Monday, January 28, 2019
Printer Friendly Version in PDF Format (11 PDF pages)
Alzheimer's Disease and Related Dementias Research Update
Richard Hodes, M.D.
Director, NIA
NIH Research Implementation Milestones drive progress and are Based on Broad Input
- 2011
- The National Alzheimer's Project Act (NAPA) was signed into law in January -- updated annually
- 2012
- AD Summit
- 2013
- ADRD Summit + Down syndrome meeting
- 2015
- AD Summit
- 2016
- ADRD Summit
- 2017
- AD Care/Services Summit -- Newest sources of milestone input
- 2018
- AD Summit -- Newest sources of milestone input
- 2019
- ADRD Summit
- 2020
- AD Care/Services Summit
https://www.nia.nih.gov/alzheimers/milestones
Timing of ADBB Releases and AD/ADRD Appropriations
- FY17 AD/ADRD milestones -- $323M extra needed
- FY17 ADBB Released July 2015
- $350M in FY16 supports acceleration of FY17 milestones
- FY17 ADBB Released July 2015
- FY18 AD/ADRD milestones - $414M extra needed
- FY18 ADBB Released August 2016
- $400M in FY17 supports acceleration of FY18 milestones
- FY18 ADBB Released August 2016
- FY19 AD/ADRD milestones - $597M extra needed
- FY19 ADBB Released July 2017
- $414M in FY18 supports acceleration of FY19 milestones
- FY19 ADBB Released July 2017
- FY20 AD/ADRD milestones -- $478M extra needed
- FY20 ADBB Released July 2018
- $425M in FY19 supports acceleration of FY20 milestones
- FY20 ADBB Released July 2018
- FY21 ADBB anticipated July 2019
Ongoing NIA AD/ADRD and Related Intervention and Prevention Trials (~140)
- 29 Early-stage Clinical Drug Development (Phase I and Phase II Clinical Trials)
- Amyloid (8)
- Neurotransmitter Receptors (3)
- Metabolism and Bioenergetics (4)
- Vasculature (3)
- Growth Factors and Hormones (1)
- Multi-target (6)
- Oxidative Stress (1)
- Other (1)
- 8 Late-stage Clinical Drug Development (Phase II/III and Phase III Clinical Trials)
- Amyloid (6)
- Vasculature (2)
- 58 Non-Pharmacological Interventions
- Exercise (14)
- Diet (5)
- Cognitive Training (14)
- Combination Therapy (7)
- Other (18)
- 7 Clinical Therapy Development for the Neuro-psychiatric Symptoms of AD/ADRD
- Pharmacological (5)
- Non-Pharmacological (2)
- 34 Care and Caregiver Interventions
- 3 Delirium/ Post-Operative Cognitive Decline Trials
Ongoing NIA AD/ADRD and Related Intervention and Prevention Trials (~140)
69% of ongoing trials are non-pharmacological
- 58 Non-Pharmacological Interventions
- Exercise (14)
- Diet (5)
- Cognitive Training (14)
- Combination Therapy (7)
- Other (18)
- 7 Clinical Therapy Development for the Neuro-psychiatric Symptoms of AD/ADRD
- Non-Pharmacological (2)
- 34 Care and Caregiver Interventions
Physical Activity Clinical Trial for AD
- Exercise in Adults With Mild Memory Problems (EXERT)
- Goal: Test effects of physical exercise on cognition, functional status, brain atrophy and blood flow, and cerebrospinal fluid biomarkers of Alzheimer's disease, in adults ages 65 to 89 year with MCI (Completion: 2022)
Diet-Based Clinical Trial for AD
- MIND Diet Intervention to Prevent Alzheimer Disease
- Goal: Test the effects of a 3-year intervention of the MIND diet (Mediterranean-DASH Intervention for Neurodegenerative Delay) on cognitive decline and brain neurodegeneration among individuals 65+ years without cognitive impairment, who are overweight and have suboptimal diets (Completion: 2022)
Cognitive Training Clinical Trial for A
- Processing Speed Training to Preserve Driving and Functional Competencies in MCI
- Goal: Test the capacity of an enriched version of processing speed training (PST) to preserve functional abilities in a clinical MCI population with quantified genetic and neuroimaging AD and comorbid cardiovascular disease biomarkers (Completion: 2020)
Caregiving Clinical Trial for AD
- Indiana University Dementia Screening Trial (IU-CHOICE)
- Goal: To examine the question of whether the benefits outweigh the harms of routine screening for dementia among older adults in primary care when the screening program is coupled with primary care practices prepared to provide care for those who screen positive (Completion:2019)
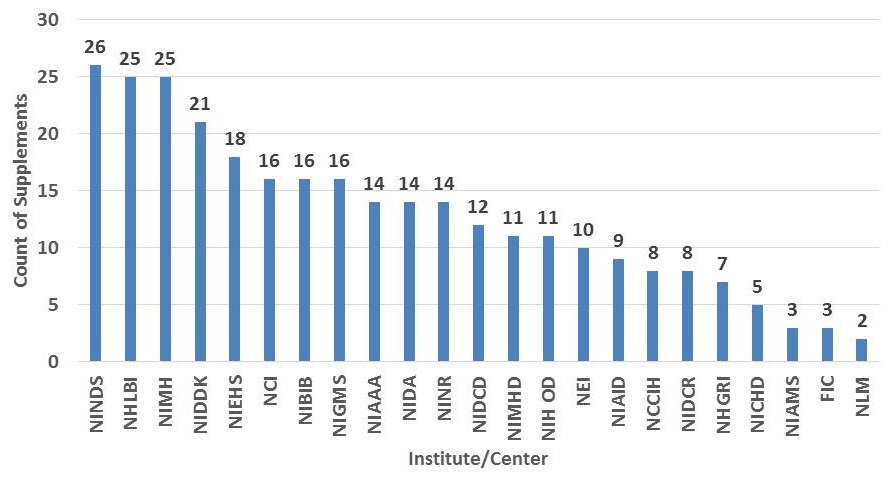
AD Supplement Funding by Institute/Center
 |
Supplement to NINR Parent Grant
- Improving Sleep Using Mentored Behavioral and Environmental Restructuring (SLUMBER)
- Goal: Test a sleep improvement program (SLUMBER) designed to teach nursing facility staff to improve common sleep-related symptoms among individuals with AD and related dementias (e.g., improve nighttime sleep environment, increase daytime activity levels and light exposure).
Supplement to NCCIH Parent Grant
- Integrating Mind Body Skills with Physical Activity to Improve Pain Outcomes in Patients with Heterogeneous Chronic Pain Conditions
- Goal: Adapt a mind body program (the Relaxation Response Resilience Program) for the specific needs of individuals with chronic pain who have documented MCI. The researchers are specifically targeting increased physical activity, with tracking devices like Fitbits.
Supplement to NIDCD Parent Grant
- Community-Delivered Affordable, Accessible Hearing Care to Reduce Symptom Burden in Alzheimer's Disease: Adaptation of HEARS Intervention
- Goal: Adapt the HEARS (Hearing Equality through Accessible Research & Solutions) intervention to older adults with ADRD and their caregivers to potentially address hearing loss and improve communication among persons with dementia.
- Brain-Computer Interface to Enhance Attention in Alzheimer's Disease
- Goal: Enhance attention and reading skills in people with mild AD through a visual-attention-to-letters training paradigm. The training uses a letter-based brain-computer interface, the Rapid Serial Visual Presentation Keyboard system, which was already established in the parent award.
NIA AD/ADRD Health Care Systems Research Collaboratory
- Goal: To improve care for persons with dementia (PWD) and their caregivers through health and long-term care systems, to include:
- primary and specialty outpatient care
- acute inpatient care
- skilled nursing and other rehabilitation facilities
- residential long-term care
- home and community-based services
- https://grants.nih.gov/grants/guide/rfa-files/rfa-ag-19-009.html
- Promote development of pragmatic trials and demonstration projects
- Develop and disseminate technical and policy guidelines/best practices for effective research collaborations
- Provide technical support for scaling up pilot studies
- Disseminate best practices for engaging stakeholders, conducting ethical research in the special circumstances of dementia care, and involving long-term and acute-care providers
REMINDER: iCare-AD/ADRD Challenge
- Improving Care for People with AD/ADRD Using Technology Challenge
- Seeks to spur the development of technology applications to improve dementia care coordination and/or care navigation
- Up to $400,000 in cash prizes may be awarded to teams or individuals that participate in the competition
- Submissions accepted from October 1, 2018 through June 30, 2019
- See https://nia.nih.gov/challenge-prize for full prize details
Save the Date
AD/ADRD Care/Services Summit (II)
March 24-25, 2020
Natcher Conference Center
Bethesda, MD
NINDS Updates
SAVE THE DATE
ALZHEIMER'S DISEASE-RELATED DEMENTIAS SUMMIT 2019: Research Challenges and Opportunities
March 14-15, 2019
Natcher Auditorium, NIH Campus,
Bethesda, MD
Julie Schneider, MD
Rush University Medical Center
Scientific Chair
Roderick A. Corriveau, PhD
NINDS, Program Director
NIH Lead
- Pleaase register online at: https://meetings.ninds.nih.gov/?ID=21149
- Attendance in person is encouraged
- Videocast will be available, and the links will be posted on the meeting website closer to the date
- Sessions on:
- Multiple Etiology Dementias
- Health Disparities
- Emerging Topics
- Nomenclature
- Frontotemporal Degeneration
- Lewy Body Dementias
- Vascular Contributions to Cognitive Impairment and Dementia
FY 2019 ADRD Research Funding Opportunities
- Diversity Notice to encourage NINDS AD/ADRD awardees to apply for administrative supplements in response to PA-18-906 Research Supplements to Promote Diversity in Health-Related Research https://grants.nih.gov/grants/guide/notice-files/NOT-NS-19-003.html
- Upcoming FOA -- Post-Stroke VCID in the U.S. including in Health Disparities Populations NOT-NS-18-092 https://grants.nih.gov/grants/guide/notice-files/NOT-NS-18-092.html
- Goal: To invite applications for a large prospective clinical research study to determine the specific subsets of stroke events that predict cognitive impairment and dementia in post-stroke populations and identify any additional clinical factors/comorbidities that influence cognitive impairment and dementia outcomes (Estimated Application Due Date: March 2019)
- Upcoming FOA -- Development and Validation of Advanced Mammalian Models for ADRD
- Goal: To invite applications that would develop improved mammalian models of Lewy body, frontotemporal, vascular, and mixed dementias that recapitulate comprehensive disease phenotypes
- 6 more new ADRD funding opportunities are planned for FY 2019
