Monday, January 27, 2020
Printer Friendly Version in PDF Format (8 PDF pages)
Congressionally Directed Medical Research Programs: Peer Reviewed Alzheimer's Research Program Overview
Anthony M. Pacifico, Ph.D.
Program Manager
The views expressed in this presentation are those of the author and may not reflect the official policy or position of the Department of the Army, Department of Defense, or the U.S. Government.
Program Overview: Introduction
- History: Initiated in 2011 to address the long-term consequences of traumatic brain injury (TBI) as they pertain to Alzheimer's disease (AD). In fiscal year 2016 (FY16), the program was expanded to include research on AD-related dementias (ADRD) as they pertain to TBI.
- Vision: To address the long-term consequences of traumatic brain injury as they pertain to Alzheimer's disease and Alzheimer's disease-related dementias.
- Mission: The PRARP's mission is devoted to (1) understanding the association between TBI and AD/ADRD and (2) reducing the burden on affected individuals and caregivers, especially in the military and Veteran communities.
Defining the Scope of the Problem
- Health Impact of AD
- U.S. -- Prevalence of AD is between 2.6 and 5.4 million cases, depending on the study; prevalence increases with age[1,2]
- Globally -- Estimated 27 million cases[3]; 5% of European population (37 million!)[4]
- Diagnosis rate is hard to estimate; cases are often missed[1]
- Centers for Disease Control and Prevention (CDC) reports 94,000 deaths in 2014 (6th leading cause of death)[5]
- Estimated U.S. health care costs in 2010: $157B to $236B[2,6]
- No cure; treatments address only symptoms
- Brookmeyer, et al. 2011. National estimates of the prevalence of Alzheimer's disease in the United States. Alzheimers Dement. Jan;7(1):61-73. doi: 10.1016/j.jalz.2010.11.007.
- Alzheimer's Association. 2016. 2016 Alzheimer's disease facts and figures. Alzheimers Dement. 2016 Apr;12(4):459-509.
- Brookmeyer, et al. 2007. Forecasting the global burden of Alzheimer's disease. Alzheimers Dement. Jul;3(3):186-91. doi: 10.1016/j.jalz.2007.04.381.
- Niu et al. 2017. Prevalence and incidence of Alzheimer's disease in Europe: A meta-analysis. Neurologia. 2017 Oct;32(8):523-532
- http://www.cdc.gov/nchs/fastats/alzheimers.htm
- Hurd, et al. 2013. Monetary Costs of Dementia in the United States. N Engl J Med. April 4; 368(14):1326-1334.
- Health Impact of TBI
- Defense and Veterans Brain Injury Center (DVBIC) reported roughly 380,000 cases since 2000[1]
- In 2014, there were approximately 2.87 million TBI Emergency Department (ED) visits/hospitalizations in the United States with 57,000 deaths[2]
- Rates of TBI-related ED visits per 100,000 population were highest among older adults aged > 75 years (1,682.0), young children aged 0-4 years (1,618.6), and individuals 15-24 years (1,010.1)[2]
- CDC-reported cost estimate for domestic TBI medical costs in 2010 was $76.3B[3]
- http://dvbic.dcoe.mil/dod-worldwide-numbers-tbi
- http://https://www.cdc.gov/traumaticbraininjury/data/index.html
- https://www.cdc.gov/cdcgrandrounds/pdf/grtbi20sep2011.pdf
- Epidemiological Data Associating TBI with Dementia
- Variety of studies, variety of outcomes
- At least 20 published studies since 1984; odds ratios range from 1 to 14
- Institute of Medicine report cites hazards ratio values based on length of loss of consciousness/post-traumatic amnesia at 2.3 for moderate and 4.5 for severe in Veterans with non-penetrating head injury[1]
- Yaffe et al. (2019) report a hazard ratio (HR) of 1.64 in female veterans (n=110K) with one or more additional comorbidities (PTSD or depression) increasing the HR up to 2.42[2]; similar but higher HR values for men (up to 3.8 for mod/severe TBI)[3]
- Confounders include: self report, lack of robust autopsy data, age at injury, post-traumatic interval, operational TBI definitions at the time of the study (e.g., use of ever versus never), use of ICD codes...
- Plassman BL, et al. 2000. Documented head injury in early adulthood and risk of Alzheimer's disease and other dementia. Neurology. Oct 24;55(8):1158-1166.
- Yaffe K, et al. 2019. Military-related risk factors in female veterans and risk of dementia. Neurology. Jan 15; 92(3):205-211.
- Barnes, et al. 2018. Association of Mild Traumatic Brain Injury With and Without Loss of Consciousness With Dementia in US Military Veterans. JAMA Neurol. 2018 Sep 1;75(9):1055-1061.
- Common Symptoms
- Memory disorders
- Behavioral Symptoms
- Executive functioning deficits
- TBI-AD/ADRD Risk Factors (or Comorbidities)
- Genetics
- Sleep, activity, nutrition
- Hypertension, diabetes
- Gender
- Age
- Research Tools Used for Both
- Autopsy
- Radioimaging (plaques, Tau)
- Magnetic resonance imaging (MRI) (functional, diffusion tensor imaging, structural [hippocampal volume])
- Cerebral spinal fluid (Aβ42, Tau, and phospho-Tau), Blood, Saliva
NIH, VA, and PRARP Governmental Funding Landscape FY11-FY19 for AD/ADRD
- PRARP Total Congressional appropriations: $123M
- PRARP "changes its course" yearly in order to leverage larger investments.
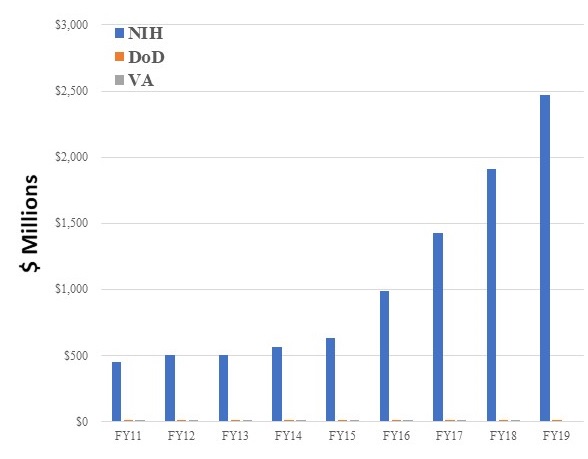 |
| NIH | DoD^ | VA | |
|---|---|---|---|
| FY11 | $448M | $15M | $12.4M |
| FY12 | $503M | $12M | $13.4M |
| FY13 | $504M | $12M | $11.6M |
| FY14 | $562M | $12M | $11.2M |
| FY15 | $631M | $12M | $13.7M |
| FY16 | $986M | $15M | $12.8M |
| FY17 | $1,423M | $15M | $11.8M |
| FY18 | $1,911M | $15M | $11.9M |
| FY19 | $2,468M* | $15M | N/A |
| *Estimated (https://report.nih.gov/categorical_spending.aspx) ^Before management and withhold costs. |
|||
Comparison of NIH[1] and PRARP Funding Using IADRP[2] Data FY14-FY18[3]
| NIH (N=7,972 Projects*) |
PRARP (N=83 Projects) |
All IADRP Partners* (Non-DoD) (N=11,429 Projects*) |
|
|---|---|---|---|
| Category A. Molecular Pathogenesis and Physiology of Alzheimer's Disease | 4,113 (51.6%)*↓ | 30 (36.1%)#↑ | 5,753 (50.4%)*↓ |
| Category B. Diagnosis, Assessment, and Disease Monitoring | 1,584 (19.9%)*↓ | 21 (25.3%)#↓ | 2,232 (19.5%)^↓ |
| Category C. Translational Research and Clinical Interventions | 1,241 (15.6%)*↓ | 8 (9.6%)#↑ | 1,852 (16.2%)*↓ |
| Category D. Population Studies (Epidemiology) | 547 (6.9%)*↑ | 9 (10.8%)*↑ | 727 (5.2%)*--- |
| Category E. Care, Support, and Health Economics of Alzheimer's Disease | 487 (6.1%)^↑ | 15 (18.1%)#↓ | 865 (7.6%)^↑ |
*0-1% Absolute change from FY13-FY17 |
|||
FY19/20 PRARP Program Cycle
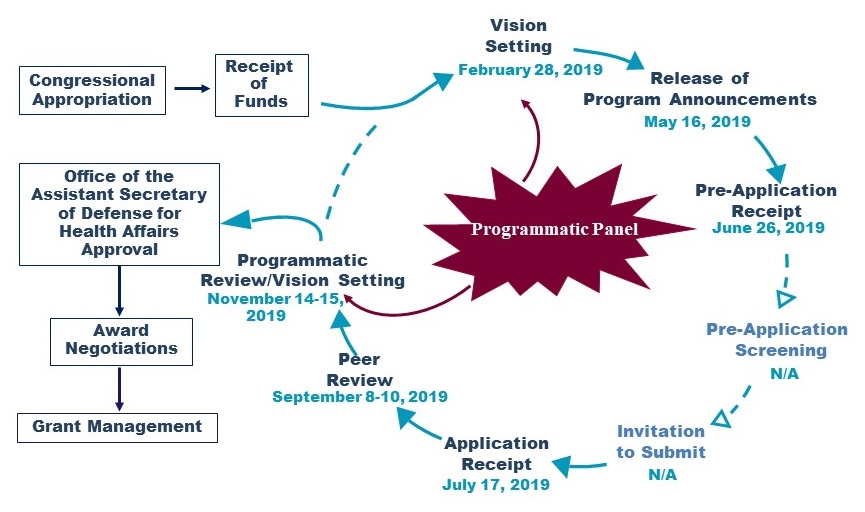 |
PRARP Portfolio Investment Strategy: Through the Years
| FY15 | FY16 | FY17 | FY18 | FY19 | |
|---|---|---|---|---|---|
| Gaps/Priorities | Paucity of Clinical Studies; Diagnostics; Research Resources; Quality of Life; Caregiver Support | Paucity of Clinical Studies; Diagnostics; Research Resources; Quality of Life; Caregiver Support; Epidemiology | Paucity of Clinical Studies; Diagnostics; Research Resources; Quality of Life; Caregiver Support; Epidemiology | Paucity of Clinical Studies; Diagnostics; Research Resources; Quality of Life; Caregiver Support; Epidemiology | Paucity of Clinical Studies; Diagnostics and Prognostics; Research Resources; Quality of Life; Family and Care Support; Epidemiology |
| Funding Opportunities |
|
|
|
|
|
| 16 Awards | 15 Awards | 18 Awards | 18 Awards | 21 Awards |
Strategic Plan: FY20 PRARP Overarching Challenges
- Challenges
- Paucity of Clinical Studies: The paucity of clinical studies to examine the interrelationship between TBI and subsequent AD/ADRD for the military, Veteran, and civilian communities
- Diagnostics and Prognostics: The need for technologies, tests, surveys, questionnaires, devices, biomarkers, or analyses to detect TBI and sequelae to include AD/ADRD utilizing new and/or pre-existing datasets
- Quality of Life: The need for technologies, assessments, interventions, or devices to benefit individuals living with the common symptoms of TBI and/or AD/ADRD
- Basic Research: Fundamental research to examine the interrelationship between TBI and subsequent AD/ADRD for the military, Veteran, and civilian communities and to translate these findings
- Epidemiology: The paucity of epidemiological research to examine the interrelationship between TBI, risk and resiliency factors, and subsequent AD/ADRD for the military, Veteran, and civilian communities
- Family and Care Support: The need for technologies, assessments, interventions, or devices that enhance the lives of those providing care and families of individuals living with the common symptoms of TBI and/or AD/ADRD
- All are equally important as they address the PRARP's Mission and Vision
- Address high-level research gaps
- Mechanism-specific
- All applications must address at least one Overarching Challenge
- Meant to align with PRARP Focus Areas (Technical Challenges; e.g., Novel Target Identification, Bioinformatics)
PRARP Research Investment FY14-FY18 by Overarching Challenge and Dollar Amount* (N=83;$63.1M)
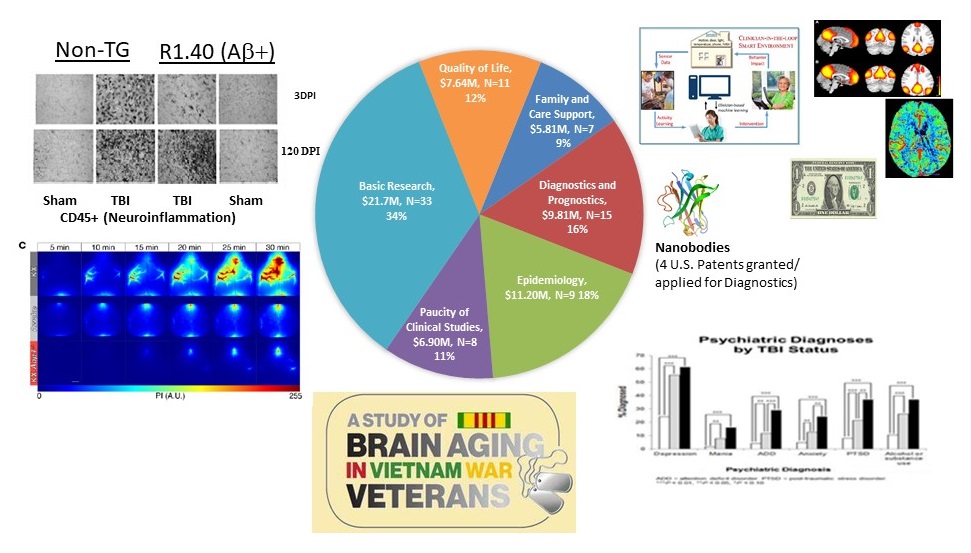 |
FY20 Award Mechanisms
- Convergence Science Research Award (CSRA)
- Intent: Support innovative or novel efforts to generate research resources, tools, or research efforts for researchers and/or practitioners in health sciences.
- Level I: Investigators at the postdoctoral level (or equivalent) or above in any scientific discipline. (Mentor required.)
- Level II: The Principal Investigator (PI) must be an independent investigator at or above the level of Assistant Professor (or equivalent).
- Maximum funding of $225,000 (Level I)/$500,000 (Level II) for direct costs (plus indirect costs) over 3 years.
- Innovation in Care and Support Award
- Intent: Support innovative research that improves the quality of life and care for individuals living with the common symptoms of TBI and/or AD/ADRD and/or their families and care providers.
- Level I: Investigators at the postdoctoral level (or equivalent) or above in any scientific discipline. (Mentor required.)
- Level II: The PI must be an independent investigator at or above the level of Assistant Professor (or equivalent).
- Maximum funding of $225,000 (Level I)/$500,000 (Level II) for direct costs (plus indirect costs) over 3 years.
- Research Partnership Award (RPA)
- Intent: To create an avenue for collaborative research partnerships between/among investigators to address a research problem or question in a manner that would be unachievable through separate efforts.
- The PI and Co-PI(s) must each be an independent investigator at or above the level of Assistant Professor (or equivalent)
- Maximum funding of $1.3M in total costs over 3 years.
- NEW: Accelerating Diagnostics for Traumatic Brain Injury Research Award (ADTBI)
- Intent: Supports high-impact, human-based, development of robust diagnostic and prognostic biomarkers for chronic TBI as they pertain to Alzheimer's disease and the related dementias.
- The PI must be an independent investigator at or above the level of Assistant Professor (or equivalent).
- The following biomarker types are encouraged:
- Imaging-Related
- Fluid-Based (e.g., Cerebrospinal Fluid [CSF], Blood, or Saliva)
- Retinal
- Wearable Devices
- Studies focused on biomarker discovery are discouraged.
- The biomarkers must correlate with clinical endpoints to include cognition and/or behavior.
- Maximum funding of $2.8M in total costs over 4 years.
- NEW: Leveraging Approaches for Innovation in Care and Support Award (LEAP-InCASA)
- Intent: Supports multi-institutional, harmonized research approaches that will advance the quality of life and care for individuals living with the common symptoms of TBI and/or AD/ADRD and/or their families and care providers.
- The PI and Co-PI(s) must each be an independent investigator at or above the level of Assistant Professor (or equivalent).
- Requires a Coordinating Center and at least two Partnering sites.
- The Coordinating Center provides overall leadership and infrastructure for all research projects.
- Coordinating and Partnering sites work together to harmonize research protocols, analyze data and publish research findings.
- Accrual for each of the research studies occurs at both the research site and coordinating center.
- Maximum funding of $2.8M in total costs over 4 years.
For more information regarding application deadlines, the full details for each award mechanism, and participation in future surveys, please register at:
https://ebrap.org/eBRAP/public/index.htm
