Monday, January 25, 2021
Printer Friendly Version in PDF Format (9 PDF pages)
Nomenclature
Ronald C. Petersen, PhD, MD
Mayo Clinic
Rochester, MN
Angela Taylor
Lewy Body Dementia Association
Nomenclature Initiative
- Progress on initiative
- Asymptomatic people with biomarkers
Nomenclature Initiative
- Progress on initiative
- Asymptomatic people with biomarkers
Nomenclature -- Words Matter
- Nomenclature issues very important
- Cut across all spheres of investigation
- Science/Research
- Clinical Care
- Public Stakeholders
- Government agencies
- Advocacy groups
- Research participants
- Under-represented groups
Confusion in the field on terminology
- Alzheimer's Disease
- Frontotemporal Degeneration
- Dementia with Lewy Bodies
- Vascular cognitive impairment-dementia
- Mixed dementias
Working Groups
- Research: Sandy Weintraub, PhD, NW University
- Clinical: Marwan Sabbagh, MD, Cleveland Clinic, Las Vegas
- Public Stakeholders: Jason Karlawish, MD, U of Pennsylvania
- Facilitator: RAND Corporation
Nomenclature -- Implications for Research
- Science/Research: Must be precise
- Clinicians: Must translate science to patients and vice versa
- Public stakeholders: Stigma, willingness to participate in research
- Sensitivity in under-represented groups
NOMENCLATURE FRAMEWORK
- Level 1: Umbrella term for neurological disorders causing cognitive impairment and dementia
- Each Working Group recommends an umbrella term
- Level 2: Age of onset; e.g.,
- Over/under 60
- Mid-life, late-life, oldest old
- Young onset, late onset
- Level 3: Continuum of cognitive impairment from none to severe (aka dementia); e.g.,
- None, slight, mild, moderate severe
- Stage 0-5
- None, MCI, mild, moderate, severe dementia
- mid/major neurocognitive disorder
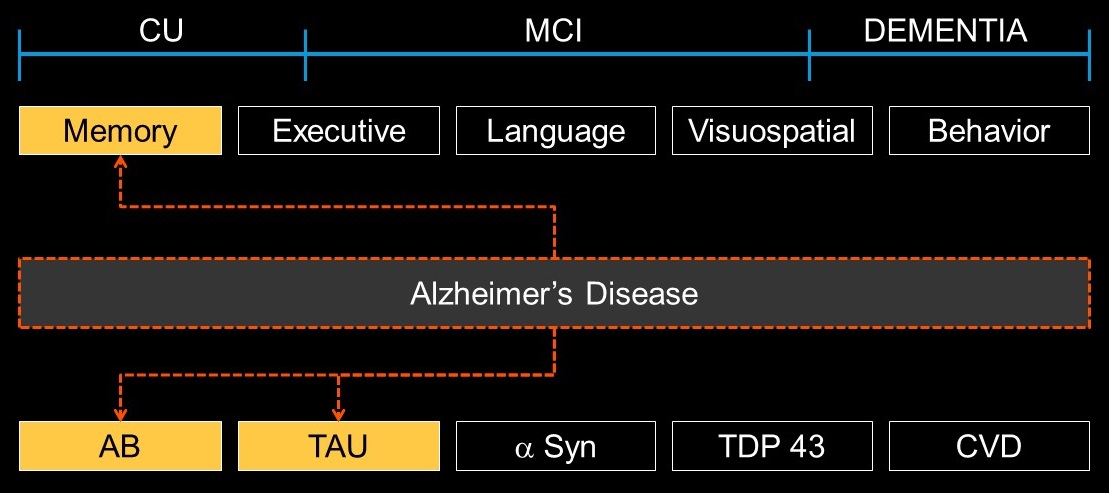
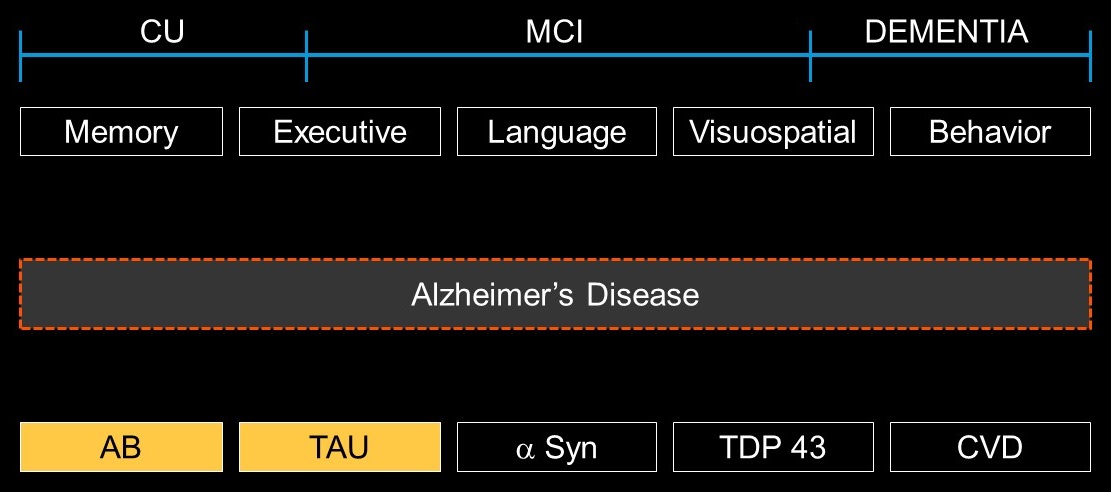
- Level 4: Syndromic description
- Descriptive, with cognitive and behavioral domains; underlying etiology not specified. May include adjectives (memory, language, executive, behavioral) and noun (disorder, spectrum, continuum) or combinations
- Level 5: Neuropathologies; e.g.,
- amyloid
- tau
- alpha synuclein
- TDP-43
- vascular
Nomenclature Initiative
- Progress on initiative
- Asymptomatic people with biomarkers
Old Conception of Alzheimer's Disease
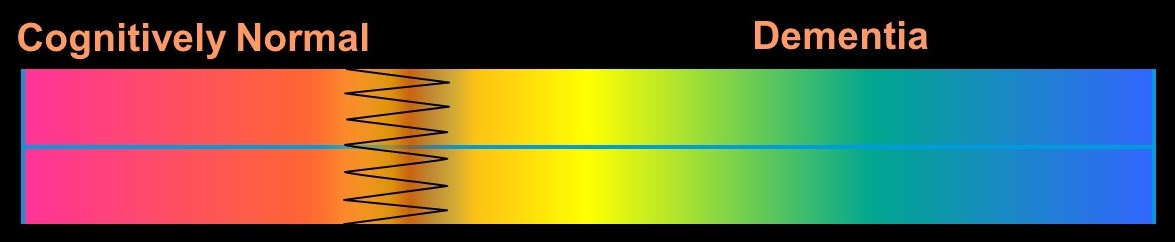 |
NINCDS-ADRDA Criteria 1984
Alzheimer's Disease as a Clinical -- Pathological Entity
Alzheimer's Disease
- 1984 -- NINCDS-ADRDA Criteria
- Clinical-Pathological definition
- 2011 -- NIA-AA Criteria
- Clinical syndrome with biomarkers for amyloid and neurodegeneration
- 2018 -- NIA-AA Framework
- Alzheimer's disease as a biological entity defined by positive biomarkers for amyloid and tau
- Clinical Spectra Independent
SEVERITY
 |
SEVERITY
 |
Plans for Future
- Work groups continue to meet
- Research/Science
- Clinical practice
- Public stakeholders
- Present research implications to NIH at workshop on February 16, 2021
Dementia Nomenclature Initiative Winter 2021 Workshop
- Goal: Update NIH and others on dementia nomenclature initiative progress; elicit feedback on working plan.
- Challenges of Current Nomenclature for Research, Clinical Care, and Public Communication
- Overview of the Dementia Nomenclature Initiative
- Report from the Research Working Group
- Working Group Updates: Clinical Care and Public Stakeholder
- Panel Discussion: Through the Lens of Health Disparities
Thank you
Questions?

