Monday, January 27, 2020
Printer Friendly Version in PDF Format (3 PDF pages)
Alzheimer's Policy Updates, 2019
Federal Policy Updates
- Fiscal Year (FY) 2020 Appropriations
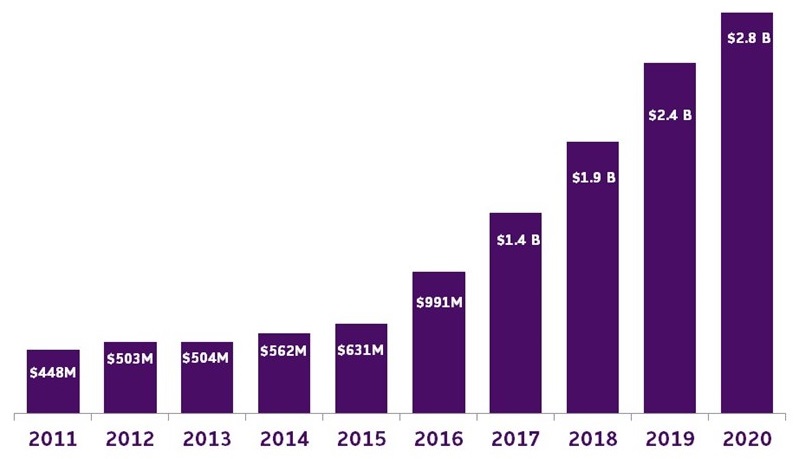
- Congress passed and the president signed into law a FY20 minibus which included an additional $350 million for Alzheimer's and dementia research at the NIH
- Total annual funding: $2.8 billion
- $10 million to implement the first year of the BOLD Act
- Extension of the Alzheimer's Semipostal Stamp
- Directs NIH to report on how it is implementing actions outlined in the National Strategy for Recruitment and Participation in Alzheimer's and Related Dementias Clinical Research to increase diversity in clinical trials
- Encourages CMS to update its definition of "detection of any cognitive impairment" element of the Annual Wellness Visit
Alzheimer's and Related Dementia Research Funding at the NIH
 |
Federal Policy Updates
- Palliative Care and Hospice Education and Training Act (PCHETA) (S.2080/H.R.647)
- October 2019 -- passed the House of Representatives
- Awaiting action in the Senate
- Lifespan Respite Care Reauthorization Act (S.995/H.R.2035)
- October 2019 -- passed Senate HELP Committee and House of Representatives
- Awaiting action by the full Senate
- EMPOWER for Health Act of 2019 (H.R.2781)
- October 2019 -- passed the House of Representatives
- Awaiting action in the Senate
- Younger-Onset Alzheimer's Disease Act (S.901/H.R.1903)
- December 2019 -- Senate released final version of the Older Americans Act including:
- Key components of the Younger-Onset Alzheimer's Disease Act
- Extends the RAISE Family Caregivers Act from 3 to 8 years
- Important elements of the Supporting America's Families and Caregivers Act
- We expect the Senate to take action in the coming weeks with both chambers coming to a final agreement on the legislation shortly thereafter
- December 2019 -- Senate released final version of the Older Americans Act including:
- Improving HOPE for Alzheimer's Act (S.880/H.R.1873)
- Continuing to work closely with the Senate Finance Committee and the House Ways and Means and Energy and Commerce Committees to ensure the bill receives a markup
- Senate Cosponsors: 48
- House Cosponsors: 199
Regulatory Advances
- Risk Adjustment:
- CMS proposed two new risk adjustment models to calculate its payments to MA plans; one model included two dementia codes: dementia with complications and dementia without complications
- CMS chose the model with the dementia codes, new methodology is now in effect
- Should lead to more accurate payment for complex beneficiaries and allow MA plans to better allocate resources
- HIPAA:
- HHS issued a Request for Information on Modifying HIPAA Rules to Improve Coordinated Care
- RFI asked specifically whether adult children should be able to access the treatment records of their parents who have early-onset dementia.
- Advisory Council has recommended that "Congress, federal agencies, and states must...address reimbursement, training, and HIPAA policy and practice barriers to enable health care providers to provide care planning with family caregivers[.]"
