Monday, April 29, 2019
Printer Friendly Version in PDF Format (9 PDF pages)
Alzheimer's Disease and Related Dementias Research Update
Richard J. Hodes, M.D.
Director, NIA
Alzheimer's Disease Immunotherapy Phase III Trials
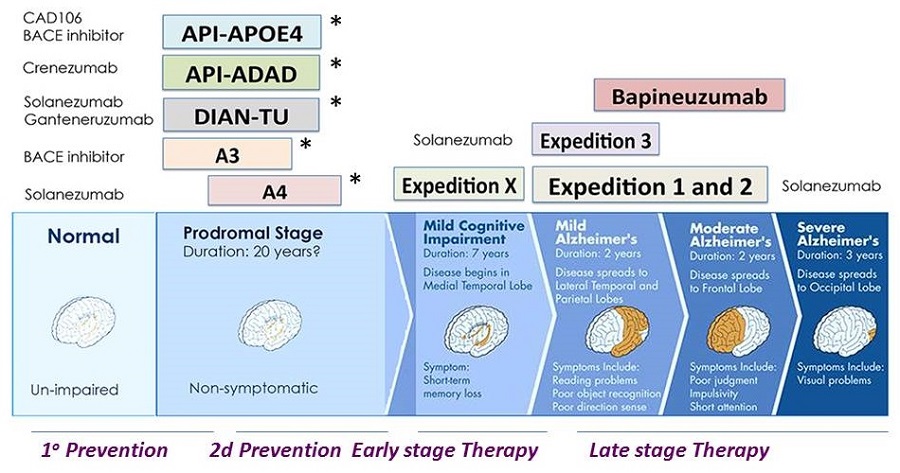 |
| * NIA sponsored (public-private partnerships |
Ongoing NIA AD/ADRD and Related Intervention and Prevention Trials (~140)
| 29 Early-stage Clinical Drug Development (Phase I and Phase II Clinical Trials) | 6 Late-stage Clinical Drug Development (Phase II/III and Phase III Clinical Trials) | 53 Non-Pharmacological Interventions | 7 Clinical Therapy Development for the Neuro-psychiatric Symptoms of AD/ADRD | 39 Care and Caregiver Interventions | 3 Delirium/ Post-Operative Cognitive Decline Trials |
|---|---|---|---|---|---|
| Amyloid (8) Receptors (4) Metabolism and Bioenergetics (4) Vasculature (3) Growth Factors and Hormones (1) Multi-target (6) Oxidative Stress (2) Other (1) |
Amyloid (5) Vasculature (1) |
Exercise (13) Diet (5) Cognitive Training (14) Combination Therapy (7) Other (14) |
Pharmacological (5) Non-Pharmacological (2) |
NIA-AD clinical trials pipeline toward 2025
 |
NIA Pipeline of Discovery
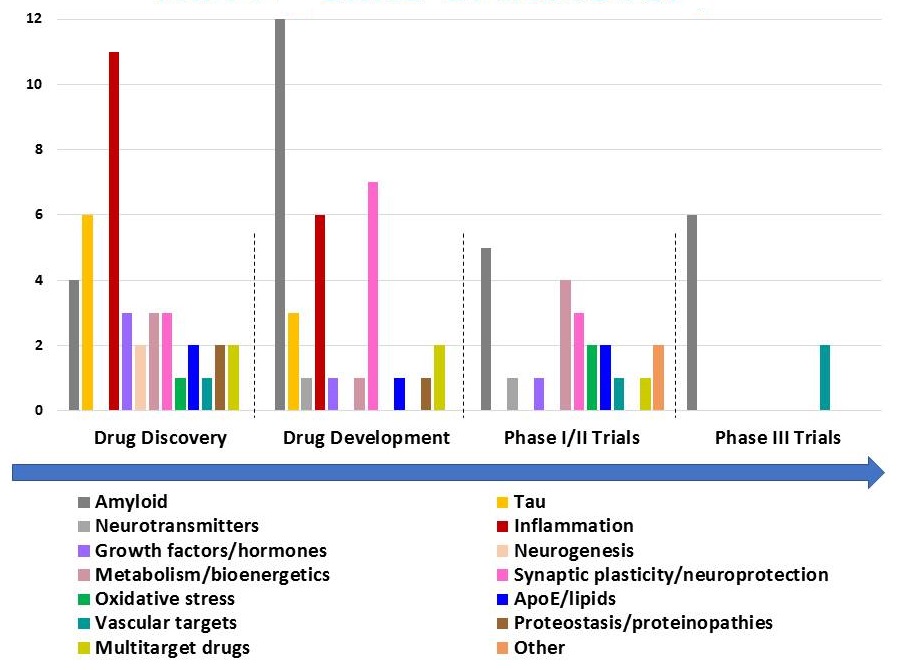 |
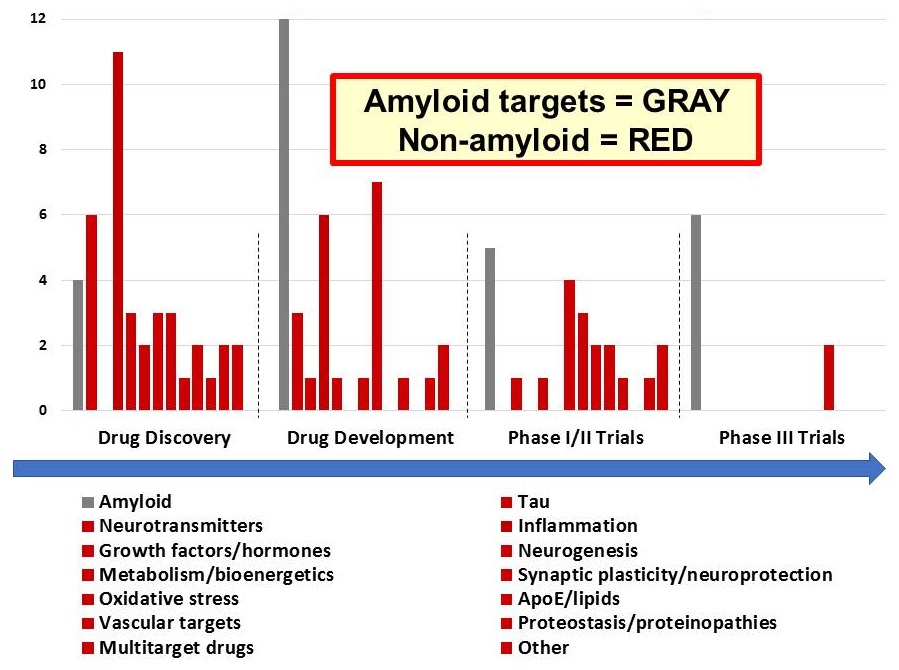 |
Genetic Regions of Interest in Alzheimer's Disease
| By Year of Discovery |
|---|
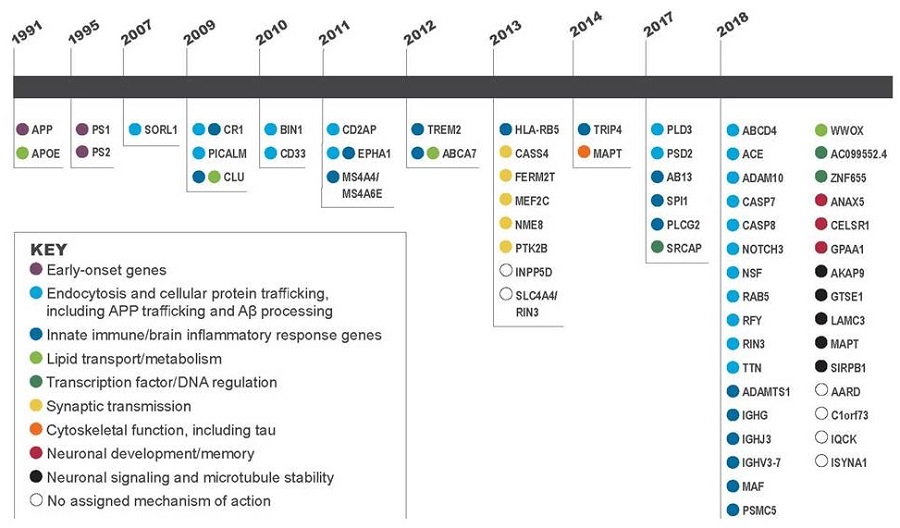 |
| NOTE: Color indicates mechanism of action in the body. See key. |
Accelerating Medicines Partnership (AMP) -- Alzheimer's Disease
COMPLETED:
- Centralized data resource established -- AMP-AD Knowledge Portal
- Rich genomic, proteomic, metabolomic human data (raw and processed) made available and being widely used -- over 2000 users to date.
- Network models of disease pathways/targets developed
- Over 100 novel candidate targets identified and being prioritized in collaboration with industry partners; experimental validation ongoing on over 20 candidate targets/pathways.
- Animal models phenotype/evaluated relative to human networks
- Web-based interface for sharing target nominations and analytical outputs developed - AGORA platform; 100 annotated target predictions and additional 480 key driver genes made publicly available.
AD Centers for Discovery of New Medicines - RFA AG 19-010
- https://grants.nih.gov/grants/guide/rfa-files/RFA-AG-19-010.html
- -Applications submitted Feb 2, 2019-
- -NIA plans to commit ~$75M over 5 years to support 2 Centers-
- Accelerate robust preclinical validation of candidate targets delivered by AMP-AD and other target discovery programs, through the development of open source tools, reagents and methods and by integrating a series of novel targets into drug discovery campaigns.
| Center Structure |
|---|
 |
AHRQ-National Academies Study
- Assessment of interventions to prevent AD-type dementia, amnestic MCI, and age-related cognitive decline
- Insufficient evidence to justify a public health campaign
- Encouraging but inconclusive evidence:
- Cognitive training
- Blood pressure management in hypertensives
- Increased physical activity
 |
| http://www.nationalacademies.org/dementia |
SPRINT Study
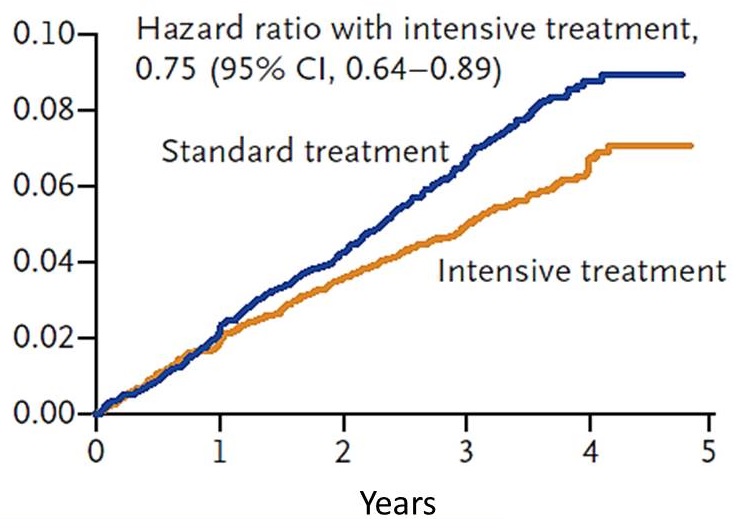 |
| The SPRINT Research Group (2015) NEJM 373(22):2103-16. |
SPRINT-MIND Research Question -- SPRINT Memory and Cognition in Decreased Hypertension
- Does intensive blood pressure control compared with standard control reduce the occurrence of dementia?
- Randomized Controlled Trial Target Systolic Blood Pressure
- Intensive Treatment Goal SBP < 120 mmHg (n= 4,278)
- Standard Treatment Goal SBP < 140 mmHg (n= 4,285)
- Randomized Controlled Trial Target Systolic Blood Pressure
The SPRINT MIND Investigators for the SPRINT Research Group (2019). JAMA, 321(6):553-561.
SPRINT-MIND: Secondary Cognitive Outcomes
- 19% reduction in the rate of developing MCI
- 15% reduction in the rate of composite MCI and probable dementia
The SPRINT MIND Investigators for the SPRINT Research Group (2019). JAMA, 321(6):553-561.
Accelerating Caregiving Research
- Multiple funding solicitations
- 2017 & 2020 care/caregiving research summits
- AD/ADRD Health Care Systems Research Collaboratory
- Systematic review of care/caregiving interventions
- NIA's first Alzheimer's prize
iCare-AD/ADRD Challenge
- Improving Care for People with AD/ADRD Using Technology Challenge
- Technology applications to improve dementia care coordination and/or care navigation
- Up to $400,000 in cash prizes
- Submissions - October 1, 2018 through June 30, 2019
- See https://nia.nih.gov/challenge-prize for full prize details and archived webinar for participants
Alzheimer's Disease-Related Dementias Summit 2019
- On March 15-16, 2019 on the main NIH Campus, Bethesda MD
- Goal: To update the research recommendations from ADRD 2016
- 7 Sessions:
- Multiple Etiology Dementias
- AD/ADRD Health Disparities
- Lewy Body Dementias
- Dementia Nomenclature
- Vascular Contributions to Cognitive Impairment and Dementia
- Frontotemporal Lobar Degeneration
- Emerging AD/ADRD -- TDP-43 in Common Dementias, Traumatic Brain Injuries and AD/ADRD Risk
- Final Recommendations will be approved by NINDS Council in September, submitted to the NAPA Council in November, and will become ADRD Research Milestones after NAPA Council approval
- Watch the archived videocast:
