. | Incidence rate for dementia/AD/CIND/dementia among those with CIND (%): 72-79: 1.89/1.66/3.93/ 10.23; 80-89: 4.22/2.42/7.31/12.38; 90 plus: 8.21/6.40/20.64/12.46; all 72+: 3.33/2.29/60.4/12.03. | Shows odds ratios for years of education, sex, race, presence of APOE allele. Estimated age of onset for dementia was 83.72. |
| Prince et al. (2015) | Meta-analysis based on 8 studies. Uses Poisson random effects models. | Incidence (%): Ages 60-64: 0.38; 65-69: 0.68; 70-74: 1.06; 75-79: 1.87; 80-84: 3.28; 85-89: 5.57; 90+: 11.2. | |
| Satizabal et al. (2016) | Framingham Heart Study, ages 60 and older in 4 periods (1977-83; 1986-91; 1992-98; 2004-08). N=5,205 (40,192 person-years). Reports overall dementia, AD, and vascular dementia using 5-year hazards; participants screened using MMSE; those who report cognitive decline and those with below-cutoff scores, which are adjusted for education and prior screening results, can receive extra screening; study reviews medical records and receives physician referrals which can also trigger screening; a dementia panel reviews every case. | Overall (age 60+) incidence over 5-year period:
| Shows differentials by age, gender, education, genomic risk factor, blood pressure, BMI, diabetes, and stroke, with age-sex adjustment. Focuses on differentials in trends rather than in levels. |
| Tom et al. (2015) | Adult Changes in Thought: members of Group Health (Seattle, WA) ages 65+ without dementia at enrollment, 1994-2008 (N=3,605). Includes refresher cohorts. Uses Cognitive Abilities Screening Instrument and cutoff score of 86 for neurological, neuropsychological, and physical examination and testing, including imaging. | Incidence (%):
| |
| Zissimopoulos et al. (2018) | HRS 2000 to 2012. Ages 65+. N=49,416 person-years. | Incidence (reported in table 3, online appendix, not in the article itself: Age 65-71: 0.53%; age 72-79: 1.23%; ages 80-89: 2.87%; age 90+: 6.20%. | |
NOTES:
| |||
| TABLE 3. Selected Studies of the Long-Range Likelihood of CI, Remaining Life Expectancy with CI, Duration of CI, and Other Longitudinal Metrics | |||
|---|---|---|---|
| Study | Data, Sample Definitions, Timing, Ages | Percent Ever Impaired | Duration and Other Longitudinal Metrics |
| Brookmeyer et al. (2002) | Baltimore Longitudinal Study of Aging, adults ages 55+, follow up from January 1985 to September 1999; N as of September 1999: 2,476. Uses a subset of 951 people who ever had a follow-up after age 55. Conduct neuropsychological tests and neurological exams after age 65. Between ages 55 and 64, uses Blessed Telephone-Information-Memory-Concentration Test. Those with low scores receive home visits. | Not reported. |
|
| Chêne et al. (2015) | Framingham Heart Study, focus on 1975-2009, original and offspring cohorts. Distinguishes AD and dementia. Total of 136,266 person-years. Some receive Kaplan-Albert neuropsychological test battery. Also uses MMSE. Extra screening by neurologist or neuropsychologist is conducted for those flagged as potential dementia. | Competing lifetime mortality-adjusted incidence from age 45 (unimpaired at 45): dementia/AD risk is 22.7/19.5% women; 13.8/10.3% men; unimpaired at age 65: women's dementia/AD risk is 24.6/21.1%; men's: 15.5/11.1%. | Does not report duration. Reports cumulative competing mortality-adjusted incidence at 10-year intervals, by sex for dementia/AD conditional on survival to age 45 (%):
|
| Crimmins et al. (2018) | HRS, ages 65+, 2000 (N=10,374) and 2010 (N=9,995); classifies into three groups: good cognition, dementia, and CIND based on cognitive tests and additional indicators for a subset of the sample (cognitive score, dementia diagnosis, neuropsychological assessment). Cutoffs for cognitive score are 0-6=dementia, 7-11=CIND, and 12-high=good; includes both community and nursing home respondents and proxy interviews (where cognitive status depends on proxy's assessment, including evaluation of IADL limitations). Also reports estimates from age 85. | Not reported. |
|
| Dufouil et al. (2018) | Framingham Heart Study, focus on 4 periods from 1977-1985 through 2004-2008. N=5,205 participants ages 60+ and dementia-free at baseline; 317 have dementia and are matched to controls. Focus on 5-year risk. | Reports Ns but not cohort rates. | Reveals increasing average age at first dementia--from 80.00 to 86.38--and age at death for those with dementia--from 85.92 to 89.40--across periods. |
| Farina et al. (2019) | HRS 2000-2014 (N=16,113 for Whites and 2,822 for Blacks). Uses TICS, with cutoff of 6 for dementia; includes proxy reports. | Not reported. |
|
| Fishman (2017) | ADAMS (856 HRS baseline participants with 308 dementia cases from sample of 1,770, with 227 deaths and 687 refusals), 2001-2009; see table 2. Estimates for those dementia-free at age 70 in 1920 birth cohort; projects for 1940 cohort. Uses multiple decrement life table techniques. | Among survivors to age 70 dementia-free: 1920 cohort: 26.9% for men, 34.7% for women; 1940 cohort: 31% for men, 37% for women. | Expected years with dementia after age 70 for the entire population dementia-free at age 70 (including those who never develop dementia): men 0.76; women: 1.74. |
| Ganguli et al. (205) | Studies adults ages 65+ from voter registration roles in southwestern Pennsylvania communities: 1,681 participants in Monongahela Valley Independent Elders Study, 1987-2002. Used MMSE and other indicators. | Not reported. | Expected years with AD for those with AD, adjusting for censoring by age, by onset age:
|
| Garcia et al. (2017) | HRS 1998-2012 (N=32,406 people, 146,593 person-years), plus public use National Health Interview Survey Linked Mortality file 1997-2009 for Hispanic mortality rates. Uses Sullivan life tables. Uses TICS score of 6 or less for dementia, 7-11 for CIND, and 12 or higher for no CI. Uses proxy respondents' reports of memory, IADL limitations. Censors those in institutions, so life tables represent civilian non-institutional population. | Not reported. | At age 50, entire non-institutional population (including those who never develop CI, expected years with dementia/expected years with CIND/% with any CI: White women: 1.6/4.1/17%; Black women: 3.9/8.4/ 40%; native-born Hispanic women: 4.7/9.4/42%; foreign-born Hispanic women: 6.0/12.0/49%; White men: 1.1/4.0/17%; Black men: 3.1/7.6/41%; native-born Hispanic men: 3.0/7.8/ 36%; foreign-born Hispanic men: 3.2/9.6/41%. |
| Gilsanz et al. (2019) | California health plan participants (Kaiser Permanente of Northern California) ages 90+ with no prior dementia diagnosis on January 1, 2010 (N=2,350). See Table 2. | Among survivors to age 90 dementia-free, cumulative rates are 22.6% (5 years) and 36.7% (10 years). Rate for 10-years is 10% higher for African Americans than Asians (44.6% and 34.2%). | |
| Helzner et al. (2008) | Washington Heights Inwood Columbia Aging project (N=323 incident AD cases with follow-up data), ages 65+. Participants followed up to 12 years. Initial 1992 sample based on Medicare enrollees from 14 Manhattan census tracts (N=2,125) plus 1999 refresher sample (N=2,183) from all of Manhattan. | Postdiagnosis survival duration (median): non-Hispanic Whites: 3.7 years; African Americans: 4.8 years; Hispanics: 7.6 years. Racial and ethnic differences not significant in multivariate model. | |
| James et al. (2014) | 2,566 adults ages 65+ (mean 78.1 years) without dementia at baseline from the Religious Orders Study and the Rush Memory and Aging Project. (Table 8 description of Bennett et al. provides more information.) | Not reported. | Median time from AD diagnosis to death was 3.8 years. |
| Johnson, Brookmeyer, and Ziegler-Graham (2007) | Baltimore Longitudinal Study of Aging (N=108 cases with AD). See Brookmeyer et al. (2002) above for detail. | Not reported. | Survival time with AD depends on onset age and functional form. At age 65, median survival ranges 7.8-10.2 years for men and 8.9-13.9 for women. At age 75, median ranges 5.1-6.5 for men and 6.3-8.0 for women. At age 85, median ranges 2.1-3.8 for men and 3.1-4.8 for women. |
| Larson et al. (2004) | 23,000 adults ages 65+ enrolled between 1987 and 1996 in the Group Health Cooperative, Seattle Washington; N with AD=521. Uses MMSE. | Not reported. | Expected years with AD, for those who develop AD: for men, median 4.2 (p25=2.3; p75=6.3), for women, median 5.7 (p25=3.1; p75=9.0). |
| Lichtenstein et al. (2018) | Canadians in British Columbia enrolled in a dementia clinic from 1997-1999 and followed (N=168). | N/A (sample is people diagnosed). | For those who develop dementia. Median survival is 7.08 years. Women: 7.42, men: 5.83. Age at assessment 80: 7.25; age >=80: 4.58. AD: 7.33, other dementias: 4.33. Education: 12: 7.33; >=12: 5.83. Not all differences significant. |
| Lièvre, Alley, and Crimmins (2008) | AHEAD (1994-2000, at least 2 interviews, N=7,228 people), living in the community, ages 70+; non-institutional restriction suggests lower bound estimate given high rates of impairment in the institutional population (see Suthers et al. 2003 below, which corrects for this). Finds significant shares recover from CI and tests robustness of finding. Uses TICS cutoff score =8 for CI in first wave and =9 in subsequent waves. | Not reported. | Expected years with CI for the entire population, including those who never develop CI:
|
| Mayeda et al. (2016) | Payment records from Kaiser Permanente Northern California for 2000-2013. (N=274,283 heath care members; total number of dementia cases is 59,555.) Sample excludes those impaired as of baseline (ages 65 plus); definition: ICD-9 diagnostic codes.1 | Cumulative 25-year risk at age 65: 35% American Indian/Alaska Native; 38% African American; 32% Latino; 25% Pacific Islander; 30% White; 28% Asian American. | |
| Mayeda et al. (2017) | 273,843 dementia-free plan participants (Kaiser Permanente of Northern California) ages 64+ followed from January 1, 2000 to December 31, 2013. 59,494 developed dementia, and 64.1% of those developing dementia died. Excludes cases missing race, multi-race, and Pacific Islanders. | Not reported. | Reports estimates by race/ethnicity (no estimates for all people in the sample). Finds shorter median time from dementia diagnosis to death for non-Hispanic Whites (3.1 years) than for Native Americans/Alaska natives (3.4 years), African Americans (3.7 years), Latinos (4.1 years), and Asian Americans (4.4 years). Differences persist after adjusting for co-morbidities. Also reports results for the cross of race-Hispanicity and age at diagnosis. |
| Mueller et al. (2019) | Meta-analysis of 11 longitudinal studies (N=22,952, 2,029 with DLB and 20,923 with AD). | Not reported. | Expected years with dementia for those who develop it. At diagnosis (average diagnosis age DLB: 76.3/AD: 77.2): 4.11 with DLB, 5.66 with AD. Differences are robust to controls. |
| Murtaugh, Spillman, and Wang (2011) | 1993 National Mortality Followback Survey (N=22,957 death records) calibrated using data from NLTCS to account for morbidity declines and 2000 Social Security Administration intermediate data to account for population longevity change. | At age 65: 19.8% (all); 14.8% (men); 24.4% (women). | Expected years with CI those who develop it. At age 65: 4.4 (all), 4.6 (women), 3.9 (men). |
| Reuser, Willekens, and Bonneux (2011) | HRS/AHEAD 1992-2004; participants in at least 2 waves. Uses TICS with cutoff point of 8 to indicate SCI plus proxy responses for poor memory. Total people/person-years: 9,834/78,180 men and 12,554/98,674 women. Uses 3 education groups: Not HS graduate, HS graduate or some college, and college degree or more. | At age 55 cumulative probability (men/women): non-Hispanic Whites: 22.6/35.9%; non-Hispanic Blacks: 43.9/53.3%; Hispanics: 46.2/60.8%; education (non-Hispanic Whites only): low: 29.6/39.2%; medium: 17.6/35.7%; high: 19.8/29.3%. | Life expectancy at age 55 by cognition status for the entire population (total years/with CI/no CI):
|
| Robitaille et al. (2018) | Compares 6 longitudinal aging studies. We focus on the 1 U.S. study: Memory and Aging project for a Northern Illinois retirement community (N=1,852, final sample 1,442. see Bennet et al. 2013 in Table 10 below). Begun in 1997 with on-going enrollment--used total of 19 waves. Uses MMSE and scores as follows: 27-30 indicates normal cognition; 23-26 indicates mild impairment; 22 or lower indicates moderate to severe impairment. Reports total and unimpaired life expectancies; we compute impaired as the residual. | Not reported. | Life expectancy by cognition status for the entire population:
|
| Seshadri et al. (2006) | Framingham Heart Study. Original cohort participants (N=4,897, 115,146 person-years) who were stroke and dementia-free at 55 years of age were followed biennially for up to 51 years. | At age 65 conditioned on dementia-free: 20% for women and 10% for men. | |
| Stallard (2011) | NLTCS, 1984-1994; HIPAA SCI focus; uses short portable mental status questionnaire. Cutoff value is 5 or more for SCI; 3 or 4 for MCI. Also uses proxy reports, Medicare records. | Not reported. | Expected years from age 65 with HIPAA-level CI for the entire population including those who do not develop CI: 1.20 unisex, 0.78 men, 1.53 women. |
| Stallard and Yashin (2016) | Compares NLTCS 1984 and 2004 (cumulative sample over 6 waves is 49,258, annual cross-sections of 16,000-21,000 with 6,000-7,500 more detailed interviews); HIPAA-level SCI focus, disaggregates community and institutional populations. | Not reported. | Expected years from age 65 with HIPAA-level CI for the entire population including those who do not develop CI, 2004 estimates: 1.20 unisex, 0.79 men, 1.55 women. |
| Suthers, Kim, and Crimmins (2003) | AHEAD (1994-2000, 7,143 in community plus 387 institutionalized respondents, at least 2 interviews), adjustments made for CI in institutions. | Not reported. | Expected years after age 70 with CI for the entire population including those who do not develop CI: 1.5 years unisex (all, after adjusting for those in institutions). Men: 1.1 years; women 1.7 years. Also expresses as percent of life and reports estimates at older ages (75, 80, 85, 90). |
| Tom et al. (2015) | Adult Changes in Thought: members Group Health (Seattle, WA) ages 65+ without dementia at enrollment, 1994-2008 (N=3,605). | Not reported. |
|
| Yang and Levey (2015) | MCBS Cost and Use files, 2000-2010 (113,811 person-years on 53,244 people); Medicare beneficiaries ages 65+; defined as one of the following: (1) response of respondent diagnosis of AD or dementia; (2) ICD-9 codes of inpatient and outpatient claims indicating dementia (ICD-9-CM 290 or 331.0); (3) outpatient prescription drugs claims showing use of drugs prescribed for dementia.2 | At age 65 (1920s and 1930s cohorts): 14.2% (all); 15.5% (women); 13.1% (men). | Expected years with dementia after age 65 for those who develop dementia: 5.3 for women and 4.7 for men. Average age of onset approximately 80. |
| Yang et al. (2012) | MCBS, 1997-2005. N=113,811 person-years from 53,244 people. Classified as having dementia if any of 3 indicators are true: (1) respondent reports that physician diagnosed AD/dementia; (2) diagnosis codes in a claim; (3) drug claims for AD.3 Also classified as having dementia in subsequent year if classified in earlier year. Simulates for virtual birth cohort born in the 1920s and 1930s. | Not reported. | Expected years with dementia for those who develop dementia: 5.1. Early-onset (=80): 6.4 years. Late onset (>80): 3.8 years. Average age of onset 80.2. |
| Zissimopoulos et al. (2018) | FEM using HRS 2000-2012; requires 2 waves of diagnosis. | 34.7% from age 51, conditioned on survival to age 65. | Expected years with diagnosis for the entire population, including those who are never diagnosed: 2.94 at age 51, conditioned on survival to age 65. Expected years with diagnosis for those diagnosed: 5.19 at age 51, conditioned on survival to age 65. |
NOTES:
| |||
| TABLE 4. Selected Studies of the Relationship between CI and Mortality Rates in the US | ||
|---|---|---|
| Study | Data, Sample Definitions, Timing, Ages | Results, Including Relevant Differentials |
| Davis et al. (2018) | Longitudinal data from National Alzheimer's Coordinating Center (N=18,103), patients over age 65 with multiple visits. Defines severity using etiological information plus clinical dementia rating score, with cutoffs of 2 for MCI, 2 for moderate AD dementia, and 3 for severe AD. | Reports single year mortality probabilities based on disease severity. At age 65/75, 1-year death probability is 1%/1% if normal cognition; 1%/2% if MCI, 4%/6% if mild AD, 9%/12% if moderate AD, 16%/20% if severe AD, and 7%/10% if non-AD CI. |
| Dufouil et al. (2018) | Framingham Heart Study, ages 60 and older in 4 periods (1977-83; 1986-91; 1992-98; 2004-08). N=5,205; focuses on 314 dementia cases and 314 matched controls (matched on age, epoch, gender, stratified by age, education, sex). Participants screened using MMSE; those with below-cutoff scores (education adjusted; considers prior screening results) can receive extra screening, as can those who report or whose family members report cognitive decline; study reviews medical records and receives physician referrals which can also trigger screening; dementia panel reviews every case. |
|
| James et al. (2014) | 2,566 persons aged 65 years and older (mean 78.1 years) without dementia at baseline from the Religious Orders Study and the Rush Memory and Aging Project. (Table 10 description of Bennett et al. (2013) provides more information.) | The mortality hazard ratio for AD dementia was 4.30 at ages 75-84 and 2.77 at ages 85+. Estimates that about one-half million deaths at ages 75+ in 2010 were attributable to dementia. |
| Langa et al. (2008) | HRS 1993 (N=7,406) and 2002 (N=7,104), ages 70 and older and living in the community (excludes nursing home residents). Uses TICS of 11 or more to denote normal function; 8-10 is MCI and 0-7 is SCI. | Death risk for those with SCI increased over time. Education was associated with higher SCI-conditional death rates. |
| Wilson et al. (2009) | 1,715 persons deemed dementia-free in a prior data collection wave from Chicago Health and Aging Project, a longitudinal study of aging and AD conducted in 3 adjacent Chicago neighborhoods (Hebert et al. [2010] and Weuve et al. [2018] described in Table 10 provide additional information.) 802 had no CI, 597 had MCI, 296 had AD, 20 had other forms of dementia. Base sample is people ages 65 and older. | During up to 10 years of observation (mean=4.7, SD=3.0), 634 individuals died (37%). Compared with people without CI, risk of death increased by about 50% for those with MCI (hazard ratio of 1.48) and was nearly 3 times greater for those with AD (hazard ratio of 2.84). Effects did not differ by race (African Americans and Whites). |
| TABLE 5. Selected Studies of the Costs of CI in the US | |||
|---|---|---|---|
| Study | Data, Sample Definitions, Timing, Ages, Cost Components Included | Results | Differentials or Other Results |
| Coe et al. (2018) | People ages 65 and older. HRS 1992-2012 matched to Medicaid and Medicare (Parts A, B, and D from 1999-2009) payment data for people in FFS plans. Match rate to the claims data was about 80%. Defines dementia using diagnostic codes.1 Uses control group of HRS respondents with same sex and birth year to determine the incremental costs of ADRD. | Over the first 5 years after diagnosis, costs per dementia case attributable to ADRD are $15,339 to traditional Medicare and $15,852 to Medicaid (2017$). Two-thirds of Medicare costs occur in first 2 years after diagnosis and are driven by inpatient use (both hospital and skilled nursing facility). Long-term care (home health and nursing home costs) drives Medicaid costs. Unlike Medicare, Medicaid's incremental costs are concentrated among longer-lived cohort members who are more likely to need LTSS and be Medicaid-eligible. | Focus on duration, including front and backloading of costs. Conditional on being alive and enrolled, Medicaid costs average $20,479 in months 1-12, increasing to $77,011 in months 49-60. |
| Hurd et al. (2013) | HRS 2000-2008; N=7,758 for AHEAD, 4,210 for CODA, 10,413 for HRS, 3,488 for war babies, 3,624 for EBB; linked to Medicare FFS records (about 70% of sample), average annual costs; self-reported out-of-pocket costs plus institutionalization; uses models based on ADAMS, TICS, and proxy reports to assign cognitive status; state-specific home health rates from Metlife (2010). Presents estimates adjusted for demographic characteristics and coexisting conditions (stroke, diabetes, heart disease, hypertension, lung disease, cancer, psychiatric problems, arthritis) and unadjusted estimates. | Conditional on dementia, estimated unadjusted/adjusted annual market costs per person age 18+ (2010$) $33,329/$28,501; out-of-pocket: $6,838/$6,194; Medicare: $5,226/ $2,752; net formal home care: $6,888/$5,678; nursing home care: $14,337/$13,876; total costs: $47,920/$41,689 if unpaid care is valued at foregone wages ($14,591/ $13,188) or $64,168/$56,290 if unpaid care valued at replacement cost ($30,989/$27,789). | Gender, age, education, race/ethnicity, marital status, household income; ADL/ IADL limits; health conditions. |
| Jutkowitz, Kane, et al. (2017) | For disease progression: Uniform Data Set of National Alzheimer's Coordinating Center; contains data from 34 AD Centers (see Jutkowitz, MacLehose, et al. 2017--N=457). Separately considers cognition, function (up to 10 limits), and behavioral psychological state (up to 12 symptoms). Measures cognition using MMSE. Annual assessments. Also uses HRS linked to published Medicare data for caregiving and costs, ADAMS, life tables, Metlife care costs. Models transitions in living arrangements. Discount rate is 3%; appears to discount to onset age. | From diagnosis (base case mean age of 83 years, survival 5 years) discounted total lifetime cost of care for a person with dementia was $321,780 (2015$). Families incurred 70% ($225,140-$135,300 in unpaid care and 89,800 out-of-pocket), Medicaid 14% ($44,090), and Medicare 16% ($52,540) of the total burden. Lifetime costs for a person with dementia were $184,500 greater (86% incurred by families) than for those without SCI. Total annual cost peaked at $89,000 and net cost peaked at $72,400. Costs increase for the first 5 years after onset, then slowly decline. | Contrasts by onset age. Considers best and worst-case scenarios, integrating differing assumptions about costs of formal and informal care. |
| Kelley et al. (2015) | HRS, Medicare fee-for-service beneficiaries who died at ages 70+ in 2005-2010. N=1,702. Examines 4 groups by cause of death, including 1 with high dementia probability using 50% threshold in Hurd et al.'s (2013) probabilistic classification. Focuses on 5-year (rather than annual) costs. Shows value of out-of-pocket expenditures; expresses as a percent of wealth 5 years before death. Uses present value to 5 years before death, 3% discount rate. Values informal care using state-specific values. Uses Metlife data for nursing home prices. | 2010 dollars. Average total cost per decedent with dementia ($287,038) was significantly greater than that of those who died of heart disease ($175,136), cancer ($173,383), or other causes ($197,286). Medicare expenditures were similar across groups, but dementia patients' mean out-of-pocket spending ($61,522) was 81% higher than for those without ($34,068); pattern for informal care is similar. Dementia group's out-of-pocket spending (median, $36,919) represented 32% of wealth measured 5 years before death compared with 11% for non-dementia group. | Race (Black/non-Black), education (not HS graduate, HS diploma or more), and marital status-sex groups (married and unmarried men and women). |
| Langa et al. (2001) | AHEAD, 1993, ages 70+, N=7,443. Defines dementia using TICS (cutoff of 8). Uses quantiles of wages for home health aides to derive informal caregivers' opportunity cost. (Bases intermediate cost on mean, low cost on 10th percentile and high cost on 90th percentile.) Estimates account for other conditions, so costs are net of other factors, not a person's total care costs. | Additional incremental yearly (net of) cost of unpaid care (appears to be 1998$) for those with dementia varies with severity: $3,630 for mild dementia; $7,420 for those with moderate dementia; and $17,700 for those with severe dementia. This yields total annual U.S. costs of $18.6 billion (high-cost: $24.5 billion; low cost: $13.4 billion). | |
| Lin et al. (2016) | Five percent sample of Medicare claims files, 2009-2013. Individuals newly diagnosed with ADRD (N=25,916) or MCI (N=2,784) are each individually matched to a control subject without ADRD or MCI. | Medicare expenditures were 42% higher in people with ADRD ($10,622 vs $15,091) and 41% higher in those with MCI ($9,728 vs $13,691) during the year before diagnosis than for controls. Medicare expenditures of those with ADRD increased to $27,126 for the year immediately after diagnosis and decreased to $17,257 during the subsequent year. For those with MCI, mean Medicare expenditures were $20,386 for the year after diagnosis and $14,286 for the subsequent year. Use of inpatient care, post-acute skilled nursing facility care, and home health care increased substantially after ADRD or MCI diagnosis. | |
| Pyenson et al. (2019) | Medicare 5% limited dataset claims file from 2006 to 2015. Sample is Medicare FFS beneficiaries age 69+ who died in 2010-2015 and had at least 3 full years of Parts A and B coverage before their year of death (N=384,008). Uses diagnoses for the 3 years prior to death plus year of death. Excludes those with Parkinson's disease or other specified diseases. | Added costs to Medicare of AD are modest (about 10-11%). In year of death, about 30% of people classified as having AD are dually eligible for Medicare and Medicaid, compared with 25% for those classified as having other dementias, and 15% for those with no dementia. | |
| Stallard (2011) | NLTCS, 1984-1994; HIPAA severe SCI focus; columns 4 and 5 of Table 6 can be combined to arrive at total dollar costs incurred while cognitively impaired. | Unconditional combined nursing home and home care costs for those with HIPAA-level CI during periods with SCI of $32,351 (in 2000$) at 65+; $39,259 at 75+; $50,992 at 85+; $54,203 at 95+. | |
| White et al. (2019) | HRS data matched to Medicare FFS claims from 1991-2012. N=4010. Those with dementia-related claims are matched to controls without dementia. | Five-year total difference in Medicare spending between those with/without dementia is about $15,700; half is incurred in the first year. Differences in costs between those with and without dementia decline after diagnosis; about 5 years after diagnosis differences are minimal. Shorter survival with dementia explains part of the difference (the estimates do not condition on survival). | Survival with dementia varies by sex. |
| Zhu et al. (2008) | Predictors study, which prospectively follows patients from 3 university-based health centers (Columbia, Johns Hopkins, Mass General) with probable AD for 7 years, 1998-2004 (N=204). Sample is not representative--well-educated, disproportionately non-Hispanic White. Focuses on home care use, including both paid home health and informal care from caregivers, as duration with dementia, and often disease severity, increases. Used modified MMSE for dementia and other tests for indications of co-morbidities (e.g., Parkinson's, psychopathology, others like heart disease). | 2004 dollars. Home care users are older, less likely to be married, have worse functional status, are more likely to have extrapyramidal symptoms than non-users. Percentage using home health increases steadily over time, from 9.9% at baseline to 34.5% in year 4; days in last 3 months among users increased from 21.9 to 56 over this period. Among formal users, 87% was paid out-of-pocket, with small amounts by Medicare, other insurance. Out-of-pocket liability increased from $20,590 at baseline to $43,031 in year 4. Informal care similarly increased over time. Among users with only informal, increase was from 24.4 hours/week to 39.4 hours/week. Similar increases were seen among those with both formal and informal care. Among all, costs increased from $12,033 to $21,148. Increases in formal were faster than increases in informal, leading to a higher ratio of formal to informal. | |
| Zhu et al. (2015) | Washington Heights-Inwood Columbia Aging Project, 1999-2010. Looks 2 years before and 2 years after dementia onset, compares to non-demented control group. Focus on Medicare spending. 3,756 participants were followed, 2,476 were matched to Medicare Beneficiary Summary file. | During the 2 years before incident dementia, rates of inpatient admissions and outpatient visits were similar for dementia patients and non-demented controls, but home health and skilled nursing care use and durable medical equipment were higher in dementia patients. With incident dementia, there is a small but significant associated excess increase in inpatient admissions but not other care areas. In the 2 years before dementia, total Medicare expenditures were higher in dementia patients than in controls. But finds no excess increases in Medicare spending after dementia onset. | |
| Zissimopoulos, Crimmins, and St. Clair (2014) | FEM simulation model, using data from HRS, ADAMS, and the MCBS. Uses 0-6 as TICS cutoff for dementia and 7-11 for mild impairment no dementia; proxy cutoffs lower. Values unpaid care using wage for aide hired from a home health agency from MetLife study. | At ages 70+ in 2010, per capita annual costs of $71,303 (2010$) for people with AD ($42,074 in paid costs, $29,229 in unpaid costs), compared to $18,374 for people without ($15,408 paid costs, $2,965 unpaid costs); includes projections for 2030, 2050 and detail on payers (Medicare, Medicaid, out-of-pocket). | Projects 9.1 million in 2050, an increase from 3.6 million in 2010 and 5.8 million in 2030. |
NOTES:
| |||
| TABLE 6. Selected Studies of the Prevalence and Intensity of Caregiving for People with CI in the US | ||
|---|---|---|
| Study | Data, Sample Definitions, Timing, Ages, Components Included | Results |
| Friedman et al. (2015) | HRS, 2010, community-dwellers ages 70+ (N=8,106). Uses dementia classification as in Hurd et al. (2013), see Table 5 above. Care refers to ADL or IADL help due to functional impairment. |
|
| Kasper et al. (2015) | NHATS/NSOC, 2011 (N=2,423 adults ages 65+ who received help with self-care, mobility, or household activities; family and unpaid caregivers N: 1,924). | Average monthly care hours: 91.7 hours with dementia, 67.5 hours without. |
| Ory et al. (1999) | National Caregiver Survey, 1996 (N=1,509). Caregivers ages 18+ who provided care in last 12 months to someone ages 50+. Dementia defined based on caregiver report that care recipient has AD, confusion, dementia, or forgetfulness. | Average years provided care: dementia caregivers: 5.10 years; non-dementia caregivers: 5.07 years. Average weekly hours of care: dementia caregivers: 17.06; non-dementia caregivers: 12.45. Percent providing 40 or more hours of care/week (includes "constant"): dementia caregivers: 28.1%; non-dementia caregivers: 17.0%. Percent using: dementia caregivers: 47.7%; non-dementia caregivers: 32.6%. |
| Spillman et al. (2014) | NHATS, 2011 (Medicare beneficiaries ages 65+). NSOC sample was drawn from 4,935 eligible informal caregivers to 2,423 eligible NHATS respondents. Response rates of 60-68%. Final sample is 2,007 caregivers for 1,369 respondents; analyses include only those who helped in last month (N=1,996). | Percent of caregivers to recipients with probable dementia: 32.5%; possible dementia: 15.3%; no dementia: 52.2%. Percent of total care hours to recipients with: probable dementia: 40.0%; possible dementia: 14.7%; no dementia: 45.3%. Average monthly hours: probable dementia: 91.96; possible dementia: 71.96; no dementia: 64.94. |
| TABLE 7. Estimates from Selected Studies of the Cost of CI in the US: Using Consistent (2020) Wage-Indexed Dollars | ||||
|---|---|---|---|---|
| Study | Component | Nominal Cost | Analysis Year | Real Cost (2020$) |
| Langa et al. (2001) | Unpaid, incremental, mean, age 70+, intermediate, annual, home care wage | Moderate: $7,420 Severe: $17,700 | 1998? | Moderate: $14,499 Severe: $34,586 |
| Hurd et al. (2013) | Unpaid, total (replacement), annual | $30,989 | 2010 | $41,937 |
| Hurd et al. (2013) | Unpaid, adjusted (replacement), annual | $27,789 | 2010 | $37,606 |
| Hurd et al. (2013) | Unpaid, total (foregone wage), annual | $14,591 | 2010 | $19,746 |
| Hurd et al. (2013) | Unpaid, adjusted (foregone wage), annual | $13,188 | 2010 | $17,847 |
| Jutkowitz, Kane, et al. (2017) | Unpaid total from diagnosis (among those receiving care) | $135,300 (discounted to apparent onset age)1 | 2015 | $158,646 |
| Jutkowitz, Kane, et al. (2017) | Unpaid net from diagnosis (among those receiving care) | $132,850 | 2015 | $155,769 |
| Jutkowitz, Kane, et al. (2017) | Medicaid from diagnosis (among those receiving care) | $44,090 | 2015 | $51,696 |
| Zissimopoulos, Crimmins, and St. Clair (2014) | Paid, gross, age 70+, annual (among those receiving care) | $42,074 | 2010 | $56,938 |
| Zissimopoulos, Crimmins, and St. Clair (2014) | Unpaid, gross, age 70+, annual (among those receiving care) | $29,229 | 2010 | $38,353 |
| Jutkowitz, Kane, et al. (2017) | Out-of-pocket total from diagnosis, gross (among those with CI) | $89,840 | 2015 | $105,339 |
| Jutkowitz, Kane, et al. (2017) | Out-of-pocket from diagnosis, net, (among those with CI) | $25,110 | 2015 | $29,442 |
| Stallard (2011) | Home care and nursing home during spells with HIPAA-level SCI, unconditional, ages 65 plus (for the entire population, including those without CI) | $32,351 | 2000 | $56,740 |
| Zhu et al. (2008) | Home care, first year of spell probable AD (among those receiving care) | $25,090 | 2004 | $39,693 |
| Zhu et al. (2008) | Home care, fourth year of spell probable AD (among those receiving care) | $43,031 | 2004 | $68,075 |
| Kelly et al. (2015) | Out-of-pocket 5-year totals, gross (among those receiving care) | $61,522 | 2010 | $77,332 |
| Coe et al. (2018) | Medicaid, net, first 5 years after diagnosis--with and without survivors? | $15,852 | 2017 | $17,766 |
| Coe et al. (2018) | Medicaid, conditional on survival and enrollment | Year 1: $20,479 Year 2: $27,547 Year 3: $36,344 Year 4: $51,449 Year 5: $77,011 | 2017 | Year 1: $22,951 Year 2: $30,872 Year 3: $40,720 Year 4: $57,660 Year 5: $86,307 |
| Coe et al. (2018) | Medicare, net, first 5 years after diagnosis--with and without survivors? | $15,339 | 2017 | $17,191 |
NOTES:
| ||||
| TABLE 8. Selected Studies Based on Long-Range Models of Cognitive Status | ||||
|---|---|---|---|---|
| Study or Model | Starting Sample, and Transition Probability Data | Indicators for CI | Model Specification (functional form, state space) for CI | Other Relevant Data and Results |
| Ahmadi-Abhari et al. (2017) | England and Wales Office for National Statistics (ONS) data and projections (5-year age-sex groups). Uses ELSA, pooled data from 2002-2013; ages 50+ (refresher cohorts maintain representativeness) for transitions (anchored to 2006 midpoint); total sample of 7,906; proxy ("consultee") interviews utilized where needed and permission was obtained. Validated age-specific dementia rates against Cognitive Function and Ageing Study-II. | ELSA administered 3 sets of tests each wave--addresses orientation to time, immediate and delayed memory, verbal fluency; additional tests addressed numeracy and literacy. Used Informant Questionnaire for Cognitive Decline for proxies. Uses relative measure of cognitive status: 1.5 standard deviations below mean for one's education group (using 3 groups: no qualification, O or A-level, or university) among 50-80 year-olds. Does not find evidence of large learning effects on tests. Defines dementia using measures of cognitive status and functional impairment or self-reported or physician diagnosis. | Integrates calendar year trends in CI with those of mortality and cardiovascular disease. Fits survival model of time to incident dementia using baseline status. Model covariates include age at entry, age squared, time in study, time squared, sex, education, and mid-life history of hypertension, diabetes, obesity. Separate process for time trend. Examined effects of lifestyle and vascular risk factors. Project people ages 35 plus using Markov model with annual transition probabilities. Predicts cardiovascular disease, life expectancy, dementia, disabled and disability-free life expectancy (figure provides overview of model structure). Mortality varies by cardiovascular status. Sensitivity analyses show bounds with less optimistic dementia decline assumptions. | Age-specific incidence and prevalence decline (20% incidence decline over 20 years that increases to 42% after accounting for net composition change, like more exercise, fewer dropouts--other composition changes did not offset), but absolute number of people with dementia is expected to increase. Longer survival among those with dementia partly drives this. |
| Brookmeyer et al. (2018) | Starts with age-gender prevalence from Census. Uses forward calculation model based on incidence rates from longitudinal epidemiological studies (Mayo Clinic's Study of Aging, tracking peoples' cognitive function in Olmstead, MN). | Differentiates preclinical from clinical, with 2 different preclinical pathways: amyloidosis (progresses to amyloidosis plus neurodegeneration and then to MCI and early clinical and clinical) or neurodegeneration, which can lead to amyloidosis or to MCI then early clinical and clinical. | Multi-state model. Nine total states--with death a pathway from each state. Later stage clinical has additive (condition on background risk) mortality risk (7.8%). Age-year specific transition probabilities between states (to allow intervention effects). | Projects baseline increase from 6.08 million in 2017 with either clinical disease (3.65 million) or MCI (2.43 million) to 15 million in 2060 (9.3 million clinical, 5.7 million mild). |
| Future Elderly Model / Future Americans Model (selected studies include Zissimopoulos et al. (2018)) | See Table 3--HRS-based model. | Differentiates normal, mild impairment, and dementia. | Dementia at ages 65+ increases from 3.6 million in 2010 to 9.1 million in 2050. | |
| Hebert et al. (2013) | Starting sample is 2010 US Census and Census age-race-sex population projections. Transition probabilities based on Chicago Health and Aging Project (10,802 people represented from 1993-2011). Focuses on those evaluated between 1997 and 2000 (2,577 evaluations on 1,913 people conducted from 1997-2010). | Only considers incident cases of AD dementia. Uses uniform, structured clinical evaluation (by nurse-clinicians, neuropsychologists plus neurologist or geriatrician review). | 432 groups defined by single year of age, sex, 2 race groups, and 3 education groups. | AD at ages 65+ increases from 4.7 million in 2010 to 13.8 million in 2050. |
| Loef and Walach (2013) | Uses estimates from meta-analyses to determine dementia differentials by BMI. Uses population estimates from United Nations Department of Economic and Social Affairs. | Varies (meta-analysis). | Uses estimates from Prince et al. (2009). | Under base assumptions, dementia increases from 4.03 million in 2010 to 6.6 million in 2030 and 10.3 million in 2050; trending obesity, increases to 7.1 million in 2030 and 11.3 million in 2050. |
| Matthews et al. (2019) | Uses prevalence rates from Medicare FFS records described above in Table 1. Uses Census age-sex-race-Hispanic origin projections. | Diagnosis codes (see Table 1). | Uses constant age-sex-specific rates that vary by race-ethnicity (70 separate groups) over time. | ADRD at ages 65+ increases from 5.0 million in 2014 to 13.9 million in 2060. |
| TABLE 9. Selected Studies of the Effects of Future Interventions on CI in the US | |||
|---|---|---|---|
| Study | Data, Sample Definitions, Timing, Ages, Cost Components Included | Interventions/Sensitivities Explored | Results, Including Relevant Differentials |
| Barnes and Yaffe (2011) | Uses relative risk estimates from meta-analyses found through searches of Cochrane Database of Systematic Reviews and PUBMED. Applies these to Alzheimer's Association population estimates on an age-specific basis where possible. Focuses on population attributable risk. Reports for both U.S. and globally. Seems to focus on 2009 prevalence. | Focus on 10-25% reductions in modifiable behaviors leading to diabetes, smoking, depression, cognitive inactivity, physical inactivity, hypertension. | Cases of AD prevented in U.S. with 10-25% reductions: Diabetes: 17,000-40,000; hypertension 40,000-100,000; obesity 36,000-91,000; depression: 67,000-172,000; physical inactivity: 90,000-230,000; smoking 50,000-130,000; cognitive inactivity (low education): 36,000-91,000. Total 184,000-492,000 cases. |
| Brookmeyer, Gray, and Kawas (1998) | Combines information from 4 epidemiological studies (Baltimore, Boston, Framingham and Rochester) to determine incidence rates. Uses multiplicative adjustments to these incidence rates to determine effects of interventions. | Delay onset by 1-2 years. | Two-year delay would lead to 2 million fewer cases in 50 years; 1-year delay would lead to 800,000 fewer cases in 50 years. |
| Brookmeyer et al. (2018) | Model described above (Table 5). Uses different proportional adjustments to each of the transition probabilities. Some adjustments may equal 1. | (1) Highly effective primary prevention; (2) moderately effective secondary prevention; (3) modestly effective secondary prevention. | First intervention would reduce 2060 prevalence (from 9.3 million) to 5.14-8.93 million, depending on assumptions; range for second intervention is 3.00-8.63 million; range for third intervention is 6.76-9.12 million. |
| Fishman (2017) | Transition probabilities based on ADAMS, described above (Table 3). Shifts incidence rates in the proportions described (i.e., assigns baseline age 70 incidence rate to age 70 + K, baseline age 71 incidence rate to age 71 + K, and so forth); alternative scenarios reduce all the age-specific rates. | Interventions for those dementia-free at age 70: (1) delay dementia onset by 1 year, is effective for 50%; (2) same as 1 but affects 90%; (3) delay dementia onset by 5 years, is effective for 50%; (4) delay dementia onset by 5 years for 90%. |
|
| Hurd, Martorell, and Langa (2015) | See Hurd et al. (2013) described in Table 5. | Assumes the SCI decline seen in Matthews et al. (2013) continues; explores recent trends in 3 co-morbidities--diabetes, hypertension, and diabetes. |
|
| Lewin Group in Alzheimer's Association (2015) | Starts with counts of people with the disease. Draws heavily from other sources, including Hebert et al. (2010) and Sloane et al. (2002). Uses 2008 MCBS for costs. Stratifies based on disease severity (mild-moderate-severe) and age (65-74, 75-84, 85+). In MCBS data, uses own or proxy report of AD diagnosis. Also uses presence of diagnostic codes. | (1) Delayed disease onset; (2) slower disease progression. | Delayed onset scenario will lead to 7.8 million in 2050 (compared to 13.5 million under baseline). |
| Sloane et al. (2002) | Starts with Census 2000 middle series, population ages 60 and older. Uses prevalence rates from meta-analysis by GAO (1998) and Evans et al. (1992). Incidence increases exponentially with age until 95 and is then held constant. Two disease stages: mild and moderate-to-severe CI. Models transition between these 2 states (average 4 years moderate). Mortality assumed to be 10-20% higher than overall for the two groups. | (1) Delayed disease onset; (2) delayed disease progression; (3) combination of 1 and 2. | Under baseline, projects 10 million people age 60+ by 2050, over 6 million mild and just under 4 million severe. Delayed onset model would imply 6.3 million total in 2050--2.1 mild and 4.2 severe. Slowed progression model yields less dramatic results. (No major differentials reported.) |
| Yang et al. (2012) | MCBS, 1997-2005. N=113,811 person-years from 53,244 people. Classified as having dementia if any of 3 indicators are true: (1) respondent reports that physician ever said person has AD/dementia; (2) diagnosis codes in a claim; (3) ever took drugs for AD.1 Also classified as having dementia in subsequent year if classified in earlier year. Simulates for virtual birth cohort born in the 1920s and 1930s. | Improved education and increased BMI for the baby boomers relative to their predecessors born in the 1920s and 1930s. Specifically considers 5% higher HS graduation rate and 10% higher obesity rate. | In the increased-education scenario, longevity increases as does dementia prevalence, but onset age also increases. On net, dementia duration declines (by 0.2 years), as do both next Medicare and, especially, Medicaid costs. In the increased-obesity scenario, longevity declines but dementia prevalence increases by 4.4%, so aggregate costs increase even if average dementia duration declines. |
| Zissimopoulos et al. (2018) | FEM simulation model, using data from HRS, ADAMS, and the MCBS (see also Table 3). | (1) 50% reduction in diabetes incidence; (2) eliminate hypertension; and (3) develop treatment that delays dementia onset. | Projected changes due to diabetes reductions were limited--no change in expected duration, and increase of 115,000 people ages 65+ with dementia. Eliminating hypertension also increased numbers living with dementia. Treatments that delay onset were projected to reduce number of people living with dementia in 2040 by 2.2 million. |
NOTES:
| |||
| TABLE 10 Miscellaneous Community-Based Studies Related to CI and Performance in the US | ||
|---|---|---|
| Study | Data, Sample Definitions, Timing, Ages Included | Results, Including Differentials |
| Bennett et al. (2013); see also Bennett et al. (2012) | Religious Orders Study (1,100 participants across US, started 1994, 550 autopsies) and the Rush Memory and Aging Project (1,550 participants from Northeastern Illinois, started 1994, 425 autopsies). Combined, more than 2,700 people, agreeing to annual evaluation (sample has above average education), including targeted imaging, and brain donation upon death. | Focuses on clinical indicators (largely aspects of the brain, like presence of plaques, tangles, and White matter) and co-morbidities. |
| Dodge et al. (2014) | Studies adults ages 65+ from voter registration roles in southwestern Pennsylvania communities: 1,644 participants in Monongahela Valley Independent Elders Study and 1,982 participants Monongahela-Youghiogheny Healthy Aging Team study, 1987-2012. Used 4 tests: 2 on trailmaking, 1 on verbal fluency for initial letters, and 1 on verbal fluency for animal names. Controls for practice effects. Focus on MCI; excluded those with SCI at baseline from study. Disaggregates by birth cohort (1882-1901, 1902-1911, 1911-1921, 1922-1931) and education (not HS graduate, HS diploma, more than HS). | Earlier cohort has steeper age declines in cognition by age. Adjusting for education, the cohort pattern remains. Suggests that researchers should integrate cohort effects into forecasting models. Educational differences in levels are large, but they do not affect rate of decline. |
| Hall et al. (2009) | African Americans ages 65+ in Indianapolis (Indianapolis-Ibadan Dementia Project) in 1992 (2,582 eligible, 2,212 enrolled, 9.6% refused, 4.7% too sick) and 2001 (4,260 eligible, 1,892 enrolled, 1,999 refused). First stage is in-home interview using the Community Screening Interview for Dementia. Second stage is full diagnostic work-up for a selected subset. | Shows no significant difference across the time periods. Sampling differences across the 2 waves may be important. |
| Hebert et al. (2010) | Cumulative total of over 10,000 people ages 65+ in Chicago Health and Aging Project, 1993-1996 and 2010. Used uniform, structured clinical evaluation; 1,695 individuals. | Logistic regression of incident AD, controlling for age, education, gender, race, and time between evaluations, shows no time trend. |
| Rajan et al. (2019) | Chicago Health and Aging Project, 1994-2012; 2,794 individuals (1,561 African American, 1,233 European American). Four urban neighborhoods on Chicago's South side--may not generalize well to U.S. population. | Prevalence and incidence of CI estimated to be about twice as high for African Americans as for European Americans. Standardized to the 2010 U.S. population, prevalence--All/African American/not African American: Age 65-74: 3.2%/11.4%/3.2%; 75-84: 16.3%/38.4%/15.4%; 85+: 52.1%/78.9%/47.0%. Incidence: 65-74: 0.4%/1.5%/0.3%; 75-84: 3.2%/6.0%/2.9%; 85+: 7.6%/11.7%/7.4%. |
| Weuve et al. (2018) | 8,906 people ages 67+r in Chicago Health and Aging Project, 1993-1996 and 2010-2012 (refresher cohorts had been added). Uses 4 tests of function. | Overall cognitive performance followed secular pattern of modest decline, inconsistent with studies showing improvement. Average scores for Blacks consistently lower than for Whites, with some evidence of narrowing until the last wave. |
| TABLE 11. Historical Cross-Sectional Prevalence of SCI at Age 65 and Older, by Sex and Age: DYNASIM 2013-2017 and NHATS 2015 | |||
|---|---|---|---|
| DYNASIM 2013-2017 | NHATS 2015 | Ratio DYNASIM/NHATS | |
| Men | |||
| 65-69 | 1.6 | 2.4 | 0.69 |
| 70-74 | 4.5 | 5.0 | 0.91 |
| 75-79 | 8.2 | 7.0 | 1.17 |
| 80-84 | 13.5 | 13.7 | 0.99 |
| 85-89 | 20.7 | 20.8 | 0.99 |
| 90+ | 29.6 | 32.0 | 0.92 |
| All 65+ | 7.1 | 7.7 | 0.92 |
| Women | |||
| 65-69 | 1.4 | 1.5 | 0.97 |
| 70-74 | 3.5 | 2.6 | 1.35 |
| 75-79 | 7.4 | 7.9 | 0.94 |
| 80-84 | 15.5 | 15.7 | 0.99 |
| 85-89 | 23.8 | 22.2 | 1.07 |
| 90+ | 38.1 | 34.3 | 1.11 |
| All 65+ | 9.0 | 8.9 | 1.01 |
| All | |||
| 65-69 | 1.5 | 1.9 | 0.81 |
| 70-74 | 3.9 | 3.7 | 1.08 |
| 75-79 | 7.8 | 7.5 | 1.04 |
| 80-84 | 14.7 | 14.9 | 0.98 |
| 85-89 | 22.7 | 21.7 | 1.05 |
| 90+ | 35.6 | 33.7 | 1.06 |
| All 65+ | 8.2 | 8.4 | 0.97 |
| SOURCES: Authors' tabulations from DYNASIM4 (runid 974, dated: April 2020) and Spillman (2019) tabulations from NHATS. NOTES: The NHATS does not have information about cognitive status for those in nursing homes. Consistent with prior literature, we assume that roughly half of all nursing home residence are severely cognitively impaired. The NHATS estimates count those who report no LTSS need as not disabled at the HIPAA-level (this affects 84 cases). Gaugler, Yu, Davila, and Shippee (2014) note that nearly two-thirds of people in nursing homes have cognitive impairment, but they include people with moderate cognitive impairment. See also discussion of statistics from the Centers for Medicare & Medicaid Services (2015) in endnote 6. | |||
| TABLE 12. Projected Cross-Sectional Age-Specific Prevalence of SCI at Ages 65 and Older: DYNASIM 2013-2055 in Selected Years, by Age | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Age | Historical | Projected | ||||||||
| 2013-17 Prevalence | 2015 Number | 2021-25 Prevalence | 2025 Number | 2031-35 Prevalence | 2035 Number | 2041-45 Prevalence | 2045 Number | 2051-55 Prevalence | 2055 Number | |
| 65-69 | 0.015 | 219,000 | 0.015 | 281,000 | 0.013 | 243,000 | 0.015 | 311,000 | 0.012 | 284,000 |
| 70-74 | 0.039 | 449,000 | 0.033 | 563,000 | 0.033 | 639,000 | 0.031 | 534,000 | 0.029 | 553,000 |
| 75-79 | 0.078 | 633,000 | 0.061 | 731,000 | 0.066 | 1,041,000 | 0.062 | 936,000 | 0.061 | 956,000 |
| 80-84 | 0.147 | 846,000 | 0.128 | 966,000 | 0.125 | 1,489,000 | 0.124 | 1,709,000 | 0.116 | 1,506,000 |
| 85-89 | 0.227 | 869,000 | 0.213 | 862,000 | 0.192 | 1,371,000 | 0.188 | 1,804,000 | 0.184 | 1,665,000 |
| 90+ | 0.356 | 802,000 | 0.355 | 921,000 | 0.306 | 1,123,000 | 0.286 | 1,737,000 | 0.296 | 2,309,000 |
| All | 0.081 | 3,818,000 | 0.070 | 4,324,000 | 0.075 | 5,906,000 | 0.085 | 7,031,000 | 0.085 | 7,273,000 |
| SOURCES: Authors' tabulations from DYNASIM4 (runid 974, dated: April 2020). NOTES: When computing prevalence rates, we pool 5 years of data to reduce year-to-year volatility. Estimates for the number of people with SCI are rounded to the nearest 1,000. Columns may not sum to totals because of rounding. | ||||||||||
| TABLE 13. Probability of Ever Experiencing SCI and Projected Life Expectancy with SCI from Age 65: By Birth Cohort, Gender, Education, and Race and Ethnicity | ||||||
|---|---|---|---|---|---|---|
| 1955-59 Birth Cohorts (reach age 65 in 2020-24) | 1965-69 Birth Cohorts (reach age 65 in 2030-34) | 1975-79 Birth Cohorts (reach age 65 in 2040-45) | ||||
| Probability Ever SCI Ages 65+ | Mean Years Probable SCI (uncond.) | Probability Ever SCI Ages 65+ | Mean Years Probable SCI (uncond.) | Probability Ever SCI Ages 65+ | Mean Years Probable SCI (uncond.) | |
| All | 0.31 | 1.54 | 0.33 | 1.65 | 0.33 | 1.72 |
| Gender | ||||||
| Men | 0.28 | 1.36 | 0.30 | 1.41 | 0.30 | 1.48 |
| Women | 0.33 | 1.72 | 0.36 | 1.88 | 0.36 | 1.97 |
| Education | ||||||
| Did not complete HS | 0.50 | 2.74 | 0.52 | 2.83 | 0.55 | 3.33 |
| HS diploma | 0.30 | 1.46 | 0.32 | 1.55 | 0.32 | 1.59 |
| Some college but less than 4 years | 0.27 | 1.31 | 0.29 | 1.46 | 0.30 | 1.44 |
| Bachelor's degree or higher | 0.30 | 1.43 | 0.29 | 1.45 | 0.29 | 1.45 |
| Race and ethnicity | ||||||
| Non-Hispanic Whites | 0.25 | 1.24 | 0.27 | 1.24 | 0.25 | 1.22 |
| Non-Hispanic Blacks | 0.42 | 2.31 | 0.41 | 2.23 | 0.44 | 2.53 |
| Hispanics | 0.48 | 2.31 | 0.48 | 2.52 | 0.46 | 2.52 |
| SOURCES: Author's tabulations from DYNASIM4 (runid 974, dated: April 2020). NOTES: Estimates of unconditional SCI years include people who never develop SCI, whereas estimates of conditional SCI include only those people who ever develop SCI. | ||||||
| TABLE 14. Average and Distribution of Projected Years with SCI from Age 65, for Those Who Become Severely Impaired: By Birth Cohort | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1955-59 Birth Cohorts (reach age 65 in 2020-24) | 1965-69 Birth Cohorts (reach age 65 in 2030-34) | 1975-79 Birth Cohorts (reach age 65 in 2040-44) | ||||||||||
| Less than 1 | 1.00-1.99 | 2.00-4.99 | 5 or more | Less than 1 | 1.00-1.99 | 2.00-4.99 | 5 or more | Less than 1 | 1.00-1.99 | 2.00-4.99 | 5 or more | |
| Distribution for All | 17.0 | 18.0 | 24.1 | 40.9 | 16.7 | 18.2 | 24.1 | 41.0 | 16.5 | 16.0 | 25.4 | 42.2 |
| Mean for all | 5.05 years | 4.99 years | 5.22 years | |||||||||
| Median for all | 3.58 years | 3.58 years | 3.83 years | |||||||||
| SOURCES: Author's tabulations from DYNASIM4 (runid 974, dated: April 2020). | ||||||||||||
| TABLE 15A. Average Projected Cost of SCI from Age 65 Onward for Paid and Unpaid LTSS (Sum): DYNASIM 1955-1959 Birth Cohorts, by Gender, Education, and Lifetime Earnings Quintile--UNCONDITIONAL | |||||
|---|---|---|---|---|---|
| Paid LTSS | Unpaid Family Care | ||||
| Total | Medicaid | Family Out-of-Pocket | Other | ||
| All | 86,200 | 34,300 | 48,100 | 3,800 | 74,400 |
| Gender | |||||
| Men | 63,100 | 25,900 | 33,800 | 3,400 | 69,000 |
| Women | 107,600 | 42,000 | 61,400 | 4,200 | 79,400 |
| Education | |||||
| Did not complete HS | 113,600 | 72,500 | 37,200 | 3,900 | 141,600 |
| HS diploma | 77,100 | 35,200 | 38,600 | 3,300 | 69,300 |
| Some college but less than 4 years | 73,700 | 29,600 | 41,300 | 2,800 | 63,500 |
| Bachelor's degree or higher | 95,700 | 21,000 | 69,500 | 5,200 | 61,300 |
| Lifetime earnings quintile | |||||
| Lowest | 96,000 | 64,700 | 28,500 | 2,800 | 92,700 |
| Second | 93,000 | 49,000 | 40,700 | 3,200 | 77,700 |
| Middle | 93,400 | 37,100 | 52,800 | 3,400 | 75,500 |
| Fourth | 77,300 | 19,500 | 52,900 | 5,000 | 67,000 |
| Highest | 75,800 | 11,000 | 60,600 | 4,200 | 63,900 |
| SOURCE: Author's tabulations from DYNASIM4 (runid 974, dated: April 2020). NOTES: Projected costs 2020 inflation-adjusted dollars. Estimates include people who never experienced severe CI. The amount of unpaid family care is computed based on the residual between LTSS need and paid care received and historical data on average hours of unpaid care received by people with dementia residing in the community. Unpaid care is valued at the median home care wage in the recipient's state of residence, based on 2019 estimates from Genworth (2019), wage-indexed to the year in which care is provided. | |||||
| TABLE 15B. Average Projected Cost of SCI from Age 65 Onward for Paid and Unpaid LTSS (PDV): DYNASIM 1955-1959 Birth Cohorts, by Gender, Education, and Lifetime Earnings Quintile--UNCONDITIONAL | |||||
|---|---|---|---|---|---|
| Paid LTSS | Unpaid Family Care | ||||
| Total | Medicaid | Family Out-of-Pocket | Other | ||
| All | 50,000 | 20,400 | 27,300 | 2,200 | 49,300 |
| Gender | |||||
| Men | 38,100 | 16,000 | 20,100 | 2,000 | 47,000 |
| Women | 61,000 | 24,500 | 34,100 | 2,400 | 51,400 |
| Education | |||||
| Did not complete HS | 73,900 | 48,000 | 23,400 | 2,600 | 100,900 |
| HS diploma | 45,600 | 21,100 | 22,600 | 1,900 | 46,300 |
| Some college but less than 4 years | 42,500 | 17,100 | 23,800 | 1,700 | 41,800 |
| Bachelor's degree or higher | 51,200 | 10,800 | 37,500 | 3,000 | 37,200 |
| Lifetime earnings quintile | |||||
| Lowest | 61,400 | 42,400 | 17,200 | 1,800 | 65,100 |
| Second | 55,300 | 29,000 | 24,400 | 1,900 | 52,500 |
| Middle | 53,600 | 21,300 | 30,300 | 2,000 | 50,200 |
| Fourth | 42,300 | 10,200 | 29,200 | 2,900 | 43,200 |
| Highest | 41,200 | 5,700 | 33,200 | 2,400 | 39,700 |
| SOURCES: Author's tabulations from DYNASIM4 (runid 974, dated: April 2020). NOTES: Projected costs are discounted to age 65 using a 2.5% real interest rate and reported in 2020 inflation-adjusted dollars. Estimates include people who never experienced SCI. The amount of unpaid family care is computed based on the residual between LTSS need and paid care received and historical data on average hours of unpaid care received by people with dementia residing in the community. Unpaid care is valued at the median home care wage in the recipient's state of residence, based on 2019 estimates from Genworth (2019), wage-indexed to the year in which care is provided | |||||
| TABLE16A. Average Projected LTSS Costs from Age 65 Onward for Those with SCI (Sum): DYNASIM 1955-1959 Birth Cohorts, by Gender, Education, and Lifetime Earnings Quintile | |||||
|---|---|---|---|---|---|
| Paid LTSS | Unpaid Family Care | ||||
| Total | Medicaid | Family Out-of-Pocket | Other | ||
| All | 281,900 | 112,100 | 157,500 | 12,400 | 243,500 |
| Gender | |||||
| Men | 223,400 | 91,800 | 119,800 | 11,900 | 244,300 |
| Women | 329,100 | 128,500 | 187,800 | 12,700 | 242,800 |
| Education | |||||
| Did not complete HS | 225,500 | 143,900 | 73,800 | 7,800 | 280,900 |
| HS diploma | 258,500 | 118,100 | 129,500 | 10,900 | 232,200 |
| Some college but less than 4 years | 274,700 | 110,300 | 154,000 | 10,300 | 236,700 |
| Bachelor's degree or higher | 364,800 | 80,000 | 265,000 | 19,800 | 233,500 |
| Lifetime earnings quintile | |||||
| Lowest | 256,100 | 172,500 | 76,100 | 7,500 | 247,300 |
| Second | 284,500 | 150,000 | 124,700 | 9,800 | 237,900 |
| Middle | 283,500 | 112,700 | 160,400 | 10,400 | 229,300 |
| Fourth | 276,300 | 69,600 | 188,900 | 17,800 | 239,200 |
| Highest | 311,200 | 45,200 | 248,900 | 17,100 | 262,200 |
| SOURCES: Author's tabulations from DYNASIM4 (runid 974, dated: April 2020). NOTES: Projected costs are reported in 2020 inflation-adjusted dollars. Estimates are restricted those who experienced SCI at ages 65 and older. The amount of unpaid family care is computed based on the residual between LTSS need and paid care received and historical data on average hours of unpaid care received by people with dementia residing in the community. Unpaid care is valued at the median home care wage in the recipient's state of residence, based on 2019 estimates from Genworth (2019), wage-indexed to the year in which care is provided. | |||||
| TABLE16B. Average Projected LTSS Costs from Age 65 Onward for Those with SCI (PDV): DYNASIM 1955-1959 Birth Cohorts, by Gender, Education, and Lifetime Earnings Quintile | |||||
|---|---|---|---|---|---|
| Paid LTSS | Unpaid Family Care | ||||
| Total | Medicaid | Family Out-of-Pocket | Other | ||
| All | 163,500 | 66,800 | 89,400 | 7,300 | 161,300 |
| Gender | |||||
| Men | 134,800 | 56,500 | 71,100 | 7,200 | 166,500 |
| Women | 186,600 | 75,000 | 104,100 | 7,400 | 157,100 |
| Education | |||||
| Did not complete HS | 146,700 | 95,300 | 46,400 | 5,100 | 200,200 |
| HS diploma | 153,000 | 70,800 | 75,800 | 6,400 | 155,400 |
| Some college but less than 4 years | 158,600 | 63,700 | 88,700 | 6,200 | 155,900 |
| Bachelor's degree or higher | 195,100 | 41,100 | 142,800 | 11,300 | 141,900 |
| Lifetime earnings quintile | |||||
| Lowest | 163,600 | 113,100 | 45,800 | 4,700 | 173,600 |
| Second | 169,200 | 88,700 | 74,700 | 5,800 | 160,800 |
| Middle | 162,700 | 64,600 | 92,000 | 6,100 | 152,500 |
| Fourth | 151,200 | 36,500 | 104,200 | 10,500 | 154,300 |
| Highest | 169,300 | 23,200 | 136,300 | 9,800 | 163,000 |
| SOURCE: Author's tabulations from DYNASIM4 (runid 974, dated: April 2020). NOTES: Projected costs are discounted to age 65 using a 2.5% real interest rate and reported in 2020 inflation-adjusted dollars. Estimates are restricted those who experienced SCI at ages 65 and older. The amount of unpaid family care is computed based on the residual between LTSS need and paid care received and historical data on average hours of unpaid care received by people with dementia residing in the community. Unpaid care is valued at the median home care wage in the recipient's state of residence, based on 2019 estimates from Genworth (2019), wage-indexed to the year in which care is provided. | |||||
| TABLE 17A. Distribution of Projected Costs of LTSS for Those Who Ever Experience SCI (Sum): DYNASIM 1955-1959 Birth Cohorts, by Gender | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Less than $10,000 | $10,000- $24,999 | $25,000- $49,999 | $50,000- $74,999 | $75,000- $99,999 | $100,000- $149,999 | $150,000- $199,999 | $200,000- $249,999 | $250,000 or More | |
| Paid Services (Family Out-of-pocket, Medicaid, Private Insurance, Other Public) | |||||||||
| Men | 36.7 | 5.6 | 4.3 | 4.4 | 3.0 | 5.2 | 5.4 | 4.7 | 30.8 |
| Women | 26.4 | 3.8 | 4.4 | 3.8 | 3.7 | 5.5 | 5.0 | 4.6 | 42.8 |
| All | 31.0 | 4.6 | 4.3 | 4.1 | 3.4 | 5.3 | 5.2 | 4.6 | 37.5 |
| Family out-of-pocket costs | |||||||||
| Men | 45.3 | 7.2 | 5.4 | 6.5 | 3.1 | 7.1 | 5.6 | 3.7 | 16.2 |
| Women | 35.6 | 5.3 | 6.6 | 5.2 | 4.4 | 6.3 | 5.3 | 4.9 | 26.3 |
| All | 40.0 | 6.2 | 6.0 | 5.8 | 3.8 | 6.7 | 5.5 | 4.4 | 21.7 |
| Medicaid costs | |||||||||
| Men | 70.0 | 1.8 | 2.4 | 2.0 | 2.3 | 3.3 | 2.8 | 2.4 | 13.0 |
| Women | 58.9 | 2.0 | 2.4 | 2.8 | 2.3 | 4.5 | 3.9 | 3.7 | 19.6 |
| All | 63.9 | 1.9 | 2.4 | 2.4 | 2.3 | 4.0 | 3.4 | 3.1 | 16.7 |
| Unpaid Family Care | |||||||||
| Men | 10.9 | 4.0 | 5.8 | 6.7 | 6.3 | 9.6 | 9.8 | 8.4 | 38.6 |
| Women | 11.1 | 2.8 | 5.8 | 7.4 | 6.1 | 10.3 | 9.6 | 10.0 | 36.8 |
| All | 11.0 | 3.4 | 5.8 | 7.1 | 6.2 | 10.0 | 9.7 | 9.3 | 37.6 |
| SOURCES: Author's tabulations from DYNASIM4 (runid 974, dated: October 2019). NOTES: Projected costs are reported in 2020 inflation-adjusted dollars. Estimates are restricted to those who experienced SCI at ages 65 and older. The amount of unpaid family care is computed based on the residual between LTSS need and paid care received and historical data on average hours of unpaid care received by people with dementia residing in the community. Unpaid care is valued at the median home care wage in the recipient's state of residence, based on 2019 estimates from Genworth (2019), wage-indexed to the year in which care is provided. | |||||||||
| TABLE 17B. Distribution of Projected Costs of LTSS for Those Who Ever Experience SCI (PDV): DYNASIM 1955-1959 Birth Cohorts, by Gender | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Less than $10,000 | $10,000- $24,999 | $25,000- $49,999 | $50,000- $74,999 | $75,000- $99,999 | $100,000- $149,999 | $150,000- $199,999 | $200,000- $249,999 | $250,000 or More | |
| Paid Services (Family Out-of-pocket, Medicaid, Private Insurance, Other Public) | |||||||||
| Men | 38.9 | 6.0 | 6.4 | 5.2 | 4.6 | 8.1 | 5.6 | 4.5 | 20.7 |
| Women | 27.7 | 5.0 | 7.0 | 5.5 | 5.1 | 7.5 | 6.7 | 6.1 | 29.4 |
| All | 32.7 | 5.5 | 6.7 | 5.3 | 4.9 | 7.8 | 6.2 | 5.4 | 25.5 |
| Family out-of-pocket costs | |||||||||
| Men | 48.8 | 7.4 | 9.2 | 5.3 | 6.5 | 6.6 | 4.4 | 3.5 | 8.4 |
| Women | 38.0 | 7.2 | 9.6 | 6.1 | 5.9 | 8.5 | 5.0 | 5.2 | 14.6 |
| All | 42.8 | 7.3 | 9.4 | 5.8 | 6.2 | 7.6 | 4.8 | 4.4 | 11.8 |
| Medicaid costs | |||||||||
| Men | 70.6 | 2.3 | 3.9 | 3.5 | 2.4 | 4.4 | 1.9 | 3.2 | 7.9 |
| Women | 59.7 | 2.8 | 4.5 | 3.9 | 4.3 | 6.2 | 5.0 | 3.4 | 10.1 |
| All | 64.6 | 2.6 | 4.2 | 3.7 | 3.5 | 5.4 | 3.6 | 3.4 | 9.1 |
| Unpaid Family Care | |||||||||
| Men | 12.2 | 5.5 | 10.0 | 8.1 | 8.0 | 13.8 | 11.7 | 9.7 | 21.1 |
| Women | 12.1 | 5.0 | 11.1 | 9.4 | 8.9 | 14.2 | 11.0 | 8.2 | 20.1 |
| All | 12.1 | 5.2 | 10.6 | 8.9 | 8.5 | 14.0 | 11.3 | 8.9 | 20.6 |
| SOURCES: Author's tabulations from DYNASIM4 (runid 974, dated: October 2019). NOTES: Projected costs are discounted to age 65 using a 2.5% real interest rate and reported in 2020 inflation-adjusted dollars. Estimates are restricted to those who experienced SCI at ages 65 and older. The amount of unpaid family care is computed based on the residual between LTSS need and paid care received and historical data on average hours of unpaid care received by people with dementia residing in the community. Unpaid care is valued at the median home care wage in the recipient's state of residence, based on 2019 estimates from Genworth (2019), wage-indexed to the year in which care is provided. | |||||||||
| TABLE 18. Projected Total LTSS Spending from Age 65 among People with Significant Disabilities and Paid Care Use, by whether Ever SCI: DYNASIM 1955-1959 Birth Cohorts, by Gender | ||||||
|---|---|---|---|---|---|---|
| All People with Significant Disabilities Who Use Paid Services | People Who Ever Experience Dementia Who Use Paid Services | People Who Do Not Experience Dementia Who Use Paid Services | ||||
| Present Value | Sum | Present Value | Sum | Present Value | Sum | |
| Total | 165,100 | 281,600 | 211,100 | 364,200 | 120,400 | 201,600 |
| Men | 145,200 | 238,700 | 185,800 | 308,200 | 107,300 | 173,900 |
| Women | 179,800 | 313,400 | 229,200 | 404,400 | 130,400 | 222,700 |
| SOURCES: Author's tabulations from DYNASIM4 (runid 974, dated: April 2020). NOTES: Projected costs are discounted to age 65 using a 2.5% real interest rate and reported in 2019 inflation-adjusted dollars. Costs as share of wealth are capped at 100% and set at that level for people who report no wealth. Conditional estimates are restricted to those who experienced SCI at ages 65 and older, and unconditional estimates include the entire population, including those who never experience SCI. "Significant" disability meets criteria set out in HIPAA. | ||||||
REFERENCES
Ahmadi-Abhari, S., M. Guzman-Castillo, P. Bandosz, M.J. Shipley, G. Muniz-Terrera, A. Singh-Manoux, M. Kivimäki, A. Steptoe, S. Capewell, M. O'Flaherty, and E.J. Brunner. 2017. "Temporal Trend in Dementia Incidence since 2002 and Projections for Prevalence in England and Wales to 2040: Modelling Study." BMJ , 358: j2856, doi:10.1136/bmj.j2856.
Aigbogun, Myrlene Sanon, Robert Stellhorn, Ann Hartry, Ross A. Baker, and Howard Fillit. 2019. "Treatment Patterns and Burden of Behavioral Disturbances in Patients with Dementia in the United States: A Claims Database Analysis." BMC Neurology, 19: 33, doi:10.1186/s12883-019-1260-3.
Alzheimer's Association. 2015. Changing the Trajectory of Alzheimer's Disease: How a Treatment by 2025 Saves Lives and Dollars. Chicago, IL: Author.
Alzheimer's Association. 2016. 2016 Alzheimer's Disease Facts and Figures. Chicago, IL: Author.
Alzheimer's Association. 2018. "Alzheimer's Association Report: 2018 Alzheimer's Disease Facts and Figures." Alzheimer's & Dementia, 14: 367-429.
Amjad, Halima, David L. Roth, Orla C. Sheehan, Constantine G. Lyketsos, Jennifer L. Wolff, and Quincy M. Samus. 2018. "Underdiagnosis of Dementia: An Observational Study of Patterns in Diagnosis and Awareness in US Older Adults." Journal of General Internal Medicine, doi:10.1007/s11606-018-4377-y.
Barnes, Deborah E., and Kristine Yaffe. 2011. "The Projected Impact of Risk Factor Reduction on Alzheimer's Disease Prevalence." Lancet Neurology, 10(9): 819-828, doi:10.1016/S1474-4422(11)70072-2.
Bennett, David A., Julie A. Schneider, Aron S. Buchman, Lisa L. Barnes, Patricia A. Boyle, and Robert S. Wilson. 2012. "Overview and Findings from the Rush Memory and Aging Project." Current Alzheimer Research, 9(6): 646-663.
Bennett, David A., Robert S. Wilson, Zoe Arvanitakis, Patricia A. Boyle, Leyla de Toledo-Morrell, and Julie A. Schneider. 2013. "Selected Findings from the Religious Orders Study and Rush Memory and Aging Project." Journal Alzheimers Disease, 33(0): S397-S403, doi:10.3233/JAD-2012-129007.
Bipartisan Policy Center (BPC). 2016. Initial Recommendations to Improve the Financing of Long-Term Care. Washington, DC: Author. http://bipartisanpolicy.org/library/long-term-care-financing-recommendations/.
Bipartisan Policy Center (BPC). 2017. Financing Long-Term Services and Supports: Seeking Bipartisan Solutions in Politically Challenges Times. Washington, DC: Author. https://cdn.bipartisanpolicy.org/wp-content/uploads/2017/07/BPC-Health-Financing-Long-Term-Services-and-Supports.pdf.
Board of Trustees, Federal Old-Age and Survivors Insurance and Disability Insurance Trust Funds. 2019. 2019 Annual Report of the Board of Trustees of the Federal Old-Age and Survivors Insurance and Disability Insurance Trust Funds. Washington, DC: Author.
Boards of Trustees of the Federal Hospital Insurance and Federal Supplementary Medical Insurance Trust Funds. 2019. 2019 Annual Report of the Boards of Trustees of the Federal Hospital Insurance and Federal Supplementary Medical Insurance Trust Funds. Washington, DC: Author.
Brayne, Carol, and Paul Calloway. 1988. "Normal Ageing, Impaired Cognitive Function, and Senile Dementia of the Alzheimer's Type: A Continuum?" Lancet, 331(8597): 1265-1267, doi:10.1016/S0140-6736(88)92081-8.
Brodaty, Henry, Katrin Seeher, and Louisa Gibson. 2012. "Dementia Time to Death: A Systematic Literature Review on Survival Time and Years of Life Lost in People with Dementia." International Psychogeriatrics, 24(7): 1034-1045.
Brookmeyer, R., N. Abdalla, C.H. Kawas, and M.M. Corrada. 2018. "Forecasting the Prevalence of Preclinical and Clinical Alzheimer's Disease in the United States." Alzheimers & Dementia, 14(2): 121-129.
Brookmeyer, R., M. Corrada, F. Curriero, and C. Kawas. 2002. "Survival Following a Diagnosis of Alzheimer's Disease." Archives of Neurology, 59: 1764-1767.
Brookmeyer, R., D.A. Evans, L. Hebert, K.M. Langa, S.G. Heeringa, B.L. Plassman, and W.A. Kukull. 2011. "National Estimates of the Prevalence of Alzheimer's Disease in the United States." Alzheimers & Dementia, 7: 61-73.
Brookmeyer, R., S. Gray, and C. Kawas. 1998. "Projections of Alzheimer's Disease in the United States and the Public Health Impact of Delaying Disease Onset." American Journal of Public Health, 88(9): 1337-42.
Broyles, Ila H., Amy Huebeler, Ira Dave, Emily Graf, Qinghua Li, Lauren Palmer, Zhanlian Feng, Sarita L. Karon, 2020. Functional Trajectories at the End of Life for Individuals with Dementia: Final Report. Washington, DC: U.S. Department of Health and Human Services, Office of the Assistant Secretary for Planning and Evaluation. https://aspe.hhs.gov/basic-report/functional-trajectories-end-life-individuals-dementia-final-report.
Centers for Disease Control and Prevention. 2011. Cognitive Impairment: A Call for Action Now! Washington, DC: U.S. Department of Health and Human Services. https://www.cdc.gov/aging/pdf/cognitive_impairment/cogimp_poilicy_final.pdf.
Centers for Medicare & Medicaid Services (CMS). 2015. Nursing Home Data Compendium: 2015 Edition. Washington, DC: U.S. Department of Health and Human Services.
Chêne, G., A. Beiser, R. Au, S.R. Preis, P.A. Wolf, C. Dufouil, and S. Seshadri. 2015. "Gender and Incidence of Dementia in the Framingham Heart Study from Mid-Adult Life." Alzheimers & Dementia, 11(3): 310-320. doi:10.1016/j.jalz.2013.10.005.
Cheng, G., C. Huang, H. Deng, and H. Wang. 2012. "Diabetes as a Risk Factor for Dementia and Mild Cognitive Impairment: A Meta-Analysis of Longitudinal Studies." Internal Medicine Journal, 42(5): 484-491.
Choi, HwaJung, Robert F. Schoeni, Linda G. Martin, and Kenneth M. Langa. 2018. "Trends in the Prevalence and Disparity in Cognitive Limitations of Americans 55–69 Years Old." Journals of Gerontology: Social Sciences, 73(S1): S29-S37, doi:10.1093/geronb/gbx155.
Coe, Norma B., Meghan M. Skira, and Eric B. Larson. 2018. "A Comprehensive Measure of the Costs of Caring for a Parent: Differences According to Functional Status." Journal of the American Geriatrics Society, 66(10): 2003-2008.
Coe, Norma, Lindsay White, Anirban Basu, and Eric Larson. 2018. "Public Spending on Acute and Long-Term Care for Alzheimer's and Related Dementias." Paper presented at Southern Economic Association Meeting, November 18, 2018.
Cohen, Marc A. 2016. "The State of the Long-Term Care Insurance Market." In E.C. Nordman, ed., The State of Long-Term Care Insurance: The Market, Challenges, and Future Innovations. Washington, DC: National Association of Insurance Commissioners and the Center for Insurance Policy and Research.
Commission on Long-Term Care. 2013. Report to Congress. Washington, DC: U.S. Senate. http://www.medicareadvocacy.org/wp-content/uploads/2014/01/Commission-on-Long-Term-Care-Final-Report-9-18-13-00042470.pdf.
Congressional Budget Office. 2013. Rising Demand for Long-Term Services and Supports for Elderly People. Washington, DC: Congress of the United States. https://www.cbo.gov/publication/44363.
Corrada, María M., Ron Brookmeyer, Annlia Paganini-Hill, Daniel Berlau, and Claudia H. Kawas. 2010. "Dementia Incidence Continues to Increase with Age in the Oldest Old The 90+ Study." Annals of Neurology, 67(1): 114-121, doi:10.1002/ana.21915.
Crimmins, Eileen M., Yasuhiko Saito, Jung Ki Kim, Yuan S. Zhang, Isaac Sasson, and Mark D. Hayward. 2018. "Educational Differences in the Prevalence of Dementia and Life Expectancy with Dementia: Changes from 2000 to 2010." Journals of Gerontology: Social Sciences, 73(S1): S20-S28, doi:10.1093/geronb/gbx135.
Cukierman, T., H.C. Gerstein, and J.D. Williamson. 2005. "Cognitive Decline and Dementia in Diabetes--Systematic Overview of Prospective Observational Studies." Diabetologia, 48: 2460-2469.
Davis, Matthew, Thomas O'Connell, Scott Johnson, Stephanie Cline, Elizabeth Merikle, Ferenc Martenyi, and Kit Simpson. 2018. "Estimating Alzheimer's Disease Progression Rates from Normal Cognition through Mild Cognitive Impairment and Stages of Dementia." Current Alzheimer Research, 15(8): 777-788, doi:10.2174/1567205015666180119092427.
Deb, Arijita, James Douglas Thornton, Usha Sambamoorthi, and Kim Innes. 2017. "Direct and Indirect Cost of Managing Alzheimer's Disease and Related Dementias in the United States." Expert Review of Pharmacoeconomics and Outcomes Research, 17(2): 189-202, doi:10.1080/14737167.2017.1313118.
Dodge, Hiroko H., Jian Zhu, Ching-Wen Lee, Chung-Chou Ho Chang, and Mary Ganguli. 2014. "Cohort Effects in Age-Associated Cognitive Trajectories." Journals of Gerontology, Series A: Biological Sciences and Medical Sciences, 69: 687-694, doi:10.1093/gerona/glt181.
Dufouil, Carole, Alexa Beiser, Geneviève Chêne, and Sudha Seshadri. 2018. "Are Trends in Dementia Incidence Associated with Compression in Morbidity? Evidence from the Framingham Heart Study." Journals of Gerontology: Social Sciences, 73(S1): S65-S72, doi:10.1093/geronb/gby001.
Erkinjuntti, Timo, Truls Østbye, Runa Steenhuis, and Vladimir Hachinski. 1997. "The Effect of Different Diagnostic Criteria on the Prevalence of Dementia." New England Journal of Medicine, 337: 1667-1674.
Evans, D.A., P.A. Scherr, N.R. Cook, M.S. Albert, H.H. Funkenstein, et al. 1992. "The Impact of Alzheimer's Disease in the United States Population." In The Oldest Old, ed. R.M. Suzman, D.P. Willis, and K.G. Manton, pp. 283-399. New York, NY: Oxford University Press.
Farina, Mateo P., Mark D. Hayward, Jung Ki Kim, and Eileen M. Crimmins. 2019. "Racial and Educational Disparities in Dementia and Dementia-Free Life Expectancy." Journals of Gerontology: Social Sciences. Advance Access.
Favreault, M. 2017. "Selected Sensitivity Analyses of DYNASIM Projections of Long-Term Services and Support Using Alternative Disability Assumptions." Report to U.S. Department of Health and Human Services, Office of the Assistant Secretary for Planning and Evaluation. Washington, DC: Urban Institute.
Favreault, Melissa M. 2018. How Might Earnings Patterns and Interactions among Certain Provisions in OASDI Solvency Packages Affect Financing and Distributional Goals? Working Paper 2018-2. Chestnut Hill, MA: Center for Retirement Research at Boston College. http://crr.bc.edu/wp-content/uploads/2018/03/wp_2018-2-1.pdf.
Favreault, M., B. Butrica, and S. Mudrazija. 2019 (forthcoming). "Understanding the Lifetime Effects of Providing Informal Care: Final Report." Report to U.S. Department of Labor. Washington, DC: Urban Institute.
Favreault, Melissa, and Judith Dey. 2015. Long-Term Services and Supports for Older Americans: Risks and Financing. Washington, DC: U.S. Department of Health and Human Services, Office of the Assistant Secretary for Planning and Evaluation. http://aspe.hhs.gov/basic-report/long-term-services-and-supports-older-americans-risks-and-financing-research-brief.
Favreault, Melissa, and Judith Dey. 2020. Long-Term Services and Supports for Older Americans: Risks and Financing, 2020. Washington, DC: US Department of Health and Human Services, Office of the Assistant Secretary for Planning and Evaluation.
Fisher, Gwenith G., Halimah Hassan, Jessica D. Faul, Willard L. Rodgers, and David R. Weir. 2017. Health and Retirement Study Imputation of Cognitive Functioning Measures: 1992-2014 (Final Release Version) Data Description. Survey Research Center. Ann Arbor, MI: University of Michigan.
Fishman, Ezra. 2017. "Risk of Developing Dementia at Older Ages in the United States." Demography, 54: 1897-1919, doi:10.1007/s13524-017-0598-7.
Freedman, Vicki A., Judith D. Kasper, Brenda C. Spillman, and Brenda L. Plassman. 2018. "Short-Term Changes in the Prevalence of Probable Dementia: An Analysis of the 2011-2015 National Health and Aging Trends Study." Journals of Gerontology: Series B, 73(S1): S48-S56, doi:10.1093/geronb/gbx144.
Friedman, Esther M., Regina A. Shih, Kenneth M. Langa, and Michael D. Hurd. 2015. "US Prevalence and Predictors of Informal Caregiving for Dementia." Datawatch. Health Affairs, 34(10): 1637-1641.
Ganguli, Mary, Hiroko H. Dodge, Changyu Shen, Rajesh S. Pandav, and Steven D, DeKosky. 2005. "Alzheimer Disease and Mortality: A 15-Year Epidemiological Study." Archives of Neurology, 62: 779-784.
Garcia, Marc A., Brian Downer, Chi-Tsun Chiu, Joseph L. Saenz, Sunshine Rote, and Rebeca Wong. 2017. "Racial/Ethnic and Nativity Differences in Cognitive Life Expectancies among Older Adults in the United States." Gerontologist, 59(2).
Gardner, Raquel C., Victor Valcour, and Kristine Yaffe. 2013. "Dementia in the Oldest Old: A Multi-Factorial and Growing Public Health Issue." Alzheimer's Research & Therapy, 5: 27, http://alzres.com/content/5/4/27.
Gaugler, Joseph E., Fang Yu, Heather W. Davila, and Tetyana Shippee. 2014. "Alzheimer's Disease and Nursing Homes." Health Affairs, 33(4): 650-657, doi:10.1377/hlthaff.2013.1268.
Genworth. 2019. Cost of Care Survey, 2019: Median Cost Data Tables.
Gilsanz, Paola, Maria M. Corrada, Claudia H. Kawas, Elizabeth Rose Mayeda, M. Maria Glymour, Charles P. Quesenberry Jr., Catherine Lee, and Rachel A. Whitmer. 2019. "Incidence of Dementia after Age 90 in a Multiracial Cohort." Alzheimer's & Dementia, doi:10.1016/j.jalz.2018.12.006.
Goldman, D.P., D. Lakdawalla, P.C. Michaud, C. Eibner, Y. Zheng, A. Gailey, I. Vaynman, J. Sullivan, B. Tysinger, D. Ermini Leaf. 2015. The Future Elderly Model: Technical Documentation.
Hales, Craig M., Margaret D. Carroll, Cheryl D. Fryar, and Cynthia L. Ogden. 2017. Prevalence of Obesity Among Adults and Youth: United States, 2015-2016. NCHS Data Brief No. 288. Hyattsville, MD: U.S. Department of Health and Human Services.
Hall, K.S., S. Gao, O. Baiyewu, K.A. Lane, O. Gureje, J. Shen, A. Ogunniyi, J.R. Murrell, F.W. Unverzagt, J. Dickens, V. Smith-Gamble, and H.C. Hendrie. 2009. "Prevalence Rates for Dementia and Alzheimer's Disease in African Americans: 1992 vs. 2001." Alzheimer's & Dementia, 5(3): 227-233, doi:10.1016/j.jalz.2009.01.026.
Hansen Hunter and Company PC. 2018. A Report on Shortfalls in Medicaid Funding for Nursing Center Care. Washington, DC: American Health Care Association. https://www.ahcancal.org/facility_operations/medicaid/Documents/2017%20Shortfall%20Methodology%20Summary.pdf.
Hatfield, Laura, Melissa M. Favreault, Thomas G. McGuire, and Michael E. Chernew. 2016. "Modeling Health Care Spending Growth of Older Adults." Health Services Research, 53 (1): 138-55.
Hebert, L.E., J.L. Bienias, N.T. Aggarwal, R.S. Wilson, D.A. Bennett, R.C. Shah, and D.A. Evans. 2010. "Change in Risk of Alzheimer Disease over Time." Neurology, 75: 786-791, doi:10.1212/WNL.0b013e3181f0754f.
Hebert, Liesi E., Jennifer Weuve, Paul A. Scherr, and Denis A. Evans. 2013. "Alzheimer Disease in the United States (2010-2050) Estimated Using the 2010 Census." Neurology, 80: 1778-1783.
Helzner, E.P., D.N. Scarmeas, S. Cosentino, M.X. Tang, N. Schupf, and Y. Stern. 2008. "Survival in Alzheimer Disease: A Multiethnic, Population-based Study of Incident Cases." Neurology, 71: 1489-1495.
Hudomiet, Péter, Michael D. Hurd, and Susann Rohwedder. 2018. "Dementia Prevalence in the United States in 2000 and 2012: Estimates Based on a Nationally Representative Study." Journals of Gerontology: Social Sciences, 73(S1): S10-S19, doi:10.1093/geronb/gbx169.
Hudomiet, Péter, Michael D. Hurd, and Susann Rohwedder. 2018. "The Relationship between Lifetime Out-of-Pocket Medical Expenditures, Dementia, and Socioeconomic Status in the U.S." Journal of the Economics of Ageing, doi:10.1016/j.jeoa.2018.11.006.
Hugo, Julie, and Mary Ganguli. 2014. "Dementia and Cognitive Impairment: Epidemiology, Diagnosis, and Treatment." Clinics in Geriatric Medicine, 30(3): 421-442, doi:10.1016/j.cger.2014.04.001.
Hurd, M.D., P. Martorell, A. Delavande, K.J. Mullen, and K.M. Langa. 2013. "Monetary Costs of Dementia in the United States." New England Journal of Medicine, 368: 1326-34.
Hurd, Michael D., Paco Martorell, and Kenneth Langa. 2015. "Future Monetary Costs of Dementia in the United States under Alternative Dementia Prevalence Scenarios." Journal Population Ageing, 8(1-2): 101-112, doi:10.1007/s12062-015-9112-4.
Jacobson, Gretchen, Anthony Damico, and Tricia Neuman 2018. "A Dozen Facts about Medicare Advantage." Data Note. San Francisco, CA: Henry J. Kaiser Family Foundation. http://files.kff.org/attachment/Data-Note-A-Dozen-Facts-About-Medicare-Advantage.
James, Bryan D., Sue E. Leurgans, Liesi E. Hebert, Paul A. Scherr, Kristine Yaffe, and David A. Bennett. 2014. "Contribution of Alzheimer Disease to Mortality in the United States." Neurology, 82: 1045-50.
Johnson, Elizabeth, Ron Brookmeyer, and Kathryn Ziegler-Graham. 2007. "Modeling the Effect of Alzheimer's Disease on Mortality." International Journal of Biostatistics, 3(1).
Joling, Karlijn J., Olin Janssen, Anneke L. Francke, Robert A. Verheij, Birgit I. Lissenberg-Witte, Pieter-Jelle Visser, Hein P.J. van Hout. 2020. "Time from Diagnosis to Institutionalization and Death in People with Dementia." Alzheimer's Dementia, 16: 662-671, doi:10.1002/alz.12063.
Jutkowitz, Eric, Robert L. Kane, Joseph E. Gaugler, Richard F. MacLehose, Bryan Dowd, and Karen M. Kuntz. 2017. "Societal and Family Lifetime Cost of Dementia: Implications for Policy." Journal of the American Geriatric Society, 65(10): 2169-2175, doi:10.1111/jgs.15043.
Jutkowitz, Eric, Richard F. MacLehose, Joseph E. Gaugler, Bryan Dowd, Karen M. Kuntz, and Robert L. Kane. 2017. "Risk Factors Associated with Cognitive, Functional, and Behavioral Trajectories of Newly Diagnosed Dementia Patients." Journals of Gerontology: Medical Sciences, 72(2): 251-258.
Kasper, Judith D., Vicki A. Freedman, and Brenda Spillman. 2013. Classification of Persons by Dementia Status in the National Health and Aging Trends Study. Technical Paper #5. Baltimore, MD: Johns Hopkins University School of Public Health. httpAvailable at https://nhats.org/.
Kasper, Judith D., Vicki A. Freedman, Brenda C. Spillman, and Jennifer L. Wolff. 2015. "The Disproportionate Impact of Dementia on Family and Unpaid Caregiving to Older Adults." Health Affairs, 34(10): 1642-1649.
Katz, Mindy J., Richard B. Lipton, Charles B. Hall, Molly E. Zimmerman, Amy E. Sanders, Joe Verghese, Dennis W. Dickson, and Carol A. Derby. 2012. "Age-specific and Sex-specific Prevalence and Incidence of Mild Cognitive Impairment, Dementia, and Alzheimer Dementia in Blacks and Whites: A Report from the Einstein Aging Study." Alzheimer Disease and Associated Disorders, 26(4): 335-43, doi:10.1097/WAD.0b013e31823dbcfc.
Kelley, Amy S., Kathleen McGarry, Rebecca Gorges, and Jonathan S. Skinner. 2015. "The Burden of Health Care Costs for Patients with Dementia in the Last 5 Years of Life." Annals of Internal Medicine, 163(10): 729-736. doi:10.7326/M15-0381.
Kubendran, Sindhu, Ross DeVol, and Anusuya Chatterjee. 2016. The Price Women Pay for Dementia: Strategies to Ease Gender Disparity and Economic Costs. Santa Monica, CA: Milken Institute.
Lamb, Vicki L., Frank A. Sloan, and Anil S. Nathan. 2008. "Dementia and Medicare at Life's End." Health Services Research, 43(2): 714-732.
Langa, K.M., M.E. Chernew, M.U. Kabeto, A.R. Herzog, M.B. Ofstedal, R.J. Willis, R.M. Wallace, L.M. Mucha, W.L. Straus, and A.M. Fendrick. 2001. "National Estimates of the Quantity and Cost of Informal Caregiving for the Elderly with Dementia." Journal of General Internal Medicine, 16: 770-778.
Langa, K.M., E.B. Larson, E.M. Crimmins, J.D. Faul, D.A. Levine, M.U. Kabeto, and D.R. Weir. 2016. "A Comparison of the Prevalence of Dementia in the United States in 2000 and 2012." JAMA Internal Medicine, 177(1): 51-58, doi:10.1001/jamainternmed.2016.6807.
Langa, K.M., E.B. Larson, J.H. Karlawish, D.M. Cutler, M.U. Kabeto, S.Y. Kim, and A.B. Rosen. 2008. "Trends in the Prevalence and Mortality of Cognitive Impairment in the United States: Is there Evidence of a Compression of Cognitive Morbidity?" Alzheimer's & Dementia, 4: 134-144, doi:10.1016/j. jalz.2008.01.001.
Larson, Eric B., Marie-Florence Shadlen, Li Wang, Wayne C. McCormick, James D. Bowen, Linda Teri, and Walter A. Kukull. 2004. "Survival after Initial Diagnosis of Alzheimer Disease." Annals of Internal Medicine, 140(7): 501-511.
LeadingAge. 2016. LeadingAge Pathways Report: Perspectives on the Challenges of Financing Long-Term Services and Supports. Washington, DC: Author. http://www.leadingage.org/sites/default/files/Pathways_Report_February_2016.pdf.
Lewin Group. 2015. "Appendix A: Methodology." Appendix to Alzheimer's Association report.
Li, Q., D. Ramirez, W. Chi, N. Chong, J. Williams, and S.L. Karon. 2017. Trend in Disability and Dementia Prevalence from 2011 to 2015: Exploratory Analysis of National Health and Aging Trends Study Data. Report to the U.S. Department of Health and Human Services, Office of the Assistant Secretary for Planning and Evaluation. Research Triangle Park, NC: RTI International.
Lichtenstein, Maya L., Nader Fallah, Benita Mudge, Ging-Yuek R. Hsiung, Dean Foti, B. Lynn Beattie, Howard H. Feldman. 2018. "16-Year Survival of the Canadian Collaborative Cohort of Related Dementias." Canadian Journal of Neurological Sciences, 45: 367-374.
Lièvre, A., D. Alley, and E.M. Crimmins. 2008. "Educational Differentials in Life Expectancy with Cognitive Impairment among the Elderly in the United States." Journal of Aging and Health, 20(4): 456-77.
Lin, Pei‐Jung, Yue Zhong, Howard M. Fillit, Er Chen, and Peter J. Neumann. 2016. "Medicare Expenditures of Individuals with Alzheimer's Disease and Related Dementias or Mild Cognitive Impairment Before and After Diagnosis." Journal of the American Geriatrics Society, 64(8), doi:10.1111/jgs.14227.
Loef, Martin, and Harald Walach. 2013. "Midlife Obesity and Dementia: Meta-Analysis and Adjusted Forecast of Dementia Prevalence in the United States and China." Obesity, 21: E51-55.
Long-Term Care Financing Collaborative. 2016. A Consensus Framework for Long-Term Care Financing Reform. Washington, DC: Convergence Center for Policy Resolution. http://morningconsult.com/wp-content/uploads/2016/02/LTCFC-Final-Report-EMBARGOED-2-22-16.pdf.
Matthews, Kevin A., Wei Xu, Anne H. Gaglioti, James B. Holt, Janet B. Croft, Dominic Mack, and Lisa C. McGuire. 2019. "Racial and Ethnic Estimates of Alzheimer's Disease and Related Dementias in the United States (2015-2060) in Adults Aged > 65 Years." Alzheimer's & Dementia, 15(1): 17-24, doi:10.1016/j.jalz.2018.06.3063.
Matthews, F.E., A. Arthur, L.E. Barnes, J. Bond, C. Jagger, L. Robinson, and C. Brayne. 2013. "A Two-Decade Comparison of Prevalence of Dementia in Individuals Aged 65 Years and Older from Three Geographical Areas of England: Results of the Cognitive Function and Ageing Study I and II." Lancet, 382(9902): 1405-12.
Mayeda, E.R., M.M. Glymour, C.P. Quesenberry, and R.A. Whitmer. 2016. "Inequalities in Dementia Incidence between Six Racial and Ethnic Groups over 14 Years." Alzheimer's & Dementia, 12(3): 216-224, doi:10.1016/j.jalz.2015.12.007.
Mayeda, Elizabeth R., M. Maria Glymour, Charles P. Quesenberry, Julene K. Johnson, Eliseo J. Pérez-Stable, Rachel A. Whitmer. 2017. "Survival after Dementia Diagnosis in Five Racial/Ethnic Groups." Alzheimer's & Dementia, 13: 761-769.
Mueller, Christoph, Pinar Soysal, Arvid Rongve, Ahmet Turan Isik, Trevor Thompson, Stefania Maggi, Lee Smith, Cristina Basso, Robert Stewart, Clive Ballard, John T. O'Brien, Dag Aarsland, Brendon Stubbs, and Nicola Veronese. 2019. "Survival Time and Differences between Dementia with Lewy Bodies and Alzheimer's Disease Following Diagnosis: A Meta-Analysis of Longitudinal Studies." Ageing Research Reviews, 50: 72-80.
Murtaugh, C.M., B.C. Spillman, and X. (D.) Wang. 2011. "Lifetime Risk and Duration of Chronic Disease and Disability." Journal of Aging and Health, 23(3): 554-77.
National Academies of Sciences, Engineering, and Medicine. 2017. Preventing Cognitive Decline and Dementia: A Way Forward. Washington, DC: National Academies Press. doi:10.17226/24782.
Neuman, Patricia, and Gretchen A. Jacobson. 2018. "Medicare Advantage Check-Up." New England Journal of Medicine, 379(22): 2163-2172.
Newhouse, Joseph P., Mary Price, J. Michael McWilliams, and Thomas G. McGuire. "How Much Favorable Selection Is Left in Medicare Advantage?" American Journal of Health Economics, 1(1): 1-26. doi:10.1162/AJHE_a_00001.
Newquist, Deborah D., Marguerite DeLiema, and Kathleen H. Wilber. 2015. "Beware of Data Gaps in Home Care Research: The Streetlight Effect and Its Implications for Policy Making on Long-Term Services and Supports." Medical Care Research and Review, 72(5): 622-640, doi:10.1177/1077558715588437.
Omorogieva, Ojo, and Joanne Brooke. 2015. "Evaluating the Association between Diabetes, Cognitive Decline and Dementia." International Journal of Environmental Research in Public Health, 12: 8281-8294.
Ory, Marcia G., Richard R. Hoffman III, Jennifer L. Yee, Sharon Tennstedt, and Richard Schulz. 1999. "Prevalence and Impact of Caregiving: A Detailed Comparison Between Dementia and Nondementia Caregivers." Gerontologist, 39(2): 177-185.
Plassman, B.L., K.M. Langa, G.G. Fisher, S.G. Heering, D.R. Weir, M.B. Ofstedal, J.R. Burke, M.D. Hurd, G.G. Potter, W.L. Rodgers, D.C. Steffens, R.J. Willis, and R.B. Wallace. 2007. "Prevalence of Dementia in the United States: The Aging, Demographics, and Memory Study." Neuroepidemiology, 29:125-132, doi:10.1159/000109998.
Plassman, Brenda L., Kenneth M. Langa, Ryan J. McCammon, Gwenith G. Fisher, Guy G. Potter, James R. Burke, David C. Steffens, Norman L. Foster, Bruno Giordani, Frederick W. Unverzagt, Kathleen A Welsh-Bohmer, Steven G. Heeringa, David R. Weir, and Robert B. Wallace. 2011. "Incidence of Dementia and Cognitive Impairment Not Dementia in the United States." Annals of Neurology, 70(3): 418-426, doi:10.1002/ana.22362.
Potter, Guy G., Brenda L. Plassman, James R. Burke, Mohammed U. Kabeto, Kenneth M. Langa, David J. Llewellyn, Mary A. M. Rogers, and David C. Steffens. 2009. "Cognitive Performance and Informant Reports in the Diagnosis of Cognitive Impairment and Dementia in African Americans and Whites." Alzheimer's and Dementia, 5(6): 445-453, doi:10.1016/j.jalz.2009.04.1234.
Prince, Martin, Anders Wimo, Maëlenn Guerchet, Gemma-Claire Ali, Yu-Tzu Wu, Matthew Prina. 2015. World Alzheimer Report 2015 The Global Impact of Dementia: An Analysis of Prevalence, Incidence, Cost and Trends. London: Alzheimer's Disease International.
Prince, Martin, Jim Jackson, Cleusa P Ferri, Renata Sousa, Emiliano Albanese, Wagner S Ribeiro, and Mina Honyashiki. 2009. World Alzheimer Report 2009. London: Alzheimer's Disease International.
Pyenson, Bruce, Tia Goss Sawhney, Charles Steffens, David Rotter, Susan Peschin, James Scott, and Ellen Jenkins, 2019. "The Real-World Medicare Costs of Alzheimer Disease: Considerations for Policy and Care." Journal of Managed Care and Specialty Pharmacy, 25(7): 800-09.
Rahman, Momotazur, Laura Keohane, Amal N. Trivedi, and Vincent Mor. 2015. "High-Cost Patients Had Substantial Rates of Leaving Medicare Advantage and Joining Traditional Medicare." Health Affairs, 34(10): 1675-1681, doi:10.1377/hlthaff.2015.0272.
Rainville, Chuck, Laura Skufca, and Laura Mehegan. 2016. Family Caregiving and Out-of-Pocket Costs: 2016 Report. Washington, DC: AARP. http://www.aarp.org/content/dam/aarp/research/surveys_statistics/ltc/20….
Rajan, Kumar B., Jennifer Weuve, Lisa L. Barnes, Robert S. Wilson, and Denis A. Evans. 2019. "Prevalence and Incidence of Clinically Diagnosed Alzheimer's Disease Dementia from 1994 to 2012 in a Population Study." Alzheimer's & Dementia 15(1): 1-7.
Reuser, Mieke, Frans J. Willekens, Luc Bonneux. 2011. "Higher Education Delays and Shortens Cognitive Impairment. A Multistate Life Table Analysis of the US Health and Retirement Study." European Journal of Epidemiology 26: 395-403. DOI 10.1007/s10654-011-9553-x
Robitaille, A., A. van den Hout, R. J. M. Machado, D. A. Bennett, I. Cuki, I. J. Deary, S. M. Hofera, E. O. Hoogendijk, M. Huisman, B. Johansson, A. V. Koval, M. van der Noordt, A. M. Piccinin, J. J. M. Rijnhart, A. Singh-Manoux, J. Skoog, I. Skoog, J. Starre, L. Vermunt, S. Clouston, G. Muniz Terrera. 2018. "Transitions across Cognitive States and Death among Older Adults in Relation to Education: A Multistate Survival Model using Data from Six Longitudinal Studies." Alzheimer's & Dementia 14: 462-472.
Rocca, W.A., R.C. Petersen, D.S. Knopman, L.E. Hebert, D.A. Evans, K.S. Hall, S. Gao, F.W. Unverzagt, K.M. Langa, E.B. Larson, and L.R. White. 2011. "Trends in the Incidence and Prevalence of Alzheimer's Disease, Dementia, and Cognitive Impairment in the United States." Alzheimer's and Dementia, 7(1): 80-93, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3026476/pdf/nihms253378.pdf.
Satizabal, C.L., A.S. Beiser, V. Chouraki, G. Chêne, C. Dufouil, and S. Seshadri. 2016. "Incidence of Dementia over Three Decades in the Framingham Heart Study." New England Journal of Medicine, 374: 523-32, http://www.nejm.org/doi/pdf/10.1056/NEJMoa1504327.
Schmitz, Al, and Chris Giese. 2019. "Is Insurance the Answer to the Long-Term-Care Financing Challenge?" Generations, 43(1): 86-88.
Seavey, Dorie, with Abby Marquand. 2011. Caring in America: A Comprehensive Analysis of the Nation's Fastest‐Growing Jobs: Home Health and Personal Care Aides. Bronx, NY: PHI. https://phinational.org/.
Selvin, Elizabeth, and Mohammed K. Ali. 2017. "Declines in the Incidence of Diabetes in the U.S.: Real Progress or Artifact?" Diabetes Care, 40: 1139-1143, doi.org/10.2337/dc16-2442
Seshadri, Sudha, Alexa Beiser, Margaret Kelly-Hayes, Carlos S. Kase, Rhoda Au, William B. Kannel, Philip A. Wolf. 2006. "The Lifetime Risk of Stroke Estimates from the Framingham Study." Stroke, 37(2): 345-350.
Sloane, P.D., S. Zimmerman, C. Suchindran, P. Reed, L. Wang, M. Boustani, and S. Sudha. 2002. "The Public Health Impact of Alzheimer's Disease, 2000-2050: Potential Implication of Treatment Advances." Annual Review of Public Health, 23: 213-31.
Spillman, Brenda C., Jennifer Wolff, Vicki A. Freedman, and Judith D. Kasper. 2014. Informal Caregiving for Older Americans: An Analysis of the 2011 National Study of Caregiving. Washington, DC: U.S. Department of Health and Human Services, Office of the Assistant Secretary for Planning and Evaluation. https://aspe.hhs.gov/report/informal-caregiving-older-americans-analysis-2011-national-study-caregiving.
Sposato, Luciano A., Moira K. Kapral, Jiming Fang, Sudeep S. Gill, Daniel G. Hackam, Lauren E. Cipriano, and Vladimir Hachinski. 2015. "Declining Incidence of Stroke and Dementia: Coincidence or Prevention Opportunity?" JAMA Neurology, 72(12): 1529-1531.
Stallard, E. 2011. "Estimates of the Incidence, Prevalence, Duration, Intensity and Cost of Chronic Disability among the U.S. Elderly." North American Actuarial Journal, 15(1): 32-58.
Stallard, P.J.E., and Anatoliy I. Yashin. 2016. Long Term Care Morbidity Improvement Study: Estimates for the Non-Insured U.S. Elderly Population Based on the National Long Term Care Survey 1984-2004. Schaumburg, IL: Society of Actuaries.
Steenland, Kyle, Felicia C. Goldstein, Allan Levey, and Whitney Wharton. 2016. "A Meta-Analysis of Alzheimer's Disease Incidence and Prevalence Comparing African-Americans and Caucasians." Journal of Alzheimer's Disease, 50(1): 71-76. doi:10.3233/JAD-150778.
Sun, Jia-Hao, Lan Tan, and Jin-Tai Yu. 2014. "Post-stroke Cognitive Impairment: Epidemiology, Mechanisms and Management." Annals of Translational Medicine, 2(8): 80.
Suthers, K., J.K. Kim, and E. Crimmins. 2003. "Life Expectancy with Cognitive Impairment in the Older Population of the United States." Journals of Gerontology: Social Sciences, 58B(3): S179-86.
Taylor, Donald H. Jr., Truls Østbye, Kenneth M. Langa, David Weir, and Brenda L. Plassman. 2009. "The Accuracy of Medicare Claims as an Epidemiological Tool: The Case of Dementia Revisited." Journal of Alzheimer's Disease, 17(4): 807-815. doi:10.3233/JAD-2009-1099.
Tom, Sarah E., Rebecca A. Hubbard, Paul K. Crane, Sebastien J. Haneuse, James Bowen, Wayne C. McCormick, Susan McCurry, and Eric B. Larson. 2015. "Characterization of Dementia and Alzheimer's Disease in an Older Population: Updated Incidence and Life Expectancy with and without Dementia." American Journal of Public Health, 105(2): 408-413.
Ujvari, Kathleen. 2018. Disrupting the Marketplace: The State of Private Long-Term Care Insurance, 2018 Update. Washington, DC: AARP Public Policy Institute. https://www.aarp.org/content/dam/aarp/ppi/2018/08/disrupting-the-marketplace-the-state-of-private-long-term-care-insurance.pdf.
U.S. Government Accountability Office (GAO; previously General Accounting Office). 1998. Alzheimer's Disease: Estimates of Prevalence in the United States. Report to the Secretary of Health and Human Services. GAO/HEHS-98-16. Washington, DC: U.S. Government Accountability Office.
Weden, Margaret M., Jeremy N.V. Miles, Esther Friedman, Jose Escarce, Christine Peterson, Kenneth M. Langa, and Regina A. Shih. 2017. "The Hispanic Paradox: Race/ethnicity and Nativity, Immigrant Enclave Residence and Cognitive Impairment among Older US Adults." J Am Geriatr Soc, 65(5): 1085-1091, doi:10.1111/jgs.14806.
Weuve, Jennifer, Kumar B. Rajan, Lisa L. Barnes, Robert S. Wilson, and Denis A. Evans. 2018. "Secular Trends in Cognitive Performance in Older Black and White U.S. Adults, 1993-2012: Findings from the Chicago Health and Aging Project." Journals of Gerontology: Social Sciences, 73(S1): S73-S81, doi:10.1093/geronb/gbx167.
White, Lindsay, Paul Fishman, Anirban Basu, Paul K. Crane, Eric B. Larson, and Norma B. Coe. 2019. "Medicare Expenditures Attributable to Dementia." Health Services Research, 54(4): 773-781.
Wilson, Robert S., Neelum T. Aggarwal, Lisa L. Barnes, Julia L. Bienias, Carlos F. Mendes de Leon, and Denis A. Evans. 2009. "Biracial Population Study of Mortality in Mild Cognitive Impairment and AD." Archives of Neurology, 66(6): 767-772, doi:10.1001/archneurol.2009.80.
Wilson, Robert S., David R. Weir, Sue E. Leurgans, Denis A. Evans, Liesi E. Hebert, Kenneth M. Langa, Brenda L. Plassman, Brent J. Small, and David A. Bennett. 2011. "Sources of Variability in Estimates of the Prevalence of Alzheimer's Disease in the United States." Alzheimer's & Dementia, 7(1): 74-79, doi:10.1016/j.jalz.2010.11.006.
Wolff, Jennifer L., Brenda C. Spillman, Vicki A. Freedman, and Judith D. Kasper. 2016. "A National Profile of Family and Unpaid Caregivers Who Assist Older Adults with Health Care Activities." JAMA Internal Medicine, 176(3): 372-79, doi:10.1001/jamainternmed.2015.7664.
Wu, Yu-Tzu, Alexa S. Beiser, Monique M.B. Breteler, Laura Fratiglioni, Catherine Helmer, Hugh C. Hendrie, Hiroyuki Honda, M. Arfan Ikram, Kenneth M. Langa, Antonio Lobo, Fiona E. Matthews, Tomoyuki Ohara, Karine Pérès, Chengxuan Qiu, Sudha Seshadri, Britt-Marie Sjölund, Ingmar Skoog, and Carol Brayne. "Trends in the Prevalence and Incidence of Dementia: A Review of Current Evidence." Nature Reviews Neurology, 13(6): 327-339.
Yang, Zhou, and Allan Levey. 2015. "Gender Differences: A Lifetime Analysis of the Economic Burden of Alzheimer's Disease." Women's Health Issues, 25(5): 436-40, http://www.whijournal.com/article/S1049-3867(15)00076-6/pdf.
Yang, Zhou, Kun Zhang, Pei-Jung Lin, Carolyn Clevenger, and Adam Atherly. 2012. "A Longitudinal Analysis of the Lifetime Cost of Dementia." Health Services Research, 47(4): 1660-1678.
Zhao, Qing-Fei, LanTan, Hui-Fu Wang, Teng Jiang, Meng-Shan Tan, LinTan, WeiXu, Jie-Qiong Li, JunWang Te-JenLai, and Jin-TaiYu. 2016. "The Prevalence of Neuropsychiatric Symptoms in Alzheimer's Disease: Systematic Review and Meta-Analysis." Journal of Affective Disorders, 190: 264-271, doi:10.1016/j.jad.2015.09.069.
Zhu, Carolyn W., Stephanie Cosentino, Katherine Ornstein, Yian Gu, Nikolaos Scarmeas, Howard Andrews, and Yaakov Stern. 2015. "Medicare Utilization and Expenditures around Incident Dementia in a Multiethnic Cohort." Journals of Gerontology A Biological Sciences Medical Sciences, 70(11): 1448-1453, doi:10.1093/gerona/glv124.
Zhu, Carolyn W., Nikolaos Scarmeas, Rebecca Torgan, Marilyn Albert, Jason Brandt, Deborah Blacker, Mary Sano, and Yaakov Stern. 2008. "Home Health and Informal Care Utilization and Costs over Time in Alzheimer's Disease." Home Health Care Services Quarterly, 27(1): 1-20.
Zissimopoulos, J., E. Crimmins, and P. St. Clair. 2014. "The Value of Delaying Alzheimer's Disease Onset." Forum for Health Economics and Policy, 18(1): 25-39. doi:10.1515/fhep-2014-0013.
Zissimopoulos, Julie M., Bryan C. Tysinger, Patricia A. St. Clair, and Eileen M. Crimmins. 2018. "The Impact of Changes in Population Health and Mortality on Future Prevalence of Alzheimer's Disease and Other Dementias in the United States." Journals of Gerontology: Social Sciences, 73(S1): S38-S47.
ABOUT THE AUTHORS
Melissa Favreault is a senior fellow in the Income and Benefits Policy Center at the Urban Institute, where her work focuses on the economic well-being and health status of older Americans and individuals with disabilities. She studies social insurance and social assistance programs and has written extensively about Medicaid, Medicare, Social Security, and Supplemental Security Income. She evaluates how well these programs serve Americans today and how various policy changes and on-going economic and demographic trends could alter outcomes for future generations. Much of her research relies on dynamic microsimulation, distributional models that she develops to highlight how educational and economic advantages shape financial outcomes, disability trajectories, health care needs, and longevity. She has a special interest in the economic risks that people face over their lives. Favreault earned her BA in political science and Russian from Amherst College, and her MA and PhD in sociology from Cornell University.
Richard W. Johnson is a senior fellow in the Income and Benefits Policy Center at the Urban Institute, where he directs the Program on Retirement Policy. His current research focuses on older Americans' employment and retirement decisions, LTSS for older adults with disabilities, and state and local pensions. Recent studies have examined job loss at older ages, occupational change after age 50, employment prospects for 50+ African Americans and Hispanics, and the impact of the 2007-2009 recession and its aftermath on older workers and future retirement incomes. He has also written extensively about retirement preparedness, including the financial and health risks people face as they approach retirement, economic hardship in the years before Social Security's early eligibility age, and the adequacy of the disability safety net.
NOTES
-
Loef and Walach (2013) provide a review of the literature on the relationship between obesity, body mass index, and cognitive decline.
-
The literature on diabetes and cognitive status is mixed and complex, but several meta-analyses suggest that diabetes increases the risk of some types of cognitive decline (Cheng et al. 2012; Cukierman, Gerstein, and Williamson 2005; Omorogieva and Brooke 2015). Dementia may also complicate diabetes management, creating a vicious circle (Omorogieva and Brooke 2015).
-
One recent meta-analysis estimated a prevalence of aggression of 40 percent among patients with Alzheimer's disease (Zhao et al. 2016). Aigbogun et al. (2019) describes the importance of other behavioral disturbances, including agitation and related symptoms.
-
This may include locked floors or wards and cameras to monitor public spaces and exits.
-
The most common form of mixed dementia, as it is sometimes called, is when Alzheimer's disease and vascular dementia occur together.
-
The Centers for Medicare & Medicaid Services (2015) estimates that in 2014 61.4 of United States nursing home residents were cognitively impaired, with 36.6 percent severely cognitively impaired and 24.8 percent mildly impaired (figure 3.11). Estimates that exclude those in institutions could thus misestimate the incidence, prevalence, and duration of cognitive impairment.
-
Brayne and Calloway (1988) provocatively suggest that dementia may be viewed as part of the continuum of the aging process.
-
This is consistent with meta-analyses (Steenlandet al. 2016).
-
Studies considering survival time with dementia sometimes use different concepts. Some, for example, compute survival time from the point at which symptoms were first reported. Others start from onset age, diagnosis age, or time of entry into a study (which may depend on one's diagnosis). Studies of remaining life expectancy with and without severe CI also examine expected duration, but they include those who never experience severe CI.
-
Deb et al. (2017) provides a review of many of the nuances in this literature.
-
Coe, Skira, and Larson (2018) point out some of the limitations of this type of valuation. Although such measures are surely superior to ignoring these costs, they do not account for how caregiving affects the caregiver's well-being.
-
Costs are assumed to vary by disease severity. The earlier paper (Alzheimer's Association 2015) uses 2008 MCBS data.
-
Like DYNASIM, the model uses many demographic variables and integrates lagged status and correlations between cognitive status and disability. A main distinction between DYNASIM and FEM is that the latter model focuses more on interrelationships between health variables and disability, and most especially chronic conditions. (DYNASIM includes these correlations, but focuses more on relationships between disability and income and wealth sources and the public policies that govern pre and post-tax and transfer income and wealth, so it is more a model of income support and less a model of disease.)
-
Some potential issues with long-running panel studies include declining representativeness of their sample because of attrition and failure to include new entrants to the population, such as immigrant families. Some studies that focus on a particular age group, like the HRS and NHATS, incorporate refresher cohorts so that the study can continue to represent accurately the population of the age group more broadly, though not necessarily at every wave.
-
By occurrence dependence, we refer to the fact that those who do not currently have disabilities but who have ever had a disability in the past are more likely to become disabled than those who have never had a disability; duration dependence refers to the fact that transition probabilities differ for those who have had disabilities for a longer time.
-
See, for example, Sun, Tan, and Yu (2014) or Sposato et al. (2015).
-
For an overview on measures of cognitive impairment in the HRS, see Fisher, Hassan, Faul, Rodgers, and Weir (2017).
-
Specifically, we assume that those whose proxies report that the person has poor memory, hallucinations, gets lost, and wanders can be classified as cognitively impaired. We tested the sensitivity of this measure to including those whose proxies report that the person cannot be left alone. For exit interviews, we generally follow the respondents' classification, but we do recode a small number of people who were classified as impaired in the prior HRS wave or who were nearly SCI in a prior wave and whose proxies report high numbers of limitations in IADLs.
-
See Hatfield et al. (2016), especially the technical appendix.
-
We assume that a small share of people with private long-term care insurance or higher incomes will purchase more expensive (presumably higher quality) care, while some people with lower incomes will purchase less expensive care.
-
We pool five years of DYNASIM data to reduce volatility in the estimates due to small cell sizes.
-
In this group, women's rates in DYNASIM are about 35 percent higher than in the NHATS, which on its face appears concerning. However, some caveats are appropriate. First, in both estimates, sample sizes are modest. Second, the women's rate in DYNASIM is still lower than the men's rate in NHATS, suggesting that it is not entirely unreasonable that we might observe this level of impairment in a large population.
-
In the projections presented in Hurd, Martorell, and Langa (2015), in contrast, age-specific prevalence does not always increase monotonically under assumptions about future decline in SCI based on rates of decline from Matthews et al. (2013).
-
Joling et al. (2020) provide recent evidence from the Netherlands.
-
Displaying this distribution of outcomes, including a small share of people who are severely cognitively impaired for a long time, is an important contribution of these projections. This so-called "tail" risk has contributed to the challenges that the private long-term care insurance industry now faces as the result of earlier policies that covered lifetime benefits. (Few newly issued policies provide lifetime benefits.)
-
Most studies report the replacement cost of unpaid care on an annual basis. When in 2020 dollars, most of the annual estimates are in the range of roughly $35,000-$42,000 (Langa et al. 2001, Zissimopoulos, Crimmins, and St. Clair 2014, Hurd et al. 2013). Our annual estimates (not reported) are broadly comparable, but sensitive to how we treat people who report providing round-the-clock care.
-
Here, we classify disability as "significant" if it meets the criteria set forward in the Health Insurance Portability and Accountability Act of 1996. See note 30 for additional detail.
Improving Health and Long-Term Care Modeling Capacity
This report was prepared under contract #HHSP233201600024I between HHS's ASPE/BHDAP and the Urban Institute. For additional information about this subject, you can visit the BHDAP home page at https://aspe.hhs.gov/bhdap or contact the ASPE Project Officers, at HHS/ASPE/BHDAP, Room 424E, H.H. Humphrey Building, 200 Independence Avenue, S.W., Washington, D.C., 20201; Judith.Dey@hhs.gov, Lauren.Anderson@hhs.gov, Helen.Lamont@hhs.gov.
Reports Available
Economic Hardship and Medicaid Enrollment in Later Life: Assessing the Impact of Disability, Health, and Marital Status Shocks
- HTML version: https://aspe.hhs.gov/basic-report/economic-hardship-and-medicaid-enrollment-later-life-assessing-impact-disability-health-and-marital-status-shocks
- PDF version: https://aspe.hhs.gov/pdf-report/economic-hardship-and-medicaid-enrollment-later-life-assessing-impact-disability-health-and-marital-status-shocks
Extended LTSS Utilization Makes Older Adults More Reliant on Medicaid Issue Brief
- HTML version: https://aspe.hhs.gov/basic-report/extended-ltss-utilization-makes-older-adults-more-reliant-medicaid-issue-brief
- PDF version: https://aspe.hhs.gov/pdf-report/extended-ltss-utilization-makes-older-adults-more-reliant-medicaid-issue-brief
Most Older Adults Are Likely to Need and Use Long-Term Services and Supports Issue Brief
- HTML version: https://aspe.hhs.gov/basic-report/most-older-adults-are-likely-need-and-use-long-term-services-and-supports-issue-brief
- PDF version: https://aspe.hhs.gov/pdf-report/most-older-adults-are-likely-need-and-use-long-term-services-and-supports-issue-brief
Risk of Economic Hardship Among Older Adults Issue Brief
- HTML version: https://aspe.hhs.gov/basic-report/risk-economic-hardship-among-older-adults-issue-brief
- PDF version: https://aspe.hhs.gov/pdf-report/risk-economic-hardship-among-older-adults-issue-brief
The Risk and Costs of Severe Cognitive Impairment at Older Ages: Literature Review and Projection Analyses
- HTML version: https://aspe.hhs.gov/basic-report/risk-and-costs-severe-cognitive-impairment-older-ages-literature-review-and-projection-analyses
- PDF version: goto
Long-Term Services and Supports for Older Americans: Risks and Financing, 2020 Research Brief
- HTML version: https://aspe.hhs.gov/basic-report/long-term-services-and-supports-older-americans-risks-and-financing-2020-research-brief
- PDF version: https://aspe.hhs.gov/pdf-report/long-term-services-and-supports-older-americans-risks-and-financing-2020-research-brief
Projections of Risk of Needing Long-Term Services and Supports at Ages 65 and Older
- HTML version: https://aspe.hhs.gov/basic-report/projections-risk-needing-long-term-services-and-supports-ages-65-and-older
- PDF version: https://aspe.hhs.gov/pdf-report/projections-risk-needing-long-term-services-and-supports-ages-65-and-older
The Risk and Costs of Severe Cognitive Impairment at Older Ages: Key Findings from our Literature Review and Projection Analyses Research Brief
- HTML version: https://aspe.hhs.gov/basic-report/risk-and-costs-severe-cognitive-impairment-older-ages-key-findings-our-literature-review-and-projection-analyses-research-brief
- PDF version: https://aspe.hhs.gov/pdf-report/risk-and-costs-severe-cognitive-impairment-older-ages-key-findings-our-literature-review-and-projection-analyses-research-brief
