Contents
- Background and Purpose
- Methodology
- Summary of Key Findings and Stakeholder Suggestions
- Conclusions and Policy Implications
A. Background and Purpose
The Office of the Assistant Secretary for Planning and Evaluation (ASPE) commissioned The Lewin Group (Lewin) to determine how and to what extent cost-effectiveness (CE) considerations are incorporated in the approval and adoption of new health technologies and the implications of not incorporating such considerations. This report examines the use of CE and other cost-health tradeoff evidence by federal and nonfederal health stakeholders, paying particular attention to the scope of authority, range and/or circumstances of use, and responsibilities for regulating CE and other economic information by the Food and Drug Administration (FDA). The role of economic evidence in decision-making also is explored in case studies of four contemporary health technologies.
B. Methodology
Lewin completed two stages of research and analysis culminating in this report. The first was an environmental scan of the current application of economic evidence in decision-making for new health technologies. The second consisted of four case studies conducted to illustrate the use of this evidence for four technologies: nucleic acid testing; Relenza (zanamivir); drug-eluting stents; and implantable cardioverter-defibrillators.
Lewin conducted primary and secondary data collection and analysis for this report. For the environmental scan, primary data were collected during semi-structured discussions with senior staff and other experts representing key federal agencies; private payers; manufacturers; and other health stakeholders from the business, academic and policy community (e.g., health economists, technology assessment organizations) involved in the innovation, adoption and diffusion of new health technologies. Secondary data collection included a review of published and unpublished peer-reviewed and other substantive literature using relevant bibliographic databases (e.g., MEDLINE/PubMed) and web-based search engines. For each case study, semi-structured discussions also were held with stakeholders with relevant expertise. Findings from these discussions were supplemented with secondary data collected from the literature and web-based resources.
After gathering data for the environmental scan and case studies, we conducted a qualitative assessment of interview responses and perspectives in the literature to perceive trends, to characterize use of evidence on CE and other health and economic tradeoffs and to compile potential options for application of cost-effectiveness analysis (CEA) as suggested by some of our sources.
C. Summary of Key Findings and Stakeholder Suggestions
Key Findings
Citing an environment of rising health care costs and insufficient access to care for many Americans, nearly all interviewees recognized potential value of using CE or other cost-health tradeoff evidence in decision-making pertaining to new health technology. At the same time, interviewees expressed caution regarding how economic evidence is and could be incorporated into policymaking. Many stressed that economic evidence should not be applied for cost control alone or rationing of safe and effective interventions, and that any considerations of cost-health tradeoffs should be inputs to a broader set of important factors mediating the introduction and use of new health care technology. Interviewees acknowledged tension in relationships among certain stakeholder groups concerning matters such as transparency, openness and clarity of the process for incorporating economic evidence.
Regarding the point in the technology lifecycle at which use of cost-health tradeoff evidence is most appropriate, interviewees offered responses ranging from the early stages of innovation to the postmarket phase. While interviewees generally were familiar with the use of CEA in one or more federal agencies, the one most frequently cited was the Agency for Healthcare Research and Quality (AHRQ), although most interviewees were aware that this agency conducts or supports these analyses but does not have regulatory or payment responsibilities. While many interviewees expressed interest in expansion of certain CE applications in the public and private sectors, none suggested that FDA incorporate CE or other cost-health tradeoff considerations in the agency's premarket or postmarket regulatory decisions. A few interviewees for the environmental scan and case studies saw some merit in having the FDA expand processes to determine the economic impact of its guidances.
Responses about the development, current use and potential use of CE and other cost-health tradeoff evidence tended to differ by the type of stakeholder interviewed. Among federal stakeholders, perspectives about the role of FDA were influenced by the extent of interaction between the interviewee's agency and FDA. The following represent the most significant findings regarding development and use of CE and other cost-health tradeoff evidence in decision-making pertaining to new health care technology. The subsequent section includes stakeholder suggestions for improving current systems or structures pertaining to the use of economic information.
1) The types and scope of health economic analysis are diverse
There is no single appropriate method of conducting CE or other cost-related analysis for health care decision-making.
- The intended use of an economic analysis should inform the most appropriate type of analysis to employ in any given instance. For instance, CEA may be most useful to a major payer considering the circumstances for covering a new technology, whereas cost-consequences analysis might be more useful to a hospital staff weighing the pros and cons of using a particular technology.
- Apart from selecting an appropriate type of economic analysis for a given circumstance, our interviewees concurred that patient health considerations are most important and that economic factors can be among multiple considerations in health policy or clinical decisions.
2) Formal use of CE evidence has been less common in the US than in certain other nations (e.g., Australia, Canada, UK)
Australia and Canada have formal systems to request and incorporate economic evidence into pharmaceutical and other technology payment decisions. The UK's National Institute for Health and Clinical Excellence (NICE) reviews economic evidence pertaining to many types of health technologies as part of the guidance that it issues to the National Health Service. Stakeholders in these systems have expressed concerns about the relatively closed nature of the Australian system, while generally commending the more accessible and transparent process of NICE.
- There are many potential explanations for the differential uptake of CE evidence in the US and abroad. The literature in this area cites potential obstacles such as methodological concerns, insufficient training, legal concerns, insufficient trust and social acceptance and health system and political barriers.
3) Among DHHS agencies and other federal agencies that influence the climate for innovation, adoption and diffusion of new health technologies, there is great variability in the ways that CE and other cost-health tradeoff information is used and in the authority to use such information
- Stakeholders repeatedly identified certain federal health agencies (e.g., AHRQ) as being involved in CE and other cost-health tradeoff studies, but were less certain about the roles of others, especially with regard to how economic evidence is used in decision-making.
- Federal agencies involved in the development of CE or other cost-health tradeoff evidence include AHRQ, the Centers for Disease Control and Prevention (CDC), Centers for Medicare & Medicaid Services (CMS), National Institutes Health (NIH) and the Veterans Administration (VA). In diverse ways, these agencies sometimes consider, review or use CE or other economic evidence to inform certain decisions (e.g., payment level, benefit structure, program impact). These agencies include AHRQ (and its US Preventive Services Task Force), CDC, CMS, Department of Defense (DoD), FDA and the VA.
- Although these agencies occasionally have some role in the development or use of CE or other economic evidence, the overall level of use of economic evidence in decision-making for new health technologies is relatively low. Across the four case studies, none of these agencies appears to have explicitly incorporated economic considerations into their decision-making processes for the four case study technologies. When economic factors were involved, stakeholders indicated that these factors were more tangential to decision-making, or that it was unclear if economic factors were considered at all.
- The extent of current and future use of CE and other cost-health tradeoff evidence by federal agencies is limited by their respective legislated missions and applicable regulations.
4) Health economists and other stakeholders suggest that, given rising health care costs and system constraints, CE and other economic evidence can provide important input to inform more effective and efficient health decision-making in the US
- Continued growth in domestic health spending of nearly 8% per year, now amounting to 16% of the gross domestic product with double-digit increases in annual health insurance premiums in each of the past four years, is adding to concerns of government, industry and consumers.
- Health care providers, payers, consumers and others increasingly are intent on achieving quality care and value for their health care dollar. Initiatives such as pay-for-performance are prominent examples of this trend.
- Many interviewees expressed that greater and more explicit adoption of CEA or other forms of economic analysis by CMS, other federal entities and private sector payers would inform more credible resource allocation and contribute to better value in health care.
5) Although both public and private stakeholders recognize the potential value of using CE or other cost-health tradeoff evidence, currently, there is no standard set of criteria for determining when economic factors are relevant and how they are to be used in decision-making
- Technology manufacturers expressed that, when they submit economic data to federal agencies like CMS, they are uncertain regarding how the information will be used and how it will affect adoption and payment of their technology.
- These industry stakeholders expressed concerns that economic evidence may be weighted too high relative to other important factors, thereby diminishing matters of clinical utility and patient access.
- Many stakeholders, particularly those from industry, perceived that current applications of CEA in health care delivery and policy decisions are lacking in transparency and resulting in somewhat unpredictable outcomes.
- Interviewees for one of the case studies also suggested that some industry and professional association stakeholders perceive that economic factors were at the root of new technology decisions, even when the decision-makers maintain that clinical evidence was the main consideration.
6) Currently, there is not a uniformly accepted standard for information included in CEAs
- Manufacturers expressed that payers provide little or no guidance regarding what should be included in CEAs to support payment decisions. As a result, manufacturers use varying assumptions and endpoints in these analyses and then, when they submit these analyses to payers, the payers find that the CEAs did not employ desired endpoints or assumptions.
- From the standpoint of public and private payers, CE models submitted by manufacturers often are insufficiently relevant to decision-making. For instance, payers indicated that manufacturers are not always explicit about assumptions used in CE models, and that these models often are not designed for interactive use by payers.
7) In the large and fragmented US health care system, there is no national, standardized process for setting priorities among health issues that could merit CEA
- Many federal and nonfederal stakeholders emphasized that the US lacks a systematic approach to determining priorities for CE research applying to interventions across a range of health conditions.
- As a result, current allocations of CE research may not address the most pressing health topics, and reviews of CE evidence may not account systematically for variations in the quality of this evidence.
8) The current role of FDA in development or use of CE evidence is very limited
- FDA's mission pertaining to health care technology focuses on reviewing evidence of safety and effectiveness pertaining to market approval and postmarket surveillance. Consideration of CE or other economic evidence in market clearance or approval of regulated technologies is not pursuant to FDA's mission, limiting the agency's purview to address these topics.
- FDA does have the responsibility to regulate claims of CE made by manufacturers about particular technologies. Many interviewees believe that FDA's regulation of such claims may stifle the availability of useful CE evidence for new health technologies unnecessarily.
- If FDA, or any other federal agency, issues new regulations meeting certain criteria, it is required to conduct a regulatory impact analysis, including analysis of the CE of such regulations, as mandated by Executive Order 12866 and Circular A-4. However, these analyses primarily gauge the impact of an entire regulation and rarely, if ever, pertain to particular health technologies that may be subject to these regulations.
9) In contrast to the impact analysis pertaining to new regulations, FDA has no statutory authority or mechanism for evaluating the economic impact of guidances
- Periodically, FDA issues guidance documents to address clinical trial design, good manufacturing practices (GMPs) or use of new technologies within the blood industry. A 2005 FDA guidance on the use of a particular type of nucleic acid testing to screen the blood supply received attention from some economists and other stakeholders. Despite the considerable additional cost of this testing and its marginal improvement in detection of pathogens, FDA did not consider the economic impact of this guidance formally.
- While FDA has no statutory authority to perform economic impact analyses of guidances, and its mission specifies evaluating safety and effectiveness, some stakeholders noted that there are no prohibitions for FDA to consider economic evidence when drafting guidance. Therefore, with no explicit restriction against doing so, it may be possible for FDA to incorporate economic evidence in this capacity.
- Stakeholders expressed openness to developing a mechanism for review of guidance documents. Stakeholders indicated that, if such a mechanism were developed, the reviewing agency would have to establish criteria for evaluating CE or economic impact, determine which stakeholders should be involved and identify an appropriate source of funding.
10) While FDA does not require economic evidence in market approval, FDA, CMS and other stakeholders (including manufacturers) are communicating more often during the review phase for new health technologies
- During internal reviews and as a result of this type of communication, FDA may consider resource utilization or other potentially cost-related endpoints (e.g., average length of stay in hospitals) if these endpoints relate directly to safety and effectiveness (e.g., associated with elevated risk of developing secondary/nosocomial infections).
- Despite increased communication among FDA and these parties, and some greater interest in CEA on the part of payers and some other stakeholders in CE evidence, this does not appear to be broadening the scope of FDA's focus beyond matters of safety and effectiveness.
11) Virtually all interviewees expressed that consideration of CE or other cost-health tradeoff evidence during market approval or postmarket surveillance could compromise or distract from the FDA's core mission of ensuring safety and effectiveness of regulated health care products
- Many stakeholders emphasized how resource-intensive FDA's responsibilities are regarding ensuring safety and effectiveness of health care technology, and that the agency currently lacks the internal capacity and statutory authority to incorporate economic evidence into its decisions.
- Some stakeholders expressed concern that weighing economic evidence at the approval phase for a new technology might result in withholding or delaying market entry of beneficial technologies. Similar concerns were expressed in stakeholder interviews conducted for the case studies. These concerns also were expressed in stakeholder interviews conducted for the case studies.
Health Stakeholder Suggestions
Stakeholders interviewed were forthcoming about contemporary development and use of CE and other economic evidence, as well as perceived limitations to potentially beneficial applications of such evidence. Some interviewees suggested ways of remedying these limitations. Themes and individual suggestions for using evidence on CE or other health and economic tradeoffs of new technologies are compiled here. Stakeholder suggestions are divided into two broad headings: 1) process and implementation considerations and 2) considerations specific to the FDA.
1) Process and Implementation Considerations
The great majority of interviewee suggestions relate to modifying the current system to better incorporate CE and other economic evidence into open and transparent policymaking processes. Overarching questions inherent to implementing such provisions address which entities might coordinate the process and potential sources of funding.
Several options emerged from stakeholder suggestions about the proper entities to coordinate a system for review and use of CE and other economic evidence. Among the federal agencies, stakeholders were most likely to identify AHRQ as the most appropriate and best equipped agency to take on this role. Many stakeholders emphasized that AHRQ currently is acting as a facilitator of CE evidence development and use already and, hence, would be a natural choice. However, others suggested that any federal entity coordinating such a process would be susceptible to political pressures that might introduce bias into activities. As such, stakeholders also suggested creating new entities to fill this role, as described below.
- Independent entity within government. Stakeholders repeatedly referenced establishing a body in the US with a role similar to that of NICE in the UK, which acts independently as a Special Health Authority to the National Health Service, providing guidance informed by clinical and economic evidence. Some stakeholders referenced the Federal Reserve (the central bank of the US) as a similar arrangement that could serve as a potential model.
- Fully independent entity. Some stakeholders favored establishing an entity that would act independently of government or industry. Among those discussed was an organization with a status similar to that of the Institute of Medicine, which would be responsible for coordinating the steps involved in setting priorities for and conducting or sponsoring CEAs. Other independent models were offered, including the Pharmacoeconomic Research Institutes (PERIs) model (which has been suggested by Princeton economist Uwe Reinhardt). PERIs would be funded to conduct economic research on drugs using funding from a small surcharge on the pharmaceutical industry.
Aside from the PERIs strategy, few suggestions emerged from this environmental scan related to funding new systems for incorporating CE or other economic evidence into policymaking. Nevertheless, stakeholders emphasized that responsibility for funding should be shared by public and private stakeholders, ideally in some form of partnership.
Stakeholder suggestions about individual steps in the process of incorporating CE or other economic evidence into decision-making fall roughly into four main categories, as depicted in Exhibit 1, along with relevant questions at each step. Suggestions are summarized according to these four categories.
Exhibit 1:
Key Considerations for Integrating CE and Other Economic Evidence into Policy 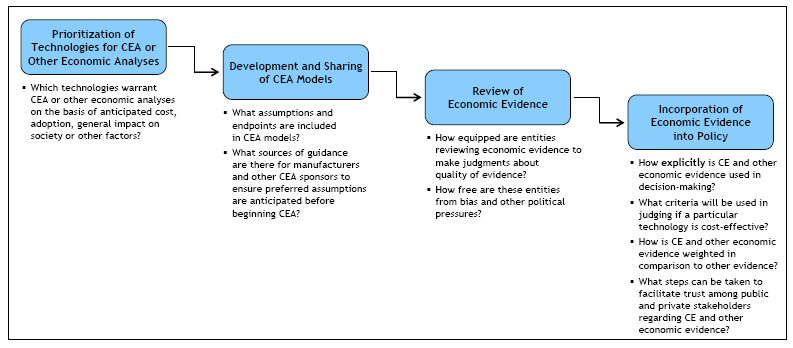
Setting Priorities among Technologies for CEA or Other Economic Analyses
Stakeholders emphasized the importance of instituting means to set priorities for determining which technologies warrant CEA or other forms of economic analysis. In suggesting approaches, some stakeholders noted that AHRQ already has instituted a process for identifying topics for clinical evidence assessments as part of its Evidence-based Practice Centers (EPC) program. Similar to the process used by NICE in the UK, the EPC program selects from among topics nominated for systematic evidence review by professional associations, payers, patient groups and other organizations. Some stakeholders suggested that this portion of the EPC process might be expanded to provide a systematic priority-setting process for implementing economic studies.
Development and Sharing of CEA Models
Manufacturers often conduct or sponsor CEAs for internal purposes and to share with decision-makers such as payers and providers. Stakeholders reported that manufacturers often submit CEA models, only to learn from payers that the models do not incorporate assumptions or endpoints preferred by the payers. From their standpoint, payers often find that models submitted by manufacturers are not interactive and that assumptions used in the models are not readily apparent. As such, stakeholders suggest the need for an objective entity or entities to help set standards about assumptions to be used in CEAs and guidelines for manufacturers to help increase transparency of models submitted to payers. Increased clarity may help to guide CEAs conducted or sponsored by technology manufacturers, so that they may be aligned better with payer expectations. This may mitigate manufacturer risk and improve timeliness of market approval and payment decisions.
Review of CE and Other Cost-health Tradeoff Evidence
In addition to establishing guidelines for developing and sharing CEA models, stakeholders suggested that an objective entity might have a role in reviewing cost-health tradeoff evidence. Some stakeholders proposed that an agency such as AHRQ could have a role in coordinating economic analyses, including evaluating the quality of available evidence and synthesizing findings from existing literature, in the current manner of AHRQ's Evidence-based Practice Centers (EPCs). Well-recognized technology assessment groups such as the Blue Cross Blue Shield (BCBSA) Technology Evaluation Center (TEC), ECRI or HAYES may have similar roles.
Incorporation of CE and Other Economic Evidence into Policy
To improve the clarity and transparency of current CEA efforts, stakeholders suggested that the private and public sector payers could facilitate greater trust among industry stakeholders by clearly establishing how economic evidence will be used (e.g., for what types of decisions) and its role relative to other technology attributes or criteria. Some stakeholders suggested that establishing a public-private partnership to develop a standard framework for use of CE and other economic evidence may enhance transparency and strengthen trust in these processes.
2) Considerations Specific to FDA
The clear consensus of the stakeholders whom we interviewed for the environmental scan and the case studies was that FDA should not consider CE or other economic factors in matters pertaining to market approval or postmarket surveillance. No stakeholder raised suggestions for using these approaches at FDA. However, stakeholders did offer suggestions pertaining to other ways in which FDA authority might affect CE or other economic evidence directly or indirectly, as follows.
- Some stakeholders have proposed ways to respond to concerns that FDA regulation of economic claims made by manufacturers can inhibit availability of CE evidence for new technologies. One health economist has suggested that FDA consider adding disclaimers about assumptions used in CEAs to products advertised using CE claims. An example of such a disclaimer could be, "This claim of cost-effectiveness is based on assumptions and simulations that may not meet the FDA criteria for claims of efficacy and safety."(1)
- Certain interviewees raised the potential importance of evaluating the economic impact of FDA's guidance documents. They noted that the agency formally has not been granted legislative authority to conduct analyses of guidance documents. However, there are no apparent restrictions upon the agency for considering economic factors in developing guidances, suggesting that FDA may be able to consider these factors. In any case, it would be necessary to allocate funding for this purpose.
The few stakeholder suggestions pertaining to use of CEA or other economic analyses by the FDA reflects their general concurrence that CEA is beyond the realm of FDA's responsibilities pertaining to marketing and postmarket surveillance of regulated health care technologies. Stakeholders emphasized that expanding the purview of the agency to include matters of CE or other economic evidence, even given a new legislative mandate, would compromise the importance of the agency's core mission pertaining to the regulated technologies.
To the extent that CMS, other public and private sector payers or health care providers become involved in using CE information in ways that increase market pressure for more cost-effective health care, this would further diminish any rationale for FDA to use CE information in regulating health technologies.
D. Conclusions and Policy Implications
At present, the level of use of economic evidence in health care decision-making is relatively low. There are several important potential implications of using, and not using, this evidence. If cost-effectiveness or other economic evidence is incorporated more in decision-making for new health technologies, the following may be relevant:
- Greater use by one party could stimulate broader use of economic evidence. If certain stakeholders, especially FDA or CMS, incorporate economic considerations to a greater extent, this could encourage more use among other stakeholders.
- If certain stakeholders adopt economic evidence into decision-making, this could encourage further economic studies to be conducted. In particular, if FDA or CMS were to begin considering explicitly such evidence, manufacturers of drugs, devices and other health technologies may be more inclined to sponsor or conduct CEAs or other economic studies in coordination with clinical data collection.
- To address concerns regarding the use of economic factors in decision-making, stakeholders may need to consider how to ensure that economic evidence is used appropriately and accounts for societal values. This could include formalizing ways of using economic evidence and ensuring transparency in relevant decision-making processes.
If the use of economic evidence in health care decision-making is not altered substantially, another set of implications could arise:
- If CEA or other economic analyses are not adopted into health technology decision-making, the need for some means of informing health care resource allocation will remain. As rising health care costs account for a larger portion of the GDP, the cost of health care technology, particularly new "high-ticket" technologies, will draw stakeholder and public attention.
- Aside from resource allocation, not using economic evidence could place financial burden upon certain stakeholders. For example, stakeholders interviewed expressed the view that, while FDA guidance documents technically are not binding, often, they are perceived that way. If economic factors are not considered during the guideline development process (e.g., costs for various stakeholders of implementing a particular technology), those responsible for implementing the technology may have trouble managing additional expenses.
- Stakeholders, including the public, may seek to become more familiar with and interested in incorporating economic factors into health care decision-making. Currently, there are concerns regarding the use of economic evidence in this context. These concerns can be addressed, at least in part, to the extent that stakeholders continue to standardize the methodology for incorporating this evidence in a transparent way.
This report provides a basis for understanding the implications of greater or lesser use of economic evidence in decision-making regarding new health technologies. These insights may be useful in informing future policymaking or other initiatives in this area.
Endnotes
1. Luce BR. What will it take to make cost-effectiveness analysis acceptable in the United States? Med Care 2005;43(7):II-44-8.
