Monday, January 28, 2019
Printer Friendly Version in PDF Format (8 PDF pages)
Testing a Dementia Care Intervention for Widespread Adoption: The COPE CT Study
Richard H. Fortinsky, PhD
Professor and Health Net, Inc. Endowed Chair in Geriatrics and Gerontology
UConn Center on Aging, University of Connecticut School of Medicine
Farmington, Connecticut
Disclosures and Acknowledgements
- No financial conflicts of interest to disclose.
- COPE CT study funded by the National Institute on Aging (R01 AG044504)
Presentation Outline
- COPE intervention overview
- COPE CT study pragmatic trial elements
- Selected findings and next steps COPE adoption potential in LTSS and health systems
COPE Intervention Phases
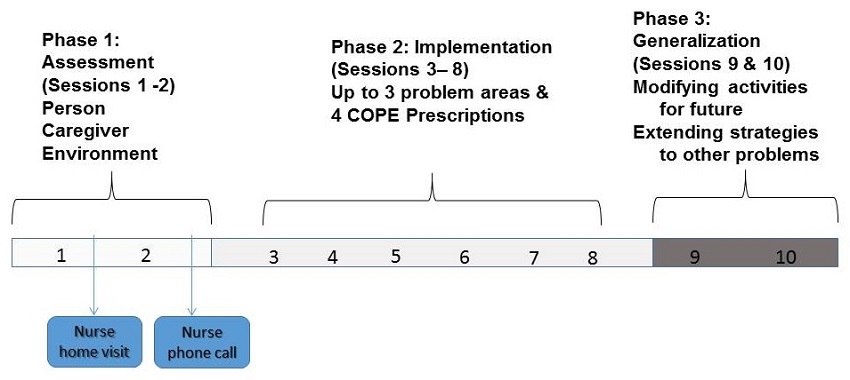 |
| Adapted from Gitlin LN et al, JAMA, 2010;304:983-991 |
Pragmatic Trial Elements of COPE CT Study Design
- Effectiveness-implementation hybrid design1
- Engages key stakeholders in decisions about study design and evaluation
- Investigators planned study with care management organization partner and Connecticut Medicaid program staff.
- Source of data : care management organization's electronic data base
- Care managers verified clients generated by electronic search
- Care managers introduced study and invited participation as part of routine monthly telephone client monitoring process.
- Achieved study enrollment goal as a result of this design process.
- 291 persons with dementia and caregivers (dyads) enrolled and randomized
- Curran GM, et al. Medical Care 2012; 50:217-226
COPE Nurse Component Selected Findings
- Of 83 persons with dementia who provided urine sample, 14% had abnormal laboratory results suggesting new medical conditions.
- Of 72 persons with dementia who provided blood, 38% had abnormal results suggesting new medical conditions.
- Renal insufficiency/dehydration and anemia most common conditions.
- COPE nurse or caregiver contacted physicians of persons with dementia to report abnormal laboratory findings.
COPE OT Component Selected Findings:Most Common Caregiver-identified Problems
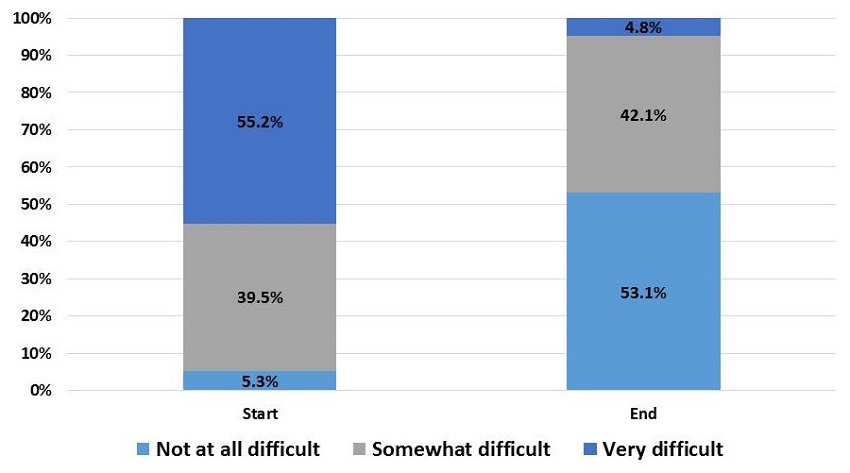
Level of Difficulty Managing Caregiver-identified Problems, Start and End of COPE Intervention
Level of Resolution of Caregiver-identified Problems at End of COPE Intervention
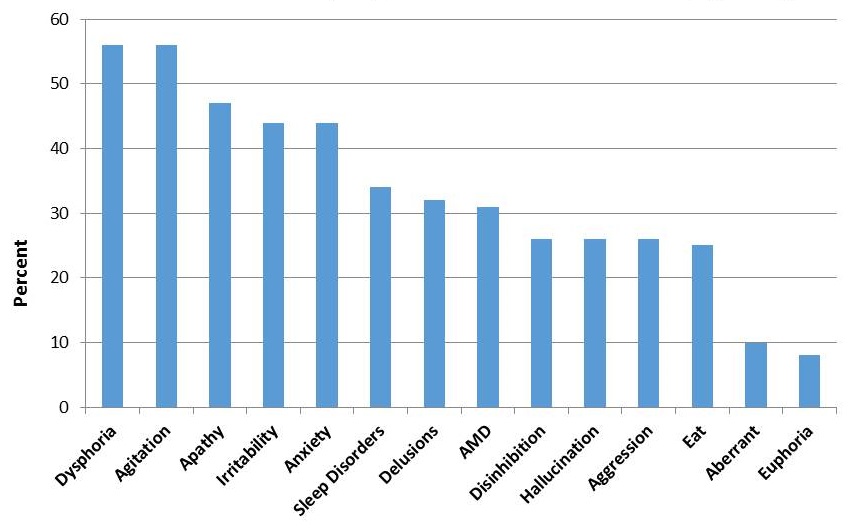
Prevalence of Neuropsychiatric Symptoms in Persons with Dementia, based on Neuropsychiatric Inventory (NPI)
COPE Effects on Neuropsychiatric Symptoms in Persons with Dementia
- Strong trend (p=0.08) in NPI total score in favor of COPE treatment group
- NPI mean score decreased in the COPE group over 4 months, no change in NPI mean score over 4 months in usual care group.
- Subgroups in which COPE had statistically significant effect on NPI score
- Medicaid-eligible persons with dementia (p=0.002)
- Dyads who lived apart (p=0.016)
- Person with dementia had higher baseline cognitive status (p=0.03)
Results: COPE Effects on Outcomes for Caregivers
- Statistically significant effect of COPE intervention on caregiver perceived well-being (p<0.001); mean score increased over 4 months in COPE group, did not change in usual care group.
- COPE showed a trend toward increasing caregiver confidence using activities (p=0.18); confidence scores increased over 4 months in both groups and in the COPE group this increase was greater.
- Subgroup analyses planned.
Next Steps in COPE CT Study
- 12 month post-randomization data will be analyzed to examine sustained effects of COPE.
- Cost-related data being collected and analyzed to determine net financial benefit of COPE.
- COPE intervention costs
- Use of health services during study period
- Willingness to pay for COPE
COPE Adoption Potential
- Medicaid-funded long-term services and supports (LTSS) such as Waiver programs ideal settings for COPE adoption.
- Medicare Advantage plans growing rapidly; many enrolling dually eligible adults, another potential reimbursement system to facilitate adoption.
- Health systems with primary care components ripe settings for adoption given growth of Patient-Centered Medical Homes & Accountable Care Organizations, with Medicare fee-for-service shared savings opportunities.
- Private pay market also possible.
COPE CT Study Investigators and Staff
- Principal Investigator:
- Richard H. Fortinsky, PhD, Center on Aging , University of Connecticut (UConn) School of Medicine
- Co-Investigators:
- Laura Gitlin, PhD, College of Nursing and Health Professions, Drexel University
- James Grady, ScD, Department of Community Medicine & Health Care, UConn School of Medicine
- Sheila Molony, PhD, APRN, GNP-BC, School of Nursing, Quinnipiac University
- Catherine Piersol, PhD, OTR/L, FAOTA, Jefferson College of Health Professions
- Laura Pizzi, PharmD, MPH, School of Pharmacy, Rutgers University
- Julie Robison, PhD, Center on Aging, UConn School of Medicine
- Project Staff at UConn Center on Aging:
- Heather McAbee-Sevick, MS, Study Coordinator
- Christine Bailey, MA, Screener & Interviewer
- Lisa Kenyon-Pesce, MPH, Recruitment Manager
- Azucena Minaya, MA, Screener & Interviewer
- Deborah Noujaim, MPH, Screener & Interviewer
- Dorothy Wakefield, MS, Data Manager & Analyst