Monday, July 29, 2019
Printer Friendly Version in PDF Format (8 PDF pages)
NAPA Clinical Subcommittee Federal Updates
Shari Ling, MD
Request for Information -- Patients Over Paperwork
- Open for Comment until August 12, 2019
- In June CMS issued a RFI on Reducing Administrative Burden to Put Patients over Paperwork that invites individuals, families and caregivers, the medical community, and other health care stakeholders to recommend further changes to rules, policies, and procedures that would shift more of clinicians' time and the health care system's resources from paperwork to high-quality care that improves health
- The RFI requests comment on areas including:
- Reporting and documentation requirements
- Coding and documentation requirements for Medicare or Medicaid payment
- Prior authorization procedures
- Policies and requirements for rural providers, clinicians, and beneficiaries
- Policies and requirements for dually enrolled (i.e., Medicare and Medicaid) beneficiaries
- Beneficiary enrollment and eligibility determination
- CMS processes for issuing regulations and policies
- Information at: https://www.federalregister.gov/documents/2019/06/11/2019-12215/request-for-information-reducing-administrative-burden-to-put-patients-over-paperwork
(Goal 2)
Programs for All-Inclusive Care for the Elderly Modernization
- In June CMS finalized a rule to update and modernize PACE requirements
- The PACE program provides comprehensive medical and social services to certain frail, elderly individuals who qualify for nursing home care but, at the time of enrollment, can still live safely in the community
- Policies finalized in the rule reflect the latest standards for PACE participants -- many of whom are “dually eligible” for both Medicare and Medicaid -- and will strengthen beneficiary protections, improve care coordination, and provide administrative flexibilities and regulatory relief for PACE organizations
- To view the final rule (CMS-4168) visit: https://www.federalregister.gov/documents/2019/06/03/2019-11087/medicare-and-medicaid-programs-programs-of-all-inclusive-care-for-the-elderly
(Strategy 2.0)
Physical and Mental Health (PMH) Integrated Care Needs and Adult Beneficiaries with Complex Care Needs (BCN) -- New Medicaid Quality Measures
- On July 10th, CMS's Medicaid Innovation Accelerator Program hosted a webinar to introduce several new measures created for voluntary use for state Medicaid programs
- One is a measure endorsed by the National Quality Forum: Follow-up care for adult Medicaid beneficiaries who are newly prescribed an antipsychotic medication, NQF #3313
- The measure is not a dementia measure -- it assesses whether Medicaid beneficiaries who are newly prescribed an antipsychotic medication receive follow-up care within four weeks
- This measure is available for states' use and is not required in any CMS reporting programs
- Additional information will be available later this summer at: https://www.medicaid.gov/state-resource-center/innovation-accelerator-program/index.html
(Strategy 2.D)
Independence at Home (IAH) 4th Year Evaluation Results
- IAH saved Medicare approximately $33 million in Year 4, or about $384 per enrolled beneficiary per month (PBPM). This follows savings from Years 1-3 that, when combined with Year 4, amount to about $63 million in savings and high-quality care for Medicare
- Results attest to the value of home-based primary care in serving complex, frail older adults, with an emphasis on a high-touch and high-tech mobile team
- Year 4 savings from the demonstration resulted from reduced hospital utilization and nursing home services; the former reduced 40% of PBPM savings
- Over a 4-year period IAH has led to over 1,800 fewer hospital admissions, over 3,400 fewer emergency department visits, and more than 400 fewer unplanned readmissions
- The effects of the demonstration on expenditures and hospital care use in years 1-4 were similar for beneficiaries with dementia and beneficiaries without dementia.
- Information at: https://innovation.cms.gov/initiatives/independence-at-home/
(Strategy 2.E)
Primary Care First Model Options
- In April CMS announced Primary Care First Model Options, a voluntary five-year payment option that rewards value and quality by offering an innovative payment structure to support delivery of advanced primary care
- The model is based on prioritizing the doctor-patient relationship; enhancing care for beneficiaries with complex chronic needs and high need seriously ill patients, reducing administrative burden, and focusing financial rewards on improved health outcomes
- In 2020, PCF will include 26 diverse regions for certain practices not currently participating in the CPC+ Model
- The model has elements specifically designed to support practices caring for patients with complex chronic needs or serious illness (Serious Illness Population)
- CMS hosted a webinar on July 24 to help the public learn more about the SIP population
- Information at: https://innovation.cms.gov/initiatives/primary-care-first-model-options/
(Strategy 2.E)
Nursing Home Arbitration Agreements and New Regulation
- In July CMS released a Notice of Proposed Rulemaking and a Final Regulation to help assure safety and quality in nursing homes
- The rules are components of CMS's five part approach to ensuring a high-quality long term care facility system, which includes strengthening requirements in nursing homes, working with states to enforce statutory and regulatory requirements, increasing transparency of nursing home performance, improving quality, and putting resident safety first by removing unnecessary burdens on providers
- In addition to protecting residents and reducing burdens, the proposed rule will help nursing homes further focus their resources on their residents by saving them $616 million in administrative costs annually that can be reinvested in care
- The CMS final rule supports residents and their families and caregivers by removing the ban on binding arbitration agreements while requiring nursing homes to ensure residents have the ability to choose the method of dispute resolution they prefer
- Information at: https://www.cms.gov/newsroom/fact-sheets/cms-issues-proposed-revision-requirements-long-term-care-facilities-arbitration-agreements and https://www.federalregister.gov/documents/2019/07/18/2019-14946/medicare-and-medicaid-programs-requirements-for-long-term-care-facilities-regulatory-provisions-to
(Strategy 2.F)
IMPACT Act Update - Interoperable Data for Post-Acute Care
- CMS is committed to addressing care plans across certain Medicare post acute settings to address problems like communication across multiple providers, poor patient recall during periods of stress, and increased cost and provider burden
- The IMPACT Act requires standardized, interoperable data in most Medicare settings, which facilitates care coordination
- Post-acute providers are required to submit assessment data to CMS for patients at admission and discharge for payment, survey purposes, public reports, and quality measurement purposes
- PAC assessment data includes administrative information (e.g. name, DOB, reason for admission), clinical information (e.g. diagnosis, cognitive status), and functional items (e.g. cognitive and physical function, co-morbidities)
- Information at: https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/Post-Acute-Care-Quality-Initiatives/IMPACT-Act-of-2014/Spotlights-and-Announcements-.html
(Strategy 2.F)
National Partnership to Improve Dementia Care in Nursing Homes: Update
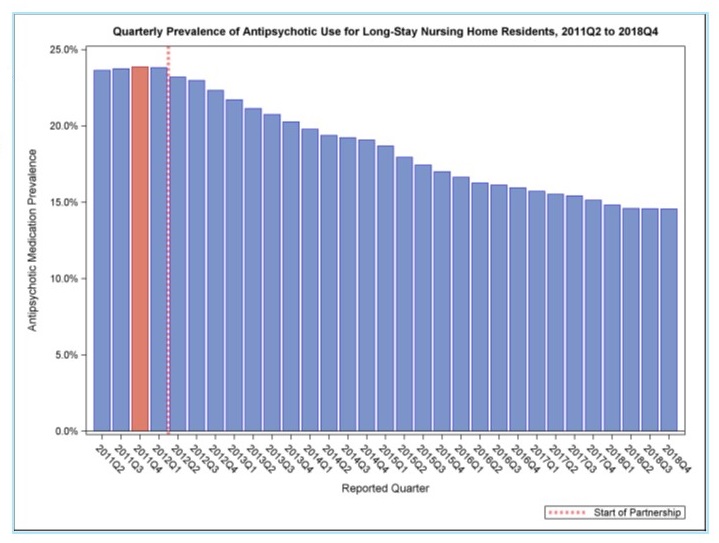 |
| Decrease of 39 percent to a national prevalence of 14.6 percent in Quarter 4, 2018 Information at: https://www.medicare.gov/nursinghomecompare/ (Strategy 3.D) |
Medicare Part D Prescriber Utilization and Payment Public Use Files (PUF)
- The Medicare Part D Prescriber PUF provides information on the specific prescription drugs prescribed by individual providers paid for under Part D
- The PUF is based on information from Prescription Drug Event records submitted by Medicare Advantage Prescription Drug) plans and by stand-alone Prescription Drug Plans
- It identifies providers by their National Provider Identifier and lists the specific prescriptions they prescribed by brand name (if applicable) and generic name
- For each prescriber and drug, the PUF also includes aggregated cost, utilization, and summarized beneficiary information, and variables that identify specific categories of drugs
- Information at: https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/Medicare-Provider-Charge-Data/Part-D-Prescriber.html
(Strategy 5.A)
Co-morbidity in Chronic Conditions including ADRD
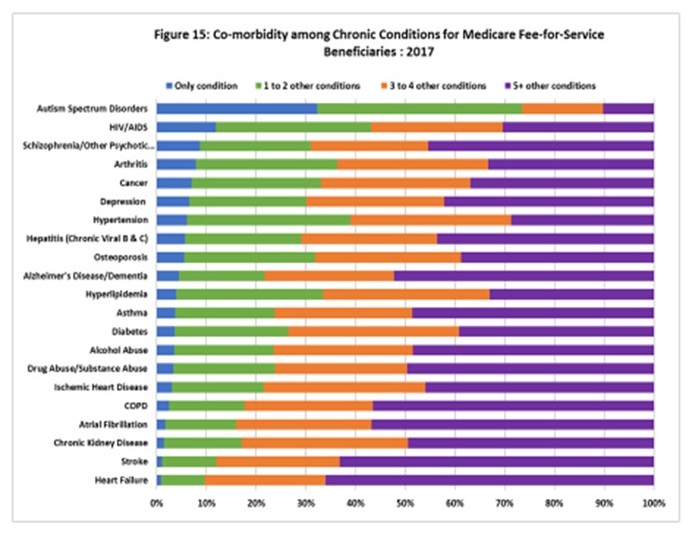 |
| https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/Chronic-Conditions/index.html |
New Continuing Education Course: Brain Health
- Sections:
- Section 1: Descriptions & Risk Fators
- Section 2: Risk Reduction & Management I
- Section 3: Risk Reduction & Management II
- Section 4: Resources & References
- Learning Outcomes:
- Explain-review brain health terminology
- Define and describe risk factors associated with age related cognitive decline, mild cognitive impairment, and dementia
- Summarize lifestyle medicine management strategies, including hypertension management, that may decrease risk
- Continuing Education:
- 1.0 CME/MOC education activity is available through ACPM's learning platform https://www.acpm.org/learn
- Cost:
- Learners can create a free login account and take the course at no cost
https://www.acpm.org/page/brainhealthcourse
New Brain Health Resources
American College of Preventive Medicine
- For your convenience, ACPM provides this collection of resources for physicians, health care professionals, and their patient populations. Materials include those developed by ACPM, its partners, and leading advocates of brain health.
- Research
- Emerging research
- Epidemiological information
- Provider Tools
- Best practices and guidelines
- Screening and diagnostic tools
- Risk factor treatment strategies
- Evidence-based community programs
- Referral strategies
- Insurance coverage
- Provider reimbursement
- Patient Materials
- Patient handouts
Brain Health Resources https://www.acpm.org/page/brainhealth
AHRQ EPC Systematic Reviews
- Diagnosis and Treatment of Clinical Alzheimer's-type Dementia (CATD): A Systematic Review
- Purpose: to summarize the evidence on:
- the accuracy of cognitive tests for identifying clinical Alzheimer's-type dementia (CATD) in individuals with suspected CATD
- the accuracy of biomarkers for identifying Alzheimer's disease (AD) in individuals with dementia
- the benefits and harms of prescription drugs and supplements for cognition, function, and behavioral and psychological symptoms of dementia (BPSD) in patients with CATD.
- Developed to inform guidelines in partnership with AAFP
- Final Report will be posted fall 2019.
- Protocol posted at: https://effectivehealthcare.ahrq.gov/topics/alzheimers-type-dementia/protocol
- Purpose: to summarize the evidence on:
- Care Interventions for People With Dementia (PWD) and their Caregivers
- Purpose: to understand the evidence base for effective dementia care interventions for persons with dementia and their caregivers, and to assess the potential for implementation of that evidence.
- Sponsored by National Institute on Aging (NIA)
- Draft Report will be posted for public comment spring 2020, and Final Report, summer 2020
- Protocol posted at: https://effectivehealthcare.ahrq.gov/topics/care-interventions-pwd/protocol
