Monday, January 27, 2020
Printer Friendly Version in PDF Format (20 PDF pages)
NINDS Update & ADRD Summit 2019 Report
Roderick Corriveau, PhD
Program Director & AD/ADRD Program Lead, NINDS
Pathway to Dementia
Alzheimer's Disease
Add
- Lifestyle Factors
- physical activity
- diet
- drug/alcohol abuse
- Environmental Factors
- education
- head trauma
- toxins/other
- Psychosocial Factors
- depression/anxiety
- Aging
- Genetic Factors
- Sex F>M
- Other Medical Risks
- hypertension
- obesity
- stroke
- heart disease
- diabetes
- metabolic
- inflammation
- certain infectious diseases
- certain medications
- Health Disparities Factors
- *BRAIN CHANGES
- *Misfolded proteins
- amyloid
- tau
- alpha synuclein
- TDP-43
- *Vascular Disorders
- injury, infarct (stroke)
- white matter disease
- blood vessel disease
- *Other Disorders
- *Misfolded proteins
- Cognitive Impairment Including Dementia
- Alzheimer's Dementia
- Lewy Body Dementias
- Vascular Dementias
- Frontotemporal Dementias
- Limbic Predominant TDP
- Mixed Dementias
- Other Cognitive Impairment
- Other Dementias
Concept by: Julie A. Schneider, MD, MS, Rush University & Roderick A. Corriveau, PhD, NINDS
The NINDS and NIA Collaborate on AD/ADRD Research
- NIA is the NIH lead for AD research and responding to the National Plan
- NINDS is the NIH lead for ADRD research (FTD, LBD, VCID), including ADRD Summits
- NAPA Goal 1: Prevent and Effectively Treat AD/ADRD by 2025
- Triennial AD, ADRD and Care Summits*
- *Dementia Care Services Research Summit NIH/NIA, March 14-15, 2020
- Research Recommendations
- Milestones
- Scientific Advances Toward Goal 1
- Triennial AD, ADRD and Care Summits*
Alzheimer's Disease-Related Dementias (ADRD)
- ADRD: Types of dementias that share cognitive and pathological features with Alzheimer's and/or commonly co-occur with typical Alzheimer's pathology
- Vascular Contributions (VCID)
- Vascular contributions to cognitive impairment and dementia (VCID)
- Frequently a part of typical clinical Alzheimer's disease
- Diverse spectrum of disorders caused by cerebro- & cardio-vascular disease
- Reducing vascular risk factors may decrease dementia risk
- Lewy Body Dementia (LBD)
- Lewy bodies, pathological hallmark of Parkinson's disease, also present in brains of people with Lewy Body Dementia (LBD, PDD)
- Dementia occurs with problems with movement, sleep, mood, & hallucinations
- Frontotemporal Degeneration (FTD)
- Onset often occurs in a person's 50s or 60s
- Progressive decline in social behavior and/or language (memory can be spared)
- Can be associated with amyotrophic lateral sclerosis (ALS)
- Mixed Dementias (MED)
- Majority of all dementia cases (age 65+) are mixed or multiple etiology dementias, mainly Alzheimer's pathology (beta-amyloid plaques and tau tangles) with cerebrovascular disease and/or Lewy bodies
NINDS-Led Summits Shape ADRD Scientific Priorities
- National Plan Action 1.A.6: NINDS to Regularly convene ADRD Summits to review progress & refine & add new ADRD research recommendations as appropriate based on scientific discoveries.
- Triennial ADRD Summits
- 2013, 2016, 2019
- Research Recommendations
- Milestones
- Scientific Advances Toward Goal 1
- Triennial ADRD Summits
- ADRD Summit Scientific Chairs
- Dr. Tom Montine (2013)
- Dr. David Holtzman (2016)
- Dr. Julie Schneider (2019)
NIH Investment in AD/ADRD Research (Millions)
| Fiscal Year: | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 (est.) |
|---|---|---|---|---|---|---|---|
| AD/ADRD | Category Not Yet Tracked |
Category Not Yet Tracked |
$631 | $986 | $1423 | $1911 | $2468 |
| Alzheimer's Disease | $504 | $562 | $589 | $929 | $1361 | $1789 | $2305 |
| Frontotemporal Dementia | $32 | $37 | $36 | $65 | $91 | $94 | $98 |
| Lewy Body Dementia | Category Not Yet Tracked |
$15 | $15 | $22 | $31 | $38 | $39 |
| Vascular Contributions to Cognitive Impairment and Dementia | Category Not Yet Tracked |
$45 | $72 | $89 | $130 | $259 | $268 |
| SOURCE: https://report.nih.gov/categorical_spending.aspx | |||||||
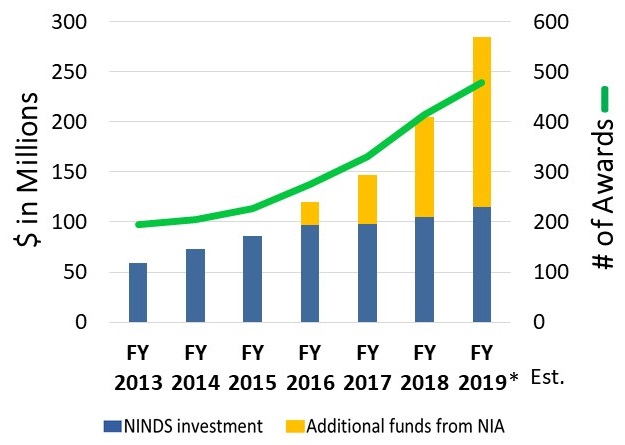
AD/ADRD Funding Strategy at NINDS
- Additional AD/ADRD funds from NIA are used by NINDS for:
- Research Responsive to AD/ADRD Initiatives
- FY19 (est.): $112 M
- Increased Investment in Investigator-Initiated AD/ADRD Research
- FY19 (est.): $58 M
- Research Responsive to AD/ADRD Initiatives
 |
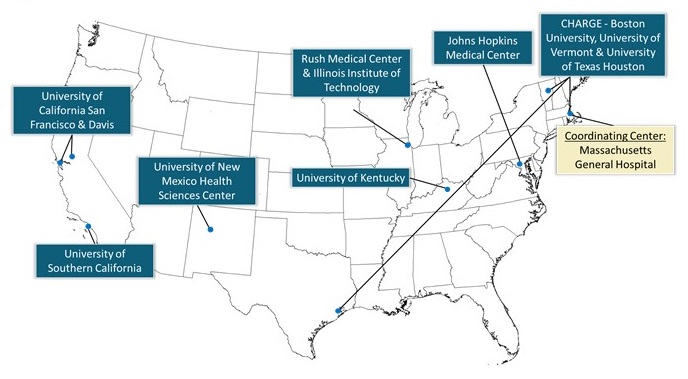
Examples of ADRD Research Initiatives: VCID, MED
- Mark VCID
- National consortium to develop and validate predictive, diagnostic, target engagement and progression biomarkers in human VCID including in clinical Alzheimer's and other mixed dementias
- detectCID
- National consortium with sites in Chicago, New York, and San Francisco
- Address the unmet need to detect cognitive impairment, including dementia in primary care across the United States
- Including in populations that experience health disparities
- Proposed clinical paradigms should utilize tools that are simple to use, standardized, and ideally take five-ten minutes or less to administer in a primary care clinical setting
 |
Examples of ADRD Research Initiatives: FTD, LBD
- Tau Center without Walls
- Interdisciplinary, multi-institute research
- Identification and validation of molecular mechanisms that contribute to tau toxicity associated with Frontotemporal Degeneration (FTD)
- 2 teams funded in 2016
- WEST COAST Linking tau proteostasis with neuronal activity in FTD
- EAST COAST Identifying genes & pathways that impact tau
- Technology Platforms GWAS, RNA Seq, Proteomics, Genome Editing, Mouse Genetics, IPSC
- Human Biology Validation Cores
- Data Coordinating Core
- The Lewy Body Dementias Biomarker Initiative
- Supports hypothesis-driven clinical research to discover biomarkers
- 5 teams funded since 2016
- Discovery
- Sample source
- Bioinformatics
- Identification
- Analytical method development
- Validation
- Clinical Utility
- Qualification
Examples of NINDS AD/ADRD Trans-NIH Collaborations
- NINDS and NIA are funding natural history studies in FTLD
- Study of individuals with a clinical diagnosis of FTD to help determine the clinical, genetic and biomarker profiles
- Study of families that have one of the three most common gene variants that cause FTD to learn more about natural history
- M2OVE-AD
- 5 awards funded by NIA and NINDS
- M2OVE-AD aims to gain a deeper understanding of risk phenotypes and mechanisms of VCID
- VCID Workshops (5/2018; 11/2019)
- Neuropathological Impact of Sleep Deficiency & Sleep Disorders (8/2018 workshop)
- AD Viewed as a Neurovascular Inflammatory Disorder (4/2017 eBriefing)
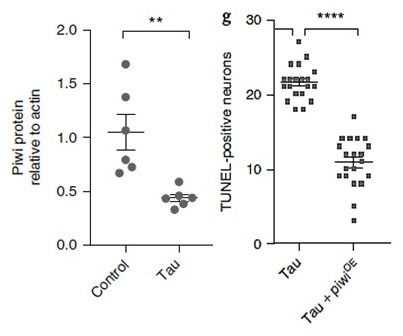
NINDS Investigator-Led ADRD Research is Increasing in Scale and Impact -- Tauopathies (FTD, AD, PSP)
- Pathogenic tau-induced piRNA depletion promotes neuronal death through transposable element dysregulation in neurodegenerative tauopathies
- Wenyan Sun[1,2,3], Hanie Samimi[4], Maria Gamez[1,3], Habil Zare[3] and Bess Frost[1,3,5]*
- 'Jumping genes' are ∼45% of human genome
- Cells utilize defenses to limit this jumping, e.g.:
- Piwi (nuclear protein) & small piwi-interacting RNAs (piRNA) help clear transposable element transcripts & keep chromatin condensed to prevent jumping
- Results suggest that tauopathy drives DNA decondensation, transposable element dysregulation (activation), and thus at a cellular level accelerates pathology
- This identifies transposable element regulation as potential pharmacological target in tauopathy
 |
NINDS ADRD Program: Summary
- NINDS leads triennial ADRD Summits (2013, 2016, 2019)
- Planning effort that: (1) Delivers ADRD Milestones to the DHHS National Plan to Address AD and (2) Informs AD Bypass Budgets
- NIH AD/ADRD budget has increased substantially; NIA shares AD/ADRD funds with the NINDS
- NIH ADRD research funding increased ∼3-fold since 2015
- NINDS AD/ADRD portfolio increased ∼3-fold since 2015
- NINDS has led 29 ADRD initiatives and programs since 2016
- Including 11 NINDS ADRD initiatives in FY 2019
- 7 additional FOA concepts approved by NINDS Council for FY2020
NINDS FY 2020 ADRD Funding Opportunity Announcement Concepts
| AD/ADRD | RFA-NS-20-005: Mechanistic Basis of TDP-43-Dependent Pathobiology in Common Dementias (R01) | Closed |
|---|---|---|
| LBD | RFA-NS-20-014: Peripheral Pathology in the Lewy Body Dementias (R01) | Closed |
| LBD | PAS-19-210: Progression Markers for Cognitive Impairment in PDD Progression Markers for Cognitive Impairment in Parkinson's Disease Dementia (R01) | Closed |
| VCID | RFA-NS-19-039: Mechanistic Basis of Diffuse White Matter Disease in VCID (R01) | Closed |
| VCID | RFA-NS-20-004: Molecular Mechanisms of Blood-Brain Barrier Function and Dysfunction in Alzheimer's Disease and Alzheimer's Related Dementias (R01) | Closed |
| VCID | RFA-NS-20-013: White Matter Lesion Etiology of Dementia in the U.S. Including in Health Disparity Populations (U19) | Active |
| VCID | RFA-NS-20-012: Clinical Trials Planning for Symptomatic Vascular Contributions to Cognitive Impairment and Dementia (VCID) (R34) | Active |
Alzheimer's Disease-Related Dementias Summit 2019
- March 14-15, 2019
- Sponsored by the National Institute of Neurological Disorders and Stroke
- National Institute on Aging
- NIH Office of Disease Prevention
- Foundation for the National Institutes of Health
- Special thanks for assistance with outreach to:
- Alzheimer's Association
- LEAD Coalition (Leaders Engaged on Alzheimer's Disease)
- Lewy Body Dementia Association
- Association for Frontotemporal Degeneration
- 2019 ADRD Summit Planning
- ∼ 6 months by >80 scientists, physicians, and administrators
- Scientific Chair: Julie Schneider
- NIH/NINDS Summit Lead: Rod Corriveau
- Steering Committee: S. Dickinson, L. Gitlin, D. Holtzman, E. Masliah, T. Montine, B. Obviagele, R. Petersen
- Session Committees
- Overarching: MED; Health Disparities; Nomenclature
- Disease-Specific: LBD; FTD; VCID; Emerging Science
- Summit Goals
- Present rationale, including scientific progress, for draft research recommendations
- Encourage discussion among group experts
- Solicit feedback and opinions from audience
- Modify recommendations based on feedback
Portions of this event were made possible by the Foundation for the National Institutes of Health with generous support from the following contributors:
- Elite
- Alzheimer's Association
- Platinum
- GHR Foundation
- Gold
- Biogen
- Silver
- Accelerate Cure/Treatments for Alzheimer's Disease (ACT-AD) Coalition
- Alzheimer's Drug Discovery Foundation
- American Stroke Association, a division of the American Heart Association
- CurePSP
- EIP Pharma
- The John A. Hartford Foundation
- WellMed Charitable Foundation
Thank You to NIH Leadership and Staff
- Walter Koroshetz (NINDS)
- Richard Hodes (NIA)
- Roderick Corriveau (NINDS)
- Debra Babcock (NINDS)
- Patrick Bellgowan (NINDS)
- Jue Chen (NHLBI)
- Sara Dodson (NINDS)
- Cerise Elliott (NIA)
- Jordan Gladman (NINDS)
- Amelie Gubitz (NINDS)
- Sophia Jeon (NINDS)
- Melinda Kelley (NIA)
- John Hsiao (NIA)
- Eliezer Masliah (NIA)
- Daniel Miller (NINDS)
- Claudia Moy (NINDS)
- Toya Rogers (NINDS)
- Jonathan Sabbagh (NINDS)
- Paul Scott (NINDS)
- Beth-Anne Sieber (NINDS)
- Margaret Sutherland
- Margo Warren (NINDS)
Approved ADRD Summit Recommendations Become National Plan Milestones
 |
ADRD Summit 2019 Results
- FACA Committee-approved ADRD Summit 2019 Report
- FACA Committee-approved prioritized ADRD research milestones (47) with timelines that will:
- Inform future NIH AD/ADRD bypass budgets and, as appropriated funds become available, corresponding research activities.
- Guide the AD/ADRD research community generally, and as such, broad implementation and execution by various federal, national and international stakeholders will be vital to their impact.
Highlights of 2019 ADRD Recommendations
- Central continuing themes across the 6 committees
- Biomarkers and Risk Profiles
- Type of Research (Basic, Translational, Clinical)
- Resource Infrastructure (biospecimens, bioinformatics, research tools, clinical trials)
- Training and Workforce Needs
- Nomenclature
- Evolving and new recommendations
- MED: trials for reversible causes; bridge science/clinical practice gap
- HD: policy relevant research and work force training in health disparities
- Nomenclature: proposes process to achieve common nomenclature
- VCID: Forward translation from basic science and reverse from clinical/trials
- LBD: expanded basic science recs (further testing hypothesis of propagation)
- FTD: Database resource
- Expansion in topics
- New emerging topics session
- TDP proteinopathy, Traumatic brain injury
- New emerging topics session
Natcher Auditorium, NIH Campus, Bethesda, MD
- Thursday, March 14, 2019
- 8:00am Welcoming Remarks and Introduction: Walter Koroshetz, MD, Director, NINDS
- 8:10am Perspective: Laura Gitlin NAPA Chair
- 8:20am Progress and Updating Research Recommendations Julie Schneider, MD, MS, Scientific Chair, and Rod Corriveau, PhD, NIH Summit Lead
- 8:40am Multiple Etiology Dementias -- Diagnosing in the 21st Century Chairs: David Knopman, MD, and Kate Possin, PhD
- 10:40am Health Disparities in AD/ADRD Chairs: Lisa Barnes, PhD and Hector Gonzalez, PhD
- 1:20pm Lewy Body Dementias Chair: Bradley Boeve, MD and Carol Lippa, MD
- 3:20pm Dementia Nomenclature Ronald Petersen, MD, PhD and Angela Taylor, BMus
- Friday, March 15, 2016
- 8:00am Vascular Contributions to Cognitive Impairment and Dementia Chairs: Donna Wilcock, PhD and Jeff Williamson, MD, MHS
- 10:10am Frontotemporal Lobar Degeneration Chairs: Leonard Petrucelli, PhD and Adam Boxer, MD, PhD
- 12:40pm Emerging Scientific Topics Chairs: Kristen Dams-O'Connor, PhD and Julie Schneider, MD, MS
- NEW - Emerging Topics:
- *TDP-43 Pathology in Common Dementias
- *TBI and AD/ADRD Risk
- >3 Hours Open Microphone Time
- NEW - Emerging Topics:
- 2:30pm Scientific Chair's Highlights and Cross-Cutting Themes Julie Schneider, MD, MS, Scientific Chair
Perspective on Prioritization of the Recommendations
- All recommendations in the the report represent very important research goals.
- Each committee was required to assign priorities starting at #1.
- However, for a research recommendation to be included in this report, it must be among the top priorities in its field.
- Timelines do not in any way reflect prioritization, but rather serve to guide planning and implementation logistics.
- Finally, ordering of sessions in no way reflects prioritization -- all sessions (MED, HD, NGO, LBD, FTLD, VCID, Nomenclature, Emerging Scientific Topics) are of equally high priority.
Multiple Separate Pathways AND Cross-Cutting Scientific Areas of Investigation Needed Across AD/ADRD
- Protein aggregation and neurodegeneration
- Protein clearance and degradation
- Vascular mechanisms, hypoxia, neurovascular unit, BBB
- Innate immune system and inflammation
- Axonal and synaptic injury and repair
- Circadian & sleep function/dysfunction
- Neurogenetics
- Systems and computational biology
Cross-Cutting Science -- CRITICAL NEED for AD and ADRD biomarkers/risk profiles
- Biomarkers that can discriminate underlying pathologies
- assessment of pre-symptomatic and symptomatic
- assessment of therapeutic response
- clinical screening
- pathology progression
- cognitive progression
- Need multiple biomarkers or markers of common processes
- Role for genetic, person-specific, multifactorial risk profiles
Cross-Cutting
- Health Disparities
- Chairs: Lisa Barnes, Hector González
- REC 1 -- Priority 1. Generate and/or improve cognitive assessment tools for populations facing AD/ADRD health disparities (1-3 y).
- REC 2 -- Priority 1. Increase availability and utilization of harmonized culturally- and linguistically-valid assessment tools within ongoing and newly generated studies of AD/ADRD and cognitive health intervention trials (1-3 y).
- Multiple Etiology Dementias
- Chairs: Dave Knopman, Kate Possin
- REC 1 -- Priority 1. Detect whether cognitive impairment is objectively present when a patient, care partner, or clinician reports cognitive, behavioral, or functional changes (3-7 y).
- REC 3 -- Priority 1. Advance basic & clinical research on common mechanisms of multi-etiology cognitive impairment and dementia (3-7 y).
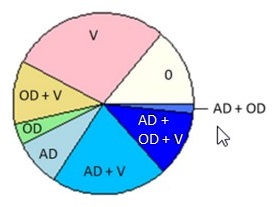
Brain Pathology is Common Late in Life; Mixed Pathologies are the Rule in Common Dementias
| Clinical Diagnosis No Cognitive Impairment ROS/MAP (N = 360) |
Clinical Diagnosis Probable AD Dementia ROS/MAP (N = 447) |
|---|---|
 |
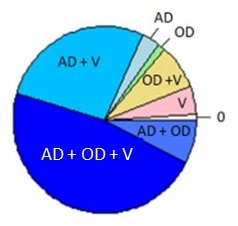 |
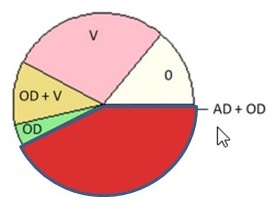
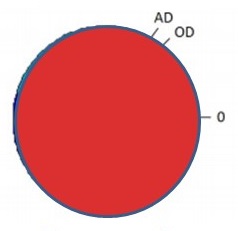
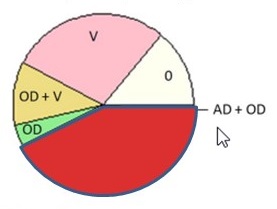
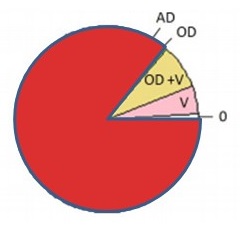
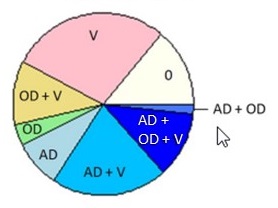
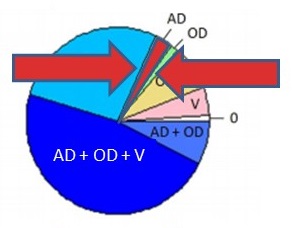
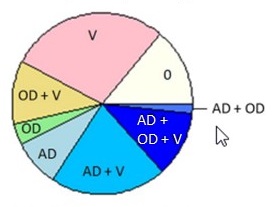
| PIE CHART KEY: AD = neuropathologic dx (plaques/tangles) OD = other neurodegenerative pathology TDP -- pathology beyond amygdala LB -- neocortical pathology V = vascular pathology -infarcts (macro/micro) -vessel disease (athero, arteriolosclerosis, CAA) 0 = no pathology detected Kapasi A, et al. Acta Neuropathol. 2017 Aug;134(2):171-186. |
|
Alzheimer's Disease (All)
| No Dementia Clinical Diagnosis = No Cognitive Impairment ROS/MAP (N = 360) |
Dementia Clinical Diagnosis = Probable AD Dementia ROS/MAP (N = 447) |
|---|---|
 |
 |
| Kapasi A, et al. Acta Neuropathol. 2017 Aug;134(2):171-186. | |
Alzheimer's Disease (Pathological)
| No Dementia Clinical Diagnosis = No Cognitive Impairment ROS/MAP (N = 360) |
Dementia Clinical Diagnosis = Probable AD Dementia ROS/MAP (N = 447) |
|---|---|
 |
 |
| Kapasi A, et al. Acta Neuropathol. 2017 Aug;134(2):171-186. | |
Alzheimer's Disease (Pure)
| No Dementia Clinical Diagnosis = No Cognitive Impairment ROS/MAP (N = 360) |
Dementia Clinical Diagnosis = Probable AD Dementia ROS/MAP (N = 447) |
|---|---|
 |
 |
| Kapasi A, et al. Acta Neuropathol. 2017 Aug;134(2):171-186. | |
Alzheimer's Disease (AD Dementia)
| No Dementia Clinical Diagnosis = No Cognitive Impairment ROS/MAP (N = 360) |
Dementia Clinical Diagnosis = Probable AD Dementia ROS/MAP (N = 447) |
|---|---|
 |
 |
| Kapasi A, et al. Acta Neuropathol. 2017 Aug;134(2):171-186. | |
What is Alzheimer's Disease?
- This Matters Because We Must Treat Individuals
- To Treat Individuals Clear Communication is Needed in:
- Training
- Research
- Clinical Practice
- Public Settings
 |
Dementia Nomenclature
- Chairs: Angela Taylor, Ron Petersen
- Focus Area 1: Dementia Nomenclature Working Groups
- REC 1 -- Priority 1. Form research, clinical practice and public stakeholder dementia nomenclature working groups (1-2 y).
- Focus Area 2: Integration and Interoperability of Dementia Nomenclature
- REC 2 -- Priority 1. Integrate and refine recommendations from the Research, Clinical Practice, and Public Stakeholder Working Groups into standardized, acceptable and accurate nomenclature that works across the spectrum of stakeholders (2-4 y).
- Dementia Nomenclature is now the topic of a NAPA Council Subcommittee led by Angela Taylor and Ron Petersen.
- LBD Chairs: Bradley Boeve, Carol Lippa
- REC 1 -- Priority 1. Prepare for and initiate clinical trials that aim to alleviate or slow the course of LBD symptoms, and delay or prevent the onset of disease (1-7 y).
- REC 5 -- Priority 1. Biomarker development (3-7 y).
- FTLD Chairs: Adam Boxer, Leonard Petrucelli
- REC 1 -- Priority 1. Clarify cellular and converging cellular mechanisms related to tau pathogenesis, C9orf72 hexanucleotide repeat expansions, GRN mutations, and other targets and pathways contributing to FTD neurodegeneration (2-10 y).
- REC 5 -- Priority 1. Develop FTD biomarkers for diagnosis, prediction and disease monitoring (2-7 y).
- VCID Chairs: Donna Wilcock, Jeff Williamson
- REC 1 -- Priority 1. Develop next‐generation experimental models and translational imaging methods for VCID (3-5 y).
- REC 4 -- Priority 1. Develop, validate and longitudinally track: (1) cognitive, physical, or other functional assessment components that indicate the presence of VCID; (2) VCID biomarkers, including when VCID is accompanied by pathological AD (3-5 y).
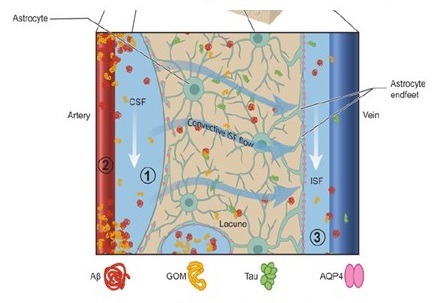
VCID: Lymphatic, Perivascular/Glymphatic
Perivascular spaces, glymphatic dysfunction, and small vessel disease
Humberto Mestre[1,2,*], Serhii Kostrikov[5,*], Rupal I. Mehta[1,2,3,4] and Maiken Nedergaard[1,2,5]
 |
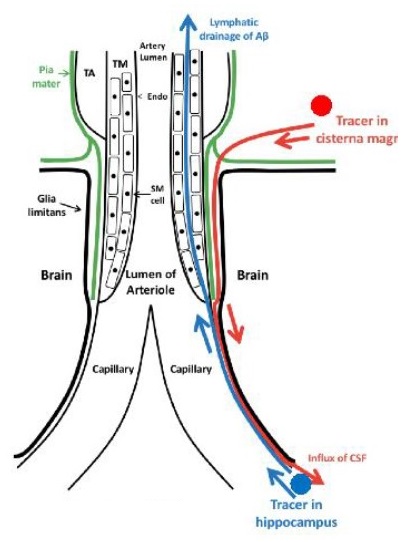 |
| Clinical Science (2017) 131 2257-2274 DOI: 10.1042/CS20160381 | Courtesy of R. Carare |
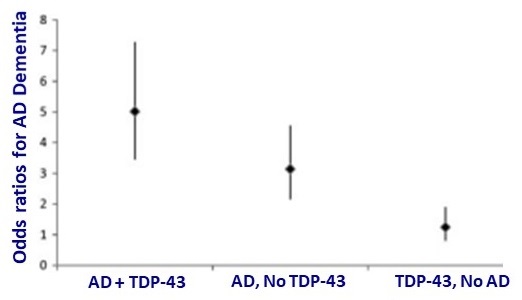
Emerging Science: TDP-43 in Common Dementias
 |
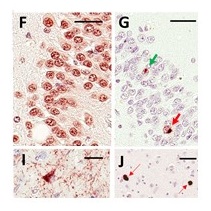 |
| Josephs KA, Lancet Neurol 2017;16(11):917-924 | Nelson et al., Brain, Volume 142, Issue 6, June 2019, Pages 1503--1527 |
Emerging Science: TBI and AD/ADRD Risk
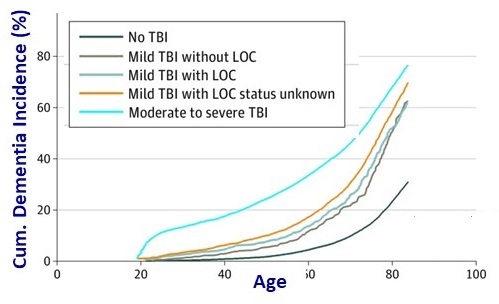 |
 |
| Barnes DE et al. JAMA Neurol 2018;75:1055 | James BD, Brain 2016;139(11):2983-2993 |
Emerging Scientific Topics
- Chairs: Kristen Dams-O'Connor, Julie Schneider
- REC 1 -- Priority 1. Develop biomarker/risk profiles to establish in vivo diagnostic criteria for TDP-43 pathology in persons without cognitive symptoms or amnestic syndromes (5-7 y).
- REC 5 -- Priority 1. Encourage cross-talk and interdisciplinary collaboration between TBI and dementia researchers (1-3 y).
All recommendations in the NINDS Council-Approved ADRD Summit 2019 Report represent critical national research pathways that are top scientific priorities toward the goal of achieving effective prevention and treatment of AD and ADRD
On behalf of the committee co-chairs, members, and with input from multiple stakeholder and the public, we respectively submit the ADRD Summit 2019 Report and Milestones to the NAPA Council.
Questions? Comments?
 |
| https://www.mindyourrisks.nih.gov/ |
