Monday, January 25, 2021
Printer Friendly Version in PDF Format (17 PDF pages)
An Update on NIA's Clinical Research Recruitment & Retention Initiatives
Holly A. Massett, PhD
Senior Advisor, Clinical Research Recruitment & Engagement
Division of Extramural Activities, NIA
Minority recruitment initiatives
- Clinical Research Operations and Management System (CROMS)
- Increase Awareness of AD/ADRD Clinical Research
- Materials Development
- Online tool: Outreach Pro
- Community Practitioner Research Network
Clinical Research Operations and Management System (CROMS)
Clinical Research Operations & Management System (CROMS)
- CROMS Purpose:
- Provide institute-wide informatics capability to track, report, and manage NIA's clinical enrollment data, activities, and grant portfolio in near real-time
- All NIA clinical trials and studies that enroll human subjects
Why an NIA-wide clinical research IT system?
- Institutional Accountability for Research
- Operational Portfolio Management
- Data Management
Timeline
- Project began in July 2020
- Competitively bid
- Awarded to Digital Infuzion (experience with NIAID, NCI, NIDCR)
- Currently on schedule, despite the many challenges COVID posed
- First fully operational release scheduled for July 2021
- Will address AD/ADRD clinical trials
- Following, will add remaining trials and studies in the NIA portfolio
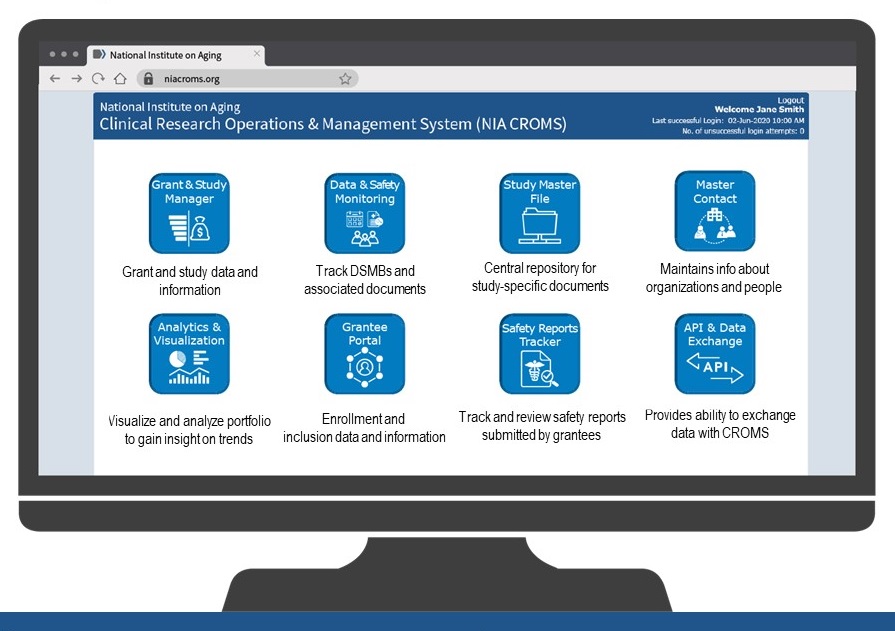
CROMS Components & Features
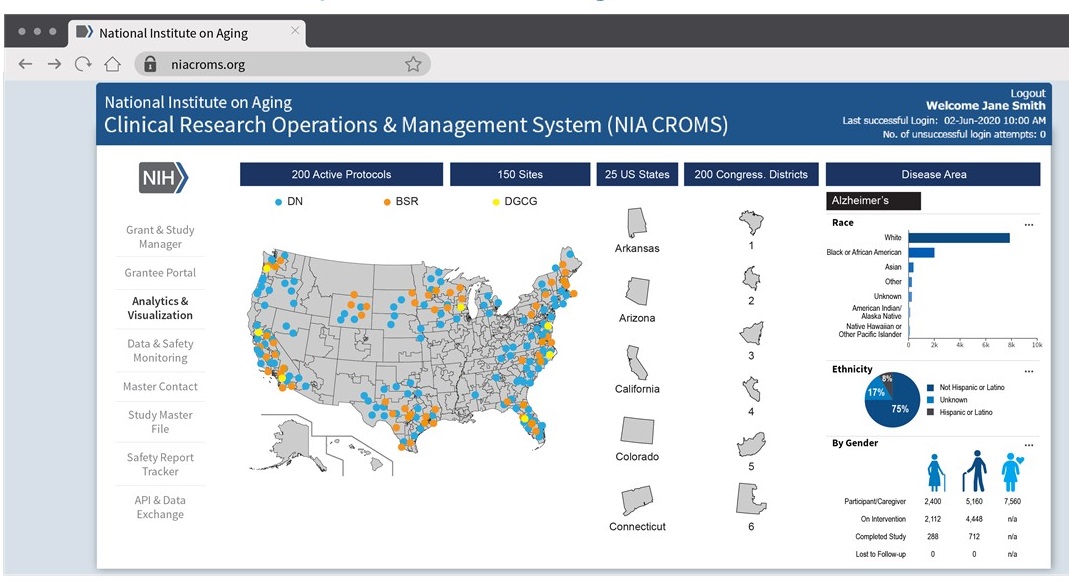
Dashboard Mockups -- Overarching View
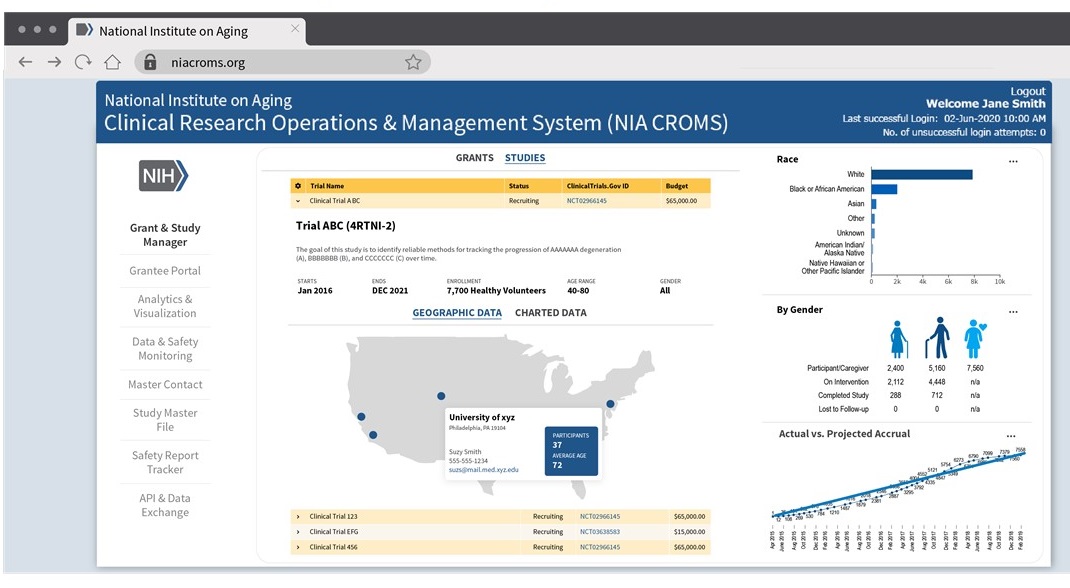
Dashboard Mockups -- Study-Specifics
Dashboard Mockup -- Site Performance
Increase Awareness of AD/ADRD Clinical Research
Well-tested Community Outreach Materials for Minority Populations
Objective
- Develop AD/ADRD clinical research materials specific to the language and cultural needs of minority populations
- Design of a suite of recruitment outreach materials for minority groups:
- African Americans (2019)
- Latinos (2020)
- Asian American & Pacific Islanders (2021)
- Native Americans (2022)
- Materials can be tailored and branded by local community organizations
Process follows health communication best practices
- Outreach Materials Development and Testing
- Conduct qualitative and quantitative research to develop and refine a suite of outreach materials tailored to increase the participation and retention of minority populations in AD/ADRD clinical studies.
- Materials include: Leave-Behinds, Fact Sheets, Image Library, Videos, Social Media Posts, and Motion Graphics
Creatives
 |
 |
| * Currently working with Asian American and Pacific Islander (AAPI) stakeholders to develop a third suite by September 2021 |
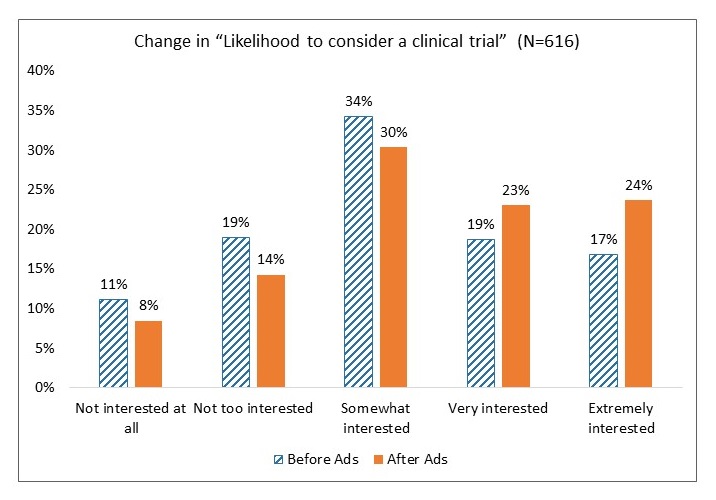
Moving the Needle: African American Research Results
 |
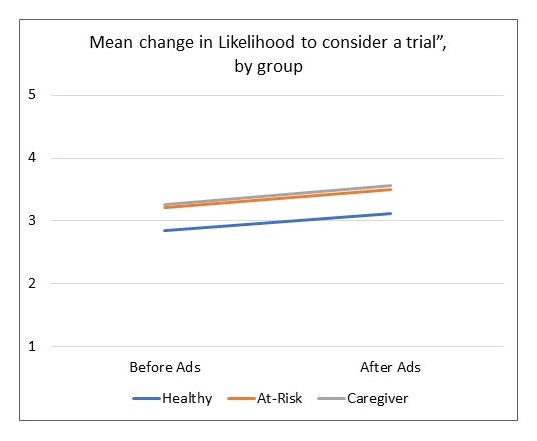 |
| All group increases are significant at p < .001 |
Moving the Needle: Latino Research Results
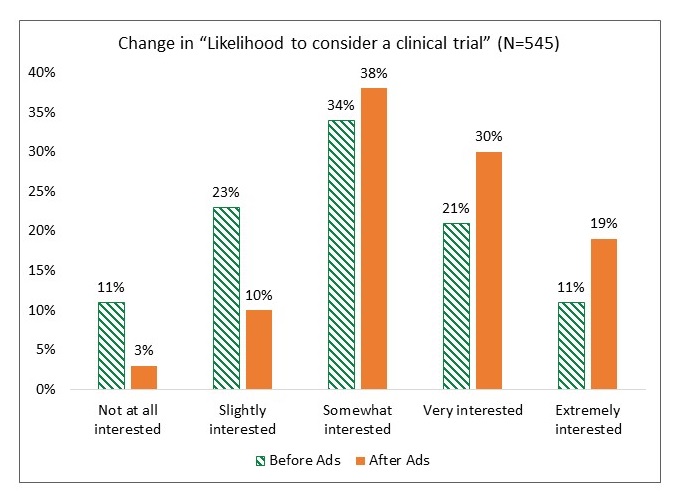 |
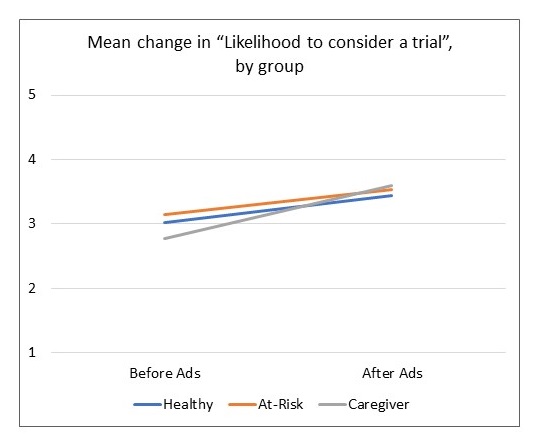 |
| All group increases are significant at p < .001 |
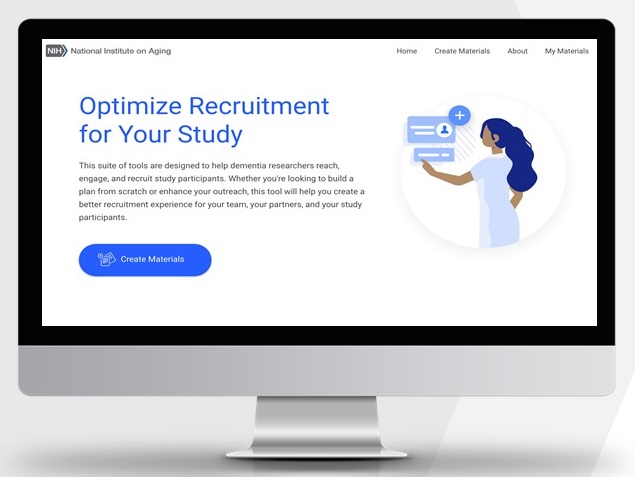
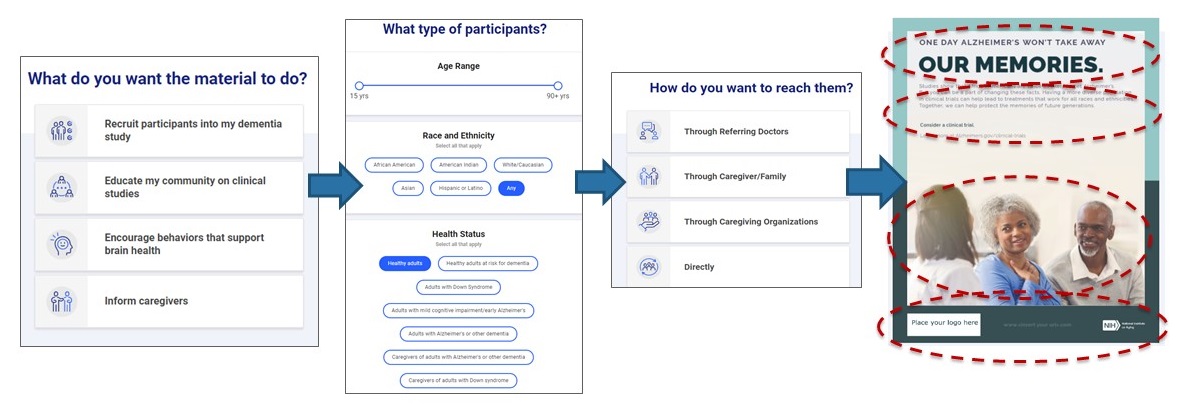
Outreach Pro: NIA's Tool for Clinical Studies Recruitment and Education
Outreach Pro: NIA's Online Tool for Clinical Studies Recruitment and Education
Objective
- Provide community stakeholders access to well-tested, evidence-based AD/ADRD clinical research materials and strategies in an easy-to-use web-based communication tool
- Users answer a brief set of questions specific to their audiences of interest
- Tool produces a “package” of tailored materials and strategies that can be branded locally for the end-user's community.
Outreach Pro
- Online tool allows the research community to access, adapt, and personalize the materials that NIA has developed and tested for underrepresented communities through a user-friendly step-by-step process:
Plans for a Community Practitioner Research Network
NIA Substantial Investments to Support Clinical Research
- Including:
- Alzheimer's Clinical Trials Consortium (ACTC)
- Trial Ready Cohort for the Prevention of Alzheimer's Dementia (TRC-PAD)
- Alzheimer's Disease Research Centers (ADRCs)
- Resource Centers for Minority Aging Research (RCMAR)
- NIA Imbedded Pragmatic AD/ADRD Clinical Trials (IMPACT) Collaboratory
- R24: Examining Diversity, Recruitment and Retention in Aging Research
- Others...
Geographic Distribution of AD/ADRD by Minority Populations
ADRD County-Level Prevalence
Blacks, Hispanic/Latinos, Asian Americans, and North American Natives
Medicare Fee for Service, 2016 |
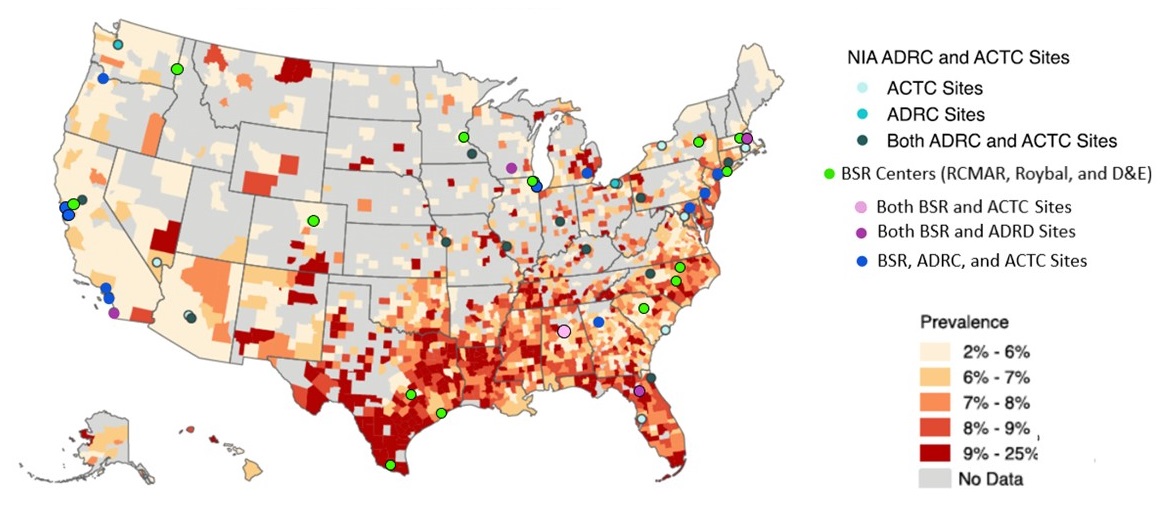 |
| Map Generated by National Alzheimer's Disease Index Powered by USAgainstAlzheimer's |
Question: How can we better enroll minorities?
- We do not adequately recruit minority participants commensurate to the disease impact
- NIA has programs and research sites across the U.S., though with potential geographic 'gaps' where minorities affected by AD live but have limited/no infrastructure
- Literature continues to report on AD/ADRD clinical research challenges with accessing minority community participation
Conclusion: Our current efforts and resources are important and necessary, but insufficient
- No matter how hard our current system works to address the inclusion of minorities, it will be difficult to address the gaps
Premise: Go to where the participants live vs. asking participants to come to us
- Build a community infrastructure of PIs and sites in minority communities with high rates of AD/ADRD
- Select sites where doctors/staff have a research interest and relevant expertise
- Nurture and incentivize community sites directly to conduct research
Complement our current resources:
- Academic Medical Centers
- Community Practitioner Sites
- Programs and Registries
NIH ICs with Community-Based Research Networks
| IC |
Network |
Description |
| NEI |
DRCR Retina Network |
160 physician offices with over 500 physicians throughout the United States and Canada to conduct high quality, collaborative clinical research |
| NIDCR |
National Dental Practice-Based Research Network (PBRN) |
Over 6,500 dental clinicians conduct research on patient health outcomes, patient satisfaction, or the efficiency of the daily delivery of care. |
| NIDA |
National Drug Abuse Treatment Clinical Trials Network (CTN) |
Treatment researchers and community-based substance abuse service providers cooperatively develop, validate, refine, and deliver new treatment options to patients. |
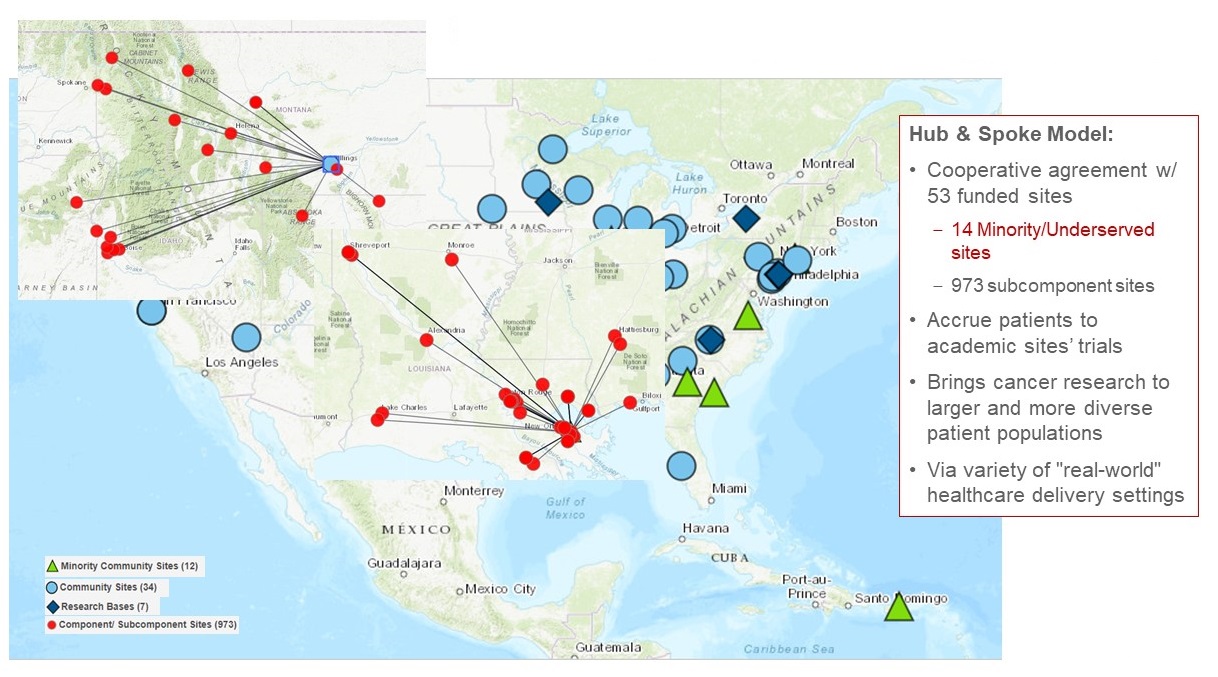
| NCI |
NCI Community Oncology Research Program (NCORP) |
Comprised of 7 Research Bases and 46 Community Sites--14 designated as Minority/Underserved Community Sites--that accrue individuals to NCI-approved cancer clinical trials and research studies. |
| NHLBI |
Pulmonary Trials Cooperative (PTC) |
Provides a framework for research leaders to work with a pool of major medical centers and community-based medical providers. |
| NINDS |
Strategies to Innovate EmeRgENcy Care Clinical Trials Network (SIREN) |
Enrollment is conducted in diverse patient populations and practice environments including academic medical centers and community ER hospitals to ensure that results can be generalized. |
| NINDS |
StrokeNet |
~500 US hospitals conduct small and large clinical trials and research studies to advance acute stroke treatment, stroke prevention, and recovery |
| NIAID |
HIV/AIDS Clinical Trials Networks |
Four publicly-funded network collaborations of community and academic sites around the world to address NIAID's five HIV/AIDS scientific priorities |
NCI Community Oncology Research Program (NCORP)
NCI Minority-based Sites' Accrual Contribution
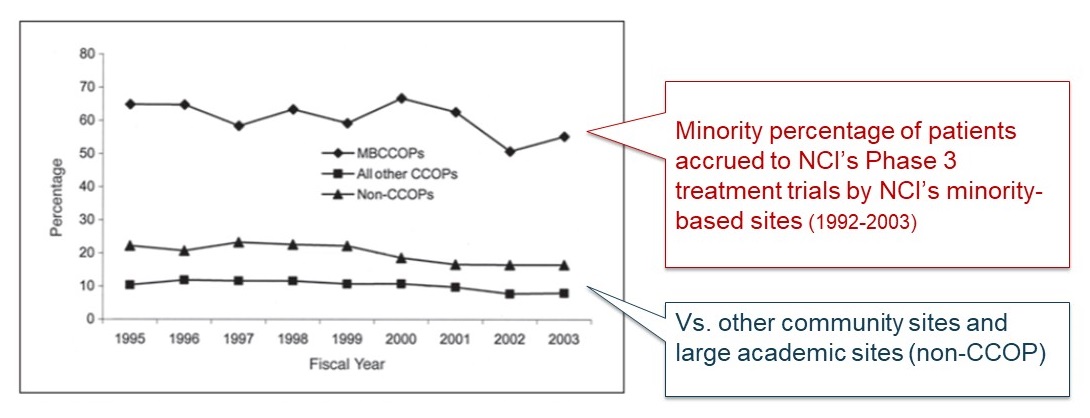 |
Fig 2. Minority percentage of patients accrued to cooperative group treatment trials by Minority-Based Community Clinical Oncology Programs (MBCCOPs), other Community Clinical Oncology Programs (CCOPs), and non-CCOP members/affiliates. Non-CCOP members of cooperative groups include academic medical centers, National Cancer Institute--designated cancer centers, and other large research institutions. Affiliates include community hospitals and oncology practices that affiliate with cooperative groups through "main memnber" institutions.
J Clin Oncol 23:5247-5254, 2004. |
Next Steps
- Internal NIA Community Practitioner Research Network Committee
- Meeting regularly to identify needs, define vision, propose strategy
- Input from the Field: One-day Community Practitioner Research Network Symposium
- Virtual meeting in early April 2021 (tbd), 12-5 pm EST
- Invite stakeholders from around country to participate in a discussion exploring potential models for a NIA community practitioner research network
Thank you!
Questions?
National Institute on Aging
https://www.nia.nih.gov/
Return to
National Alzheimer's Project Act Home Page
Advisory Council on Alzheimer's Research, Care, and Services Page
Advisory Council on Alzheimer's Research, Care, and Services Meetings Page