Friday, July 30, 2018
Printer Friendly Version in PDF Format (9 PDF pages)
How Prepared is the U.S. Health Care System for a Future Alzheimer's Treatment?
Jodi Liu, Jakub Hlavka, Richard Hillestad, Soeren Mattke
RAND Health
Why focus on Alzheimer's treatment now?
- Alzheimer's dementia affects over 5 million Americans today
- Guarded optimism for Alzheimer's disease-modifying therapies in development
- Treatment paradigm has shifted to early stage disease
- Estimated 13.8 million Americans with mild cognitive impairment
- Objective: Quantify the potential mismatch between supply and demand for the delivery of a future Alzheimer's treatment
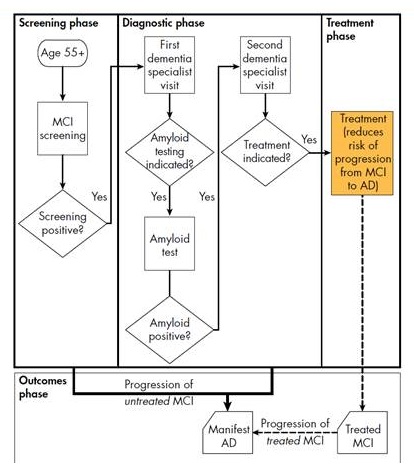
Alzheimer's disease progression and clinical pathway
- No cognitive impairment
- Mild cognitive impairment (MCI)
- Assume treatment reduces the risk of transitioning from MCI to dementia
- Alzheimer's dementia
Model assumptions and framework
- Key assumptions:
- Disease-modifying therapy for early-stage Alzheimer's disease available in 2020
- Therapy delivered by intravenous infusion every 4 weeks for a year
- Treatment reduces relative risk of progression from MCI to Alzheimer's dementia by 50%
- Dementia specialists: neurologists, geriatricians, geriatric psychiatrists
- Uptake rates based on expert input
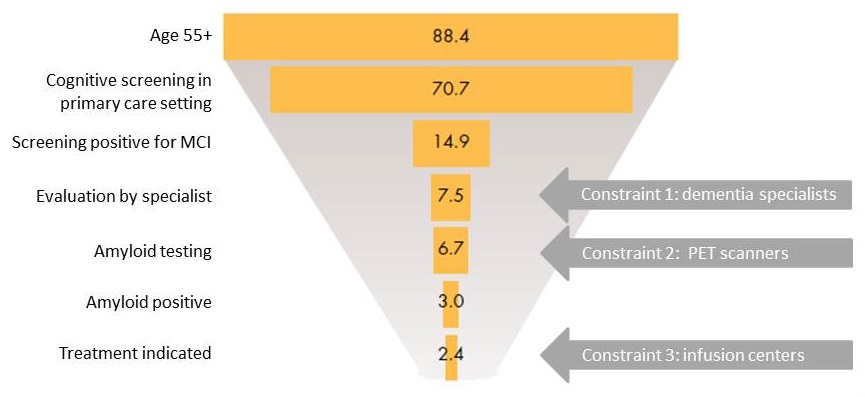
Snapshot of potential patients in 2019 (millions)
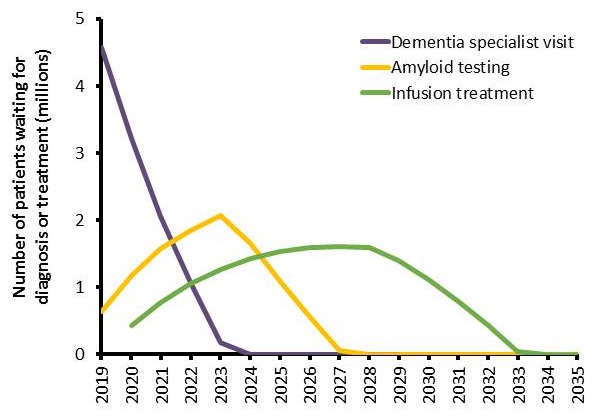
Millions of patients projected to be on waitlists
- Initial bottleneck for specialist visits with 4.6 million patients
- With treatment first available in 2020, wait lists expected until 2034
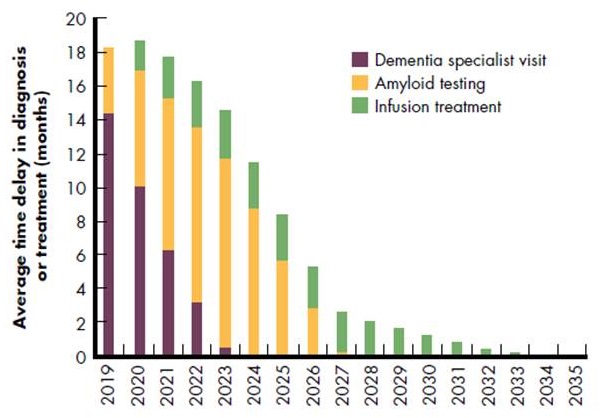
Projected wait times are extensive
- Average 14-month wait for specialists in 2019
- Average 11-month wait for testing in 2023 Infusion waits until 2034
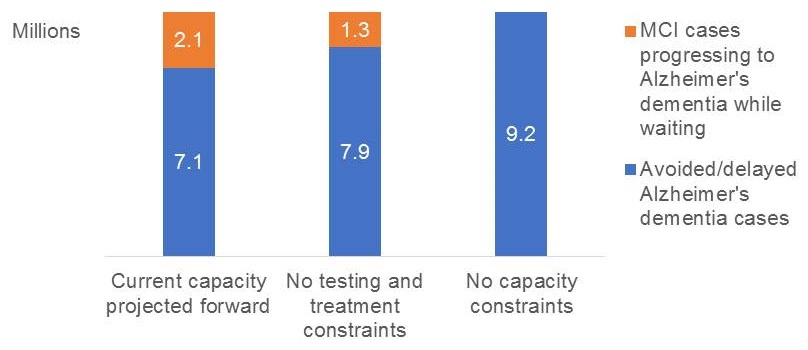
- 2.1 million MCI cases could progress to Alzheimer's dementia while on wait lists
Specialist shortage is most urgent issue
- Binding constraint and unlikely to resolve
- Improve productivity
- More specific secondary screening test for MCI reduce number of false positives or to prioritize based on risk
- Task shifting during evaluation process
- Qualifying providers from other specialties
- Train physicians in larger specialties (internal medicine, general psychiatry) and potentially mid-level providers in dementia care
- Use telemedicine to facilitate access to dementia care specialists by primary care providers
Range of diagnostic options could be expanded
- Expanding capacity for PET scans technically feasible but probably inefficient
- High fixed and variable cost
- Insufficient volume for cyclotrons in rural areas
- Building up capacity to handle prevalent cases when treatment is first approved would lead to idle capacity later
- Use of CSF assay is a possible solution
- Samples can be obtained in most clinics and sent to central labs
- Less expensive per test
- Blood and retinal tests in development
Access to amyloid PET limited by geographic gaps in cyclotrons that manufacture tracers
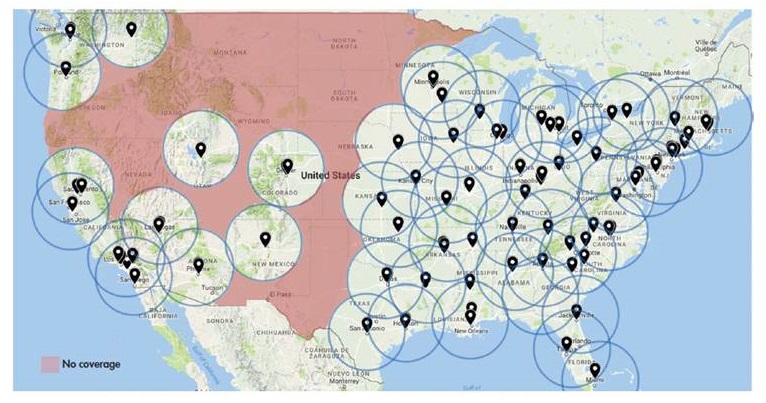 |
SOURCE: Society of Nuclear Medicine and Molecular Imaging as of October 6, 2017 (undated); Cardinal Health (2017).
RAND RR2272-3 |
Home infusions could play an important role
- Expected prevalent cases imply the need to triple existing infusion chair capacity
- Increase would likely lead to idle capacity later
- Home infusion delivery could increase capacity in the short run without fixed infrastructure
- Covered for patients in traditional Medicare starting 2020
- Already covered by many Medicare Advantage plans
- Nature of treatment could allow home infusion
- Short duration, cognitively intact patients, rare acute reactions
What are the implications for a research agenda?
- Development of better screening tools to reduce the large number of patients entering care pathway probably has high ROI
- "rescoring" of MMSE
- Better tests for primary or secondary screening or risk stratification
- Non-invasive biomarkers
- Demonstration projects for scalable delivery models that leverage specialist time better are needed
- Will also help to address regional access issues
- Capacity constraints on diagnostic testing and infusion delivery are likely to be addressed with current efforts
- Assuming adequate reimbursement
Summary
- Disease-modifying AD therapy would be a breakthrough
- Simulation suggests that US healthcare system is ill-prepared to deliver a therapy to the large number of prevalent cases
- As many as 2.1 million patients might develop AD because of delays in access to care under current capacity assumptions
- Increasing capacity to deliver a potential therapy would involve payment policy, regulatory requirements, workforce considerations, and capacity planning
Thank you
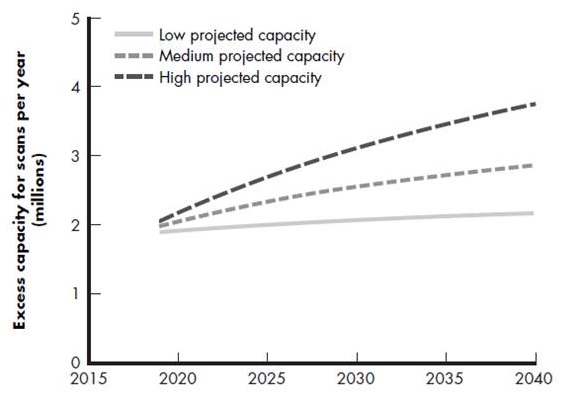
PET scan capacity
- Versus demand for 6.7 million scans in 2019 (plus geographic gaps)
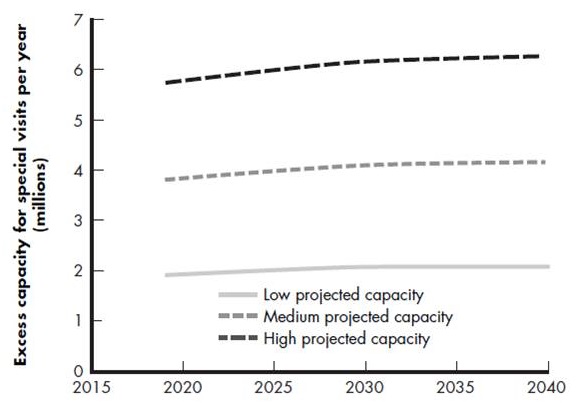
Dementia specialist workforce capacity
- Versus demand for 7.5 million evaluation visits and 3 million indication visits in 2019
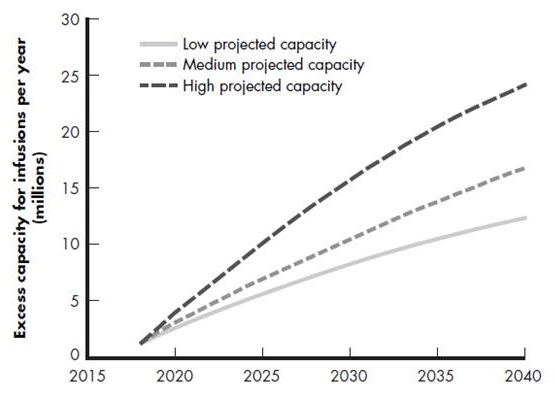
Infusion capacity
- Versus demand for 33 million infusions in 2020
MCI progresses to dementia while patients wait, 2020-2040