Monday, July 29, 2019
Printer Friendly Version in PDF Format (8 PDF pages)
Alzheimer's Disease and Related Dementias Research Update
Richard J. Hodes, M.D.
Director, NIA
NIH Research Implementation Milestones are Based on Broad Input
- 2011: The National Alzheimer's Project Act (NAPA) was signed into law in January -- updated annually
- 2012: AD Summit
- 2013: ADRD Summit & Down syndrome mtg
- 2015: AD Summit
- 2016: ADRD Summit
- 2017*: AD Care/Services Summit
- 2018*: AD Summit
- 2019: ADRD Summit
- 2020: AD Care/Services Summit
* Newest sources of milestone input
https://www.nia.nih.gov/research/milestones
Combined External and Internal Input -- FY21 AD/ADRD Bypass Budget
- Input at 2012-2019 meetings:
- Academic research community
- Industry
- Non-governmental organizations
- Other Input:
- 2017 Request for Information
- Development of comprehensive milestones (NIH staff) -- based on summit and other input
- Trans-NIH (including NINDS) staff discussion; milestones edited to ensure comprehensive inclusion of priorities for FY21
- NIH staff "price" the milestones
- Final budget estimate for FY21
Bypass Budget Language
SEC. 230. Hereafter, for each fiscal year through fiscal year 2025, the Director of the National Institutes of Health shall prepare and submit directly to the President for review and transmittal to Congress, after reasonable opportunity for comment, but without change, by the Secretary of Health and Human Services and the Advisory Council on Alzheimer's Research, Care, and Services, an annual budget estimate (including an estimate of the number and type of personnel needs for the Institutes) for the initiatives of the National Institutes of Health pursuant to the National Alzheimer's Plan, as required under section 2(d)(2) of Public Law 111-375.
Trans-NIH Input for FY21 ADBB
- 18 Institutes/Centers/Offices contributed to the development of this year's professional judgment budget:
- NIA
- NEI
- NHLBI
- NIAAA
- NIAMS
- NIBIB
- NICHD
- NIDA
- NIDCD
- NINR
- NIDCR
- NIDDK
- NIEHS
- NIMH
- NIMHD
- NINDS
- FIC
- NIH Office of AIDS Research
Using CADRO as a Framework
- The eight CADRO (Common Alzheimer's Disease Research Ontology) categories provide the overarching framework for the FY21 AD Bypass Budget and narrative.
- CADRO provides the framework for IADRP (the International Alzheimer's Disease Research Portfolio) and will allow tracking of implementation in the budget areas in future years.
Professional Judgment Budget FY 2021, Projected Costs and Additional Resources Needed
| Molecular Pathogenesis and Pathophysiology of Alzheimer's Disease | $57,500,000 |
|---|---|
| Diagnosis, Assessment, and Disease Monitoring | $20,000,000 |
| Translational Research and Clinical Interventions | $139,550,000 |
| Epidemiology | $55,000,000 |
| Care and Caregiver Support | $34,550,000 |
| Research Resources | $84,500,000 |
| Alzheimer's Disease-Related Dementias | $43,000,000 |
| Staffing Needs, Support, and Miscellaneous | $3,811,444 |
| Projected Costs for New AD/ADRD Research | $437,911,444 |
| Less Funding from Prior Appropriations that is Available for New AD/ADRD Research | ($84,000,000) |
| Additional FY 2021 Resources Needed for New AD/ADRD Research | $353,911,444 |
Distribution of FY 2021 Projected Costs Across Research Areas
| Total Projected Costs: $437,911,444* Additional Resources Needed for New Research: $353,911,444 |
|---|
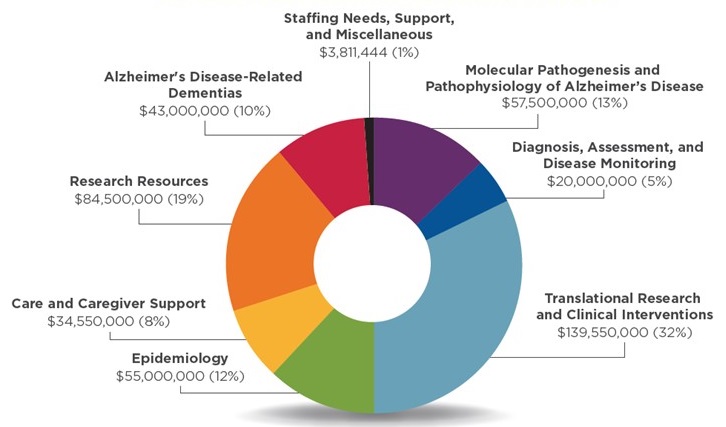 |
| * In FY 2021, the projected costs of resources needed for new research to enhance investigator-initiated research grants and initiatives to meet the 2025 treatment/prevention goal is $438 million. This estimate will be reduced by $84 million in funding that is projected to become available after completion of previously funded AD/ADRD research initiatives. As a result, the additional resources needed for new research in the FY 2021 budget is $354 million. |
Total Resource Needs, FY21
| FY 2020 President's Budget Request for AD/ADRD Research (baseline estimate) | $2,142,000,000 |
|---|---|
| Difference Between FY 2020 President's Budget Request and FY 2019 Appropriation for AD/ADRD Research | $326,000,000 |
| Additional FY 2021 Resources Needed for New AD/ADRD Research | $353,911,444 |
| TOTAL FY 2021 Resources Needed for AD/ADRD Research | $2,821,911,444 |
Timing of ADBB Releases and AD/ADRD Appropriations
- FY17 AD/ADRD milestones - $323M extra needed
- FY17 ADBB Released July 2015
- $350M in FY16 supports acceleration of FY17 milestones
- FY17 ADBB Released July 2015
- FY18 AD/ADRD milestones - $414M extra needed
- FY18 ADBB Released August 2016
- $400M in FY17 supports acceleration of FY18 milestones
- FY18 ADBB Released August 2016
- FY19 AD/ADRD milestones - $597M extra needed
- FY19 ADBB Released July 2017
- $414M in FY18 supports acceleration of FY19 milestones
- FY19 ADBB Released July 2017
- FY20 AD/ADRD milestones - $478M extra needed
- FY20 ADBB Released July 2018
- $425M in FY19 supports acceleration of FY20 milestones
- FY20 ADBB Released July 2018
- FY21 AD/ADRD milestones - $354M extra needed
AD/ADRD Progress Report -- An important part of the ADBB
- The AD/ADRD Bypass Budget narrative includes many examples of recent science advances across multiple research topics -- basic to clinical, care/caregiving, etc.
- https://www.nia.nih.gov/about/together-we-succeed-accelerating-research-on-alzheimers-disease-and-related-dementias
Implementation Tracking
- IADRP: http://iadrp.nia.nih.gov/t
- Will continue to offer detailed tracking of awards under the CADRO categories
- Web-based tool for tracking funding initiatives and activities: https://www.nia.nih.gov/research/milestones
- Newly revised
- Aimed at addressing the research milestones associated with NAPA
Complex Considerations for this and Future Bypass Budg
- Past/current appropriations, including funds that have allowed us to address milestones "in advance"
- Progress on milestones -- what have we accomplished? what new milestones are emerging?
- Funds that are becoming available each year as prior awards end
- Incorporating new milestones, including focus on care/services, target identification and drug development
- Emerging investments by other groups -- what gaps are best filled by the NIA/NIH?
- Successes in recruiting new and established researchers to AD/ADRD research and capacity of the field
Save the Date!
Research Summit on Dementia Care
The 2020 National Research Summit on Care, Services, and Supports for Persons with Dementia and Their Caregivers
March 24-25, 2020
Natcher Conference Center
NIH Main Campus, Bethesda, MD
Current List of Steering Committee Members
- Jennifer Wolff, Johns Hopkins University, Steering Committee Co-chair
- David Reuben, UCLA, Steering Committee Co-chair
- Susan Beane, Healthfirst
- Malaz Boustani, Indiana University School of Medicine
- Chris Callahan, Indiana University School of Medicine
- Elena Fazio, NIA
- Jason Karlawish, University of Pennsylvania
- Ian Kremer, LEAD Coalition
- Helen Lamont, HHS ASPE
- Katie Maslow, GSA
- Michael Monson, Centene Corporation
- Vincent Mor, Brown University
- Joanne Pike, Alzheimer's Association
- Robyn Stone, LeadingAge
- Sheryl Zimmerman, University of North Carolina
- Pending invitations remain, including:
- Person Living with Dementia
- Caregiver to a Person Living with Dementia
