Friday, April 29, 2016
NINDS Alzheimer’s Disease Related Dementias Summit 2016
Walter J. Koroshetz, M.D.
Director, National Institute of Neurological Disorders and Stroke, NIH
Alzheimer's Disease-Related Dementias: Research Challenges and Opportunities
- Sponsored by:
- National Institute of Neurological Disorders and Stroke
- In partnership with:
- National Institute on Aging
- NIH Office of Disease Prevention
- FNIH
- Alzheimer's Association
- Accelerate Cure/Treatments for Alzheimer's Disease
- American Heart Associaton/American Stroke Association
- Association for Frontotemporal Degeneration
- Axovant
- BrightFocus Foundation
- Leaders Engage on Alzheimer's Disease
- http://ninds.nih.gov/ADRelatedDementias2016
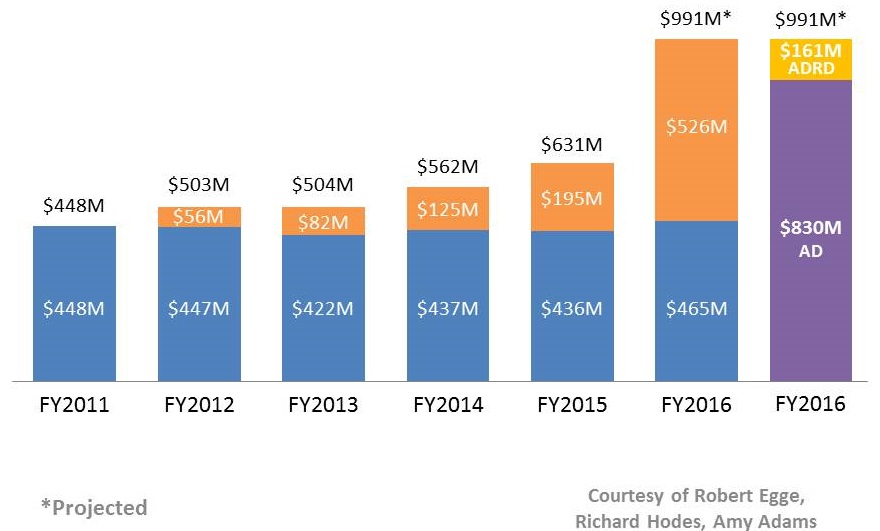
NIH Investment in the ADRDs
| 2012 | 2013 | 2014 | 2015 | |
|---|---|---|---|---|
| Frontotemporal Dementia (FTD) | $26 | $32 | $37 | $36 |
| Lewy Body Dementia | Not Determined | Not Determined | $15 | $15 |
| Vascular Cognitive Impairment/Dementia | Not Determined | Not Determined | $45 | $72 |
| ADRDs | Not Determined | Not Determined | Not Determined | $120 |
| Source: https://report.nih.gov/categorical_spending.aspx | ||||
AD/ADRD Research Increases at NIH
 |
NINDS Participation in Initiatives Responsive to ADRD 2013
- RFA-NS-16-019: Small Vessel Vascular Contributions to Cognitive Impairment & Dementia (VCID) Biomarkers Consortium: Coordinating Center (U24).
- RFA-NS-16-020: Small Vessel Vascular Contributions to Cognitive Impairment & Dementia (VCID) Biomarkers Development Projects (UH2/UH3).
- RFA-NS-16-021: Mechanistic Basis of Diffuse White Matter Disease in Vascular Contributions to Cognitive Impairment & Dementia (VCID)(R01).
- RFA-NS-16-022: Biomarkers for the Lewy Body Dementias (U01).
- RFA-NS-16-023: Center without Walls for the Identification & Validation of Molecular Mechanisms Contributing to Tau Pathogenesis & Associated Neurodegeneration in Frontotemporal Degeneration (U54).
- PAR-15-349 (led by NIA, NINDS joined): Health Disparities & AD (R01).
Underway (NIA/NINDS Collaboration): M2OVE AD Consortium
- Established by RFA-AG-15-010: Interdisciplinary Research to Understand the Vascular Contributions to Alzheimer’s Disease.
- 2016 ADRD Summit Planning
- 6 months by >80 scientists, physicians, and administrators
- Scientific Chair: David Holtzman
- NIH/NINDS Summit Lead: Rod Corriveau
- Steering Committee: Karen Chen, Tom Montine, Bruce Obviagele, Ron Petersen, Tony Phelps
- Session Committees
- Overarching: MED; Health Disparities; NGO
- Disease-Specific: LBD; FTD; VCID
- ADRD Summit Goals
- Committees present rationale for revisions to the research recommendations and timelines
- Provoke discussion among group experts
- Solicit feedback and opinions from audience
The ADRD 2016 Recommendations and Report will be finalized and informed by input at this Summit
A Major Question to be AddressedRegarding AD and ADRD
How can the various disorders of cognition be compared and assessed and why?
Diagram
- Clinical Spectrum CN - MCI - Dementia [Clinical]
- Aß [Pathology]
- TAU [Pathology]
- Other [Pathology]
- Vascular Disease [Pathology]
- Alpha Synuclein [Pathology]
- TDP-43 [Pathology]
Diagram
- Clinical Spectrum CN - MCI - Dementia [Clinical]
- Aß [Pathology]
- PET/CSF [Biomarker]
- Rx? [Therapy]
- PET/CSF [Biomarker]
- TAU [Pathology]
- PET/CSF [Biomarker]
- Rx? [Therapy]
- PET/CSF [Biomarker]
- Other [Pathology]
- ? [Biomarker]
- Rx? [Therapy]
- ? [Biomarker]
- Vascular Disease [Pathology]
- MRI [Biomarker]
- Rx? [Therapy]
- MRI [Biomarker]
- Alpha Synuclein [Pathology]
- CSF? [Biomarker]
- Rx? [Therapy]
- CSF? [Biomarker]
- TDP-43 [Pathology]
- CSF? [Biomarker]
- Rx? [Therapy]
- CSF? [Biomarker]
- Aß [Pathology]
MED Chairs: Dave Knopman, David Bennett
Diagnosing Dementia in the 21st Century
- Detect cognitive impairment when patient or relative voices a concern to health care providers
Aspirational versus Operational
Session 1 Prioritized Recommendations -- Multiple Etiology Dementias
- Improving Diagnostic Skills in the Community
- Detect cognitive impairment when patient or relative voices a concern to health care providers (5-7 y; 2017)
- Develop differential diagnosis of symptomatic cognitive impairment (5-7 y; 2017)
- Develop diagnostics/biomarkers in asymptomatic individuals (3-5 y for improved biomarkers for non-AD dementias; 2017)
- Basic & Clinical Research in Interactions between Dementia Pathophysiologies
- Promote basic and clinical research in multietiology dementia (5-7 y; 2017)
- Determining the Role for Screening for Cognitive Dysfunction
- Screen for clinically relevant cognitive impairment in the absence of a cognitive complaint (indefinite; 2017)
- Revisiting the Nosology of Cognitive Impairment in Late Life
- Address the inconsistent nomenclature in dementia research and care (3 y; 2017)
NGO Chairs: Susan Dickinson, Howard Fillit
Catalyzing Research Through Unique Programs and Partnerships
- Establish more effective communication between NIH and the NGOs on activities and progress toward the ADRD goals in the off-years between the triennial ADRD Research Summits
Session 2 Prioritized Recommendations -- Non-Governmental Organizations
- Catalyzing Research through Unique Programs and Partnerships
- Establish more effective communication between NIH and the NGOs on activities and progress toward the ADRD goals in the off-years between the triennial ADRD Research Summits (ongoing; 2016)
- Nomenclature Standards when Discussing Dementia
- Organize a working group of dementia stakeholders, including founding partnerships with disparities communities, to review the current nomenclature used in public awareness, clinical care and research, and to propose strategies to help advance early differential diagnosis and the understanding of dementia and its underlying causes (ongoing; 2017)
Health Disparities Chair: Jennifer Manly
Treatment and Prevention Strategies
- Assess epidemiology and mechanistic pathways of disparities in health burden of ADRDs (1-6 y; 2016)
Session 3 Prioritized Recommendations -- Health Disparities
- Treatment and Prevention Strategies
- Assess epidemiology and mechanistic pathways of disparities in health burden of ADRDs (1-6 y; 2016)
- Enhance the design of trials of vascular health interventions to improve their application to aging diverse populations (2-7 y; 2017)
- Monitoring Changes in ADRD Disparities
- Develop a system to monitor the magnitude and trends in disparities in incidence of ADRDs (2-8 y; 2017)
- Assessment
- Improve tools for assessment of disparities in risks, preclinical disease characteristics, and costs of ADRD among disparities populations by leveraging existing data and cohorts, designing targeted studies, and using advanced psychometric analyses for improving tools for assessment of disparities in risks, preclinical disease characteristics, and costs of ADRD among disparities populations (1-4 y; 2016)
- Increase utilization of culturally- and linguistically-appropriate assessment tools within ongoing and newly generated studies of ADRDs and vascular health intervention trials (1-2 y; 2016)
- Community Partnerships, Recruitment, and Retention
- Generate an AD/ADRD Health Disparities Task Force that is specifically designed to provide guidance and expertise for community engagement, study design, recruitment and retention into sites to ensure recruitment of diverse populations into newly generated epidemiological studies and clinical trials (1-2 y; 2016)
- Develop novel community engagement and outreach methods and identify existing methods to facilitate engagement, understanding and partnership with disparities populations (1-2 y; 2016)
Special Joint Session: Nomenclature
Angela Taylor, Dave Knopman
- LBD Chairs: Karen Marder, Dennis Dickson
- Initiate clinical trials for motor and non-motor manifestations of LBD using existing and newly developed symptomatic therapies
- FTLD Chairs: Michael Hutton, William Seeley
- Basic: Clarify the mechanism of tau pathogenesis and associated neurodegeneration
- Clinical: Clarify the mechanism of tau pathogenesis and associated neurodegeneration
- VCID Chairs: Steve Greenberg, Tom Carmichael
- Basic: Develop next generation experimental models & translational imaging methods for VCID
- Clinical: Develop and validate longitudinally tracked noninvasive biomarkers of key vascular processes related to VCID
Session 4 -- Prioritized Recommendations: LBD
- Establish Longitudinal Cohorts with Common Measures, Culminating in Autopsy Studies
- Initiate clinical trials for motor and non-motor manifestations of LBD using existing and newly developed symptomatic therapies (1-3 y; 2016).
- Create longitudinal clinical, biological, and imaging resources for DLB and PDD from the earliest stages to autopsy to improve early detection and diagnosis (5-10 y; 2016).
- Discover Disease Mechanisms through Brain Mapping and Genetics
- Characterize and catalogue LBD pathology in the CNS and PNS using genomics, expression arrays, metabolomics, and proteomics (3-5 y; 2018).
- Identify novel common and rare genetic variants, epigenetic changes, and environmental influences that impact risk for and clinical features of LBD (5-7 y; 2016)
- Develop and Validate Biological and Imaging Biomarkers
- Develop validated imaging approaches to 1) enhance the differential diagnostic accuracy of LBD 2) detect latent and prodromal LBD 3) monitor disease progression (3-5 y; 2016)
- Develop biomarkers for LBD, and the contribution of Alzheimer’s and other pathologic changes using new (Rec. 2) or existing longitudinal studies (5-7 y; 2017)
- Model Disease Processes to Develop Potential Symptomatic and Disease Modifying Therapies
- Create new animal, cellular, and in vitro models focused on a-synuclein and AD pathophysiologic processes in LBD to capture clinical heterogeneity. (7-10 y; 2017)
- Develop disease-modifying interventions for LBD based on discovering biomarkers, molecular targets, and genetic and environmental modifiers (7-10 y; 2018)
Session 5 Prioritized Recommendations -- Frontotemporal Lobar Degeneration
- Basic Science: Pathogenesis and Toxicity
- Clarify the mechanism of tau pathogenesis and associated neurodegeneration (2-5 y; 2016/2017)
- Determine the molecular basis for C9ORF72 expansion- and GRN mutation-related neurodegeneration (3-10 y; 2016/2017)
- Determine the mechanism of TDP-43 and FUS pathogenesis and toxicity (3-10 y; 2016/2017)
- Develop better FTD in vivo and cell-based model systems (1-3 y; 2016/2017)
- Clinical Science
- Expand efforts to genotype patients with FTD and identify new genes (1-3 y; 2017/2018)
- Develop FTD biomarkers for diagnosis and disease progression (3-7 y; 2017/2018)
- Create an international FTD clinical trial network (1-3 y; 2017/2018)
- Understand phenotypic heterogeneity and natural history (>10 y; in progress)
Session 6 Prioritized Recommendations -- VCID: Small Vessel Disease & AD/Vascular Interactions
- Basic Mechanisms and Experimental Models
- Develop next generation experimental models and translational imaging methods for VCID (1-7 y; 2016)
- Encourage basic science that investigates impact of aging, AD pathology, and genes on peri-/para-vascular clearance mechanisms, the neurovascular unit, and cerebrovascular function (3-7 y; 2018)
- Encourage basic science investigating effect of vascular risk factors/genes and atherosclerosis on AD-related neurodegeneration (3-7 y; 2018)
- Human-Based Studies
- Develop (1-3 y; 2016) and validate (3-7 y; 2018) longitudinally tracked noninvasive markers of key vascular processes related to VCID
- Determine interrelationships (cross-sectional and longitudinal) among aging, vascular disease & risk factors, resilience factors, genetic variants, amyloid, tau and neurodegeneration (3-7 y; 2018)
- Identify lifestyle and vascular interventions to treat, prevent or postpone VCID (7-10 y; 2022)
Mind Your Risks for Healthy Brain Aging
|
|
 |
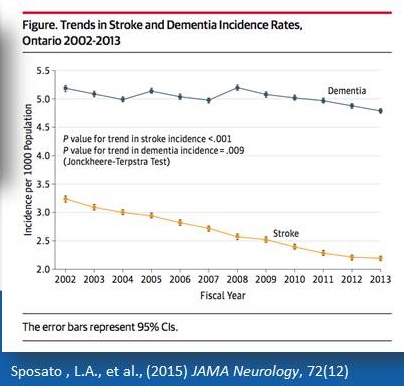 |
Mind Your Risks Campaign
 |
| https://mindyourrisks.nih.gov/ |
Conclusion
- The summits, in particular this ADRD Summit, determine the direction of the research questions which translate directly into funding opportunities
Questions?
Walter J. Koroshetz, M.D.
Director
National Institute of Neurological Disorders and Stroke
Email: koroshetzw@ninds.nih.gov
Website: http://www.ninds.nih.gov/
Follow me @NINDSdirector
