Monday, January 25, 2016
LTSS Federal Update
ACL-Jane Tilly/Erin Long; ASPE-Rohini Khillan; IHS-Bruce Finke; NIA-Richard Hodes; VA-Susan Cooley
Goal 1: Research
- FY2016 Omnibus Bill that President Obama signed into law on December 18, gave NIH an increase of $350 million for research into Alzheimer's disease, including LTSS. NIH is investing in research milestones from the 2012 & 2015 Research Summits, along with additional input.
- NIA/NIH issued 2014-2015 Alzheimer's Disease Progress Report: Advancing Research toward a Cure in December 2015. See research progress made and promising research opportunities in Category E. Care and Caregiver Support.
- ACL/CDC/NIA webinar series had up to 800 attendees each. Webinars were on Sep.22,Oct. 21 & Nov. 17. Archived materials, with CE availability, can be accessed at http://www.aoa.acl.gov/AoA_Programs/HPW/Alz_Grants/index.aspx#resources;
Goal 1 (cont) NIH LTSS Research Opportunities
4 AD-LTSS-related FOAs issued September of 2015. First application due date was December 11, 2015. Applications to be reviewed in March of 2016.
- Research on Informal and Formal Caregiving for Alzheimer's Disease (R01-PAR-15-348) & (R21-PAR-15-351) for basic and translational research on caregiving for individuals with Alzheimer's disease (AD), at the individual, family, community, and population level.
- Emerging Directions for Addressing Health Disparities in Alzheimer's Disease (R01-PAR-15-349) & (R03-PAR-15-350) to study health disparities in Alzheimer's disease (AD) and related disorders.
Goals 2 & 3: Services Expansion & Quality
- Examining Models of Dementia Care: Examination of selected models of dementia care across settings using a framework to understand what challenges providers face in implementing models. Project involves an environmental scan and a series of five case studies. Results will inform future research related to developing quality measures and standards. Project runs from October 2015 through September 2016. (ASPE)
- Kansas forum: Linda Elam, PhD, MPH Deputy Assistant Secretary, Disability, Aging and Long-Term Care Policy gave the keynote address at an HHS-University of Kansas panel on Alzheimer's research and care in November 2015.
- REACH: The REACH Intervention in Indian Country has 22 certified individuals providing services in 9 Tribal communities. (IHS & VA)
- VA Innovative Models of Dementia Care: VA shared with federal agencies a report on Veterans Health Administration innovative dementia care models that are person-centered alternatives to institutional care. This completed National Alzheimer's Plan Action Number 2.E.3, on 11/17/15. Federal Partners disseminated the report to relevant groups, and further dissemination efforts are under consideration.
Clinical Services Subcommittee Federal Update
October, 2015 - January, 2016
Bruce Finke, MD
Shari Ling, MD
Indian Health Service
- Banner Alzheimer's Institute sponsored a National Conference on Alzheimer's Disease and Dementias in Native American Communities on October 15 and 16, 2015
- The meeting drew a wide audience from Indian Country, including front line service providers, AI/AN researchers, and individuals with policy expertise
- There is follow-up planning underway
CMS Updates Quality Strategy
National Quality Forum Scan of Home and Community-Based Services
- As part of its continuing work for HHS to examine quality measurement in home and community-based services and identify gaps, NQF released a report in December, 2015: http://www.qualityforum.org/ProjectMaterials.aspx?projectID=77692
- Committee appointees will discuss the results in January and March of 2016
- There are continuing opportunities for public comment
Medicaid Home Health Benefit Update
Medicaid Innovation Center Community Services Addition
Targeted Demonstration for Dual Eligible Beneficiaries
Accountable Health Communities Model
- In January, 2016 CMS announced a new demonstration program to identify beneficiary social needs and generate referrals to community-based services
- Social issues may affect health and increase risk
- The AHC model will build alignment between clinical and community-based services at the local level and improve beneficiary awareness of community services: https://innovation.cms.gov/initiatives/ahcm
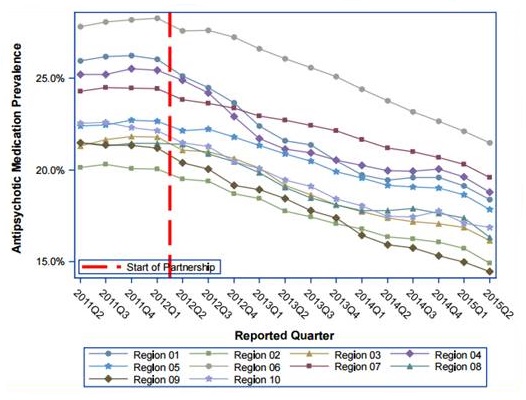
Partnership to Improve Dementia Care Overview
Progress to date - Reducing Use of Antipsychotic Medication
2014 Focused Dementia Care Survey Pilot Objectives
Nursing Home Quality Strategy
- A successful stakeholder meeting was held January 19, 2016 to help shape the draft CMS Nursing Home Strategic Plan
- There will be additional opportunities for public and private stakeholders to participate as the Strategy evolves
NAPA Research Progress Report
Richard J. Hodes, M.D.
Director
National Institute on Aging
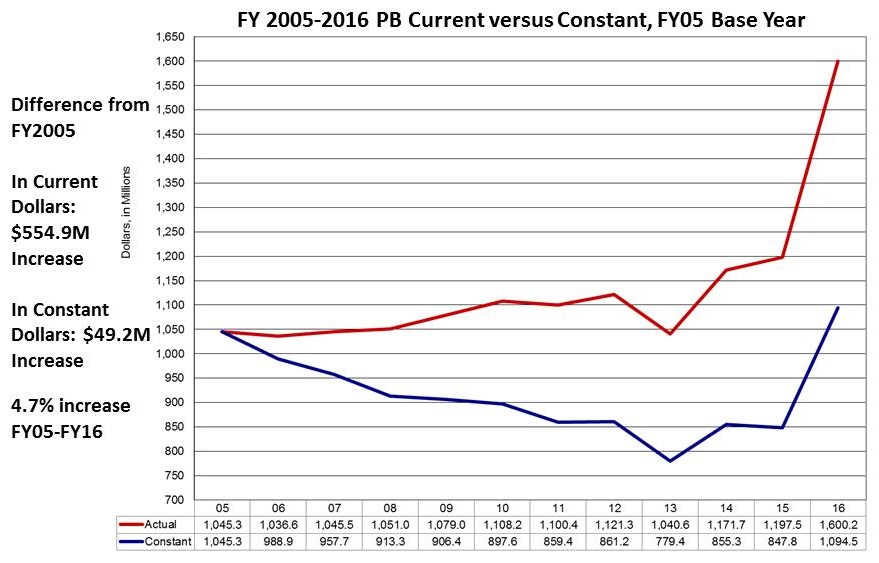
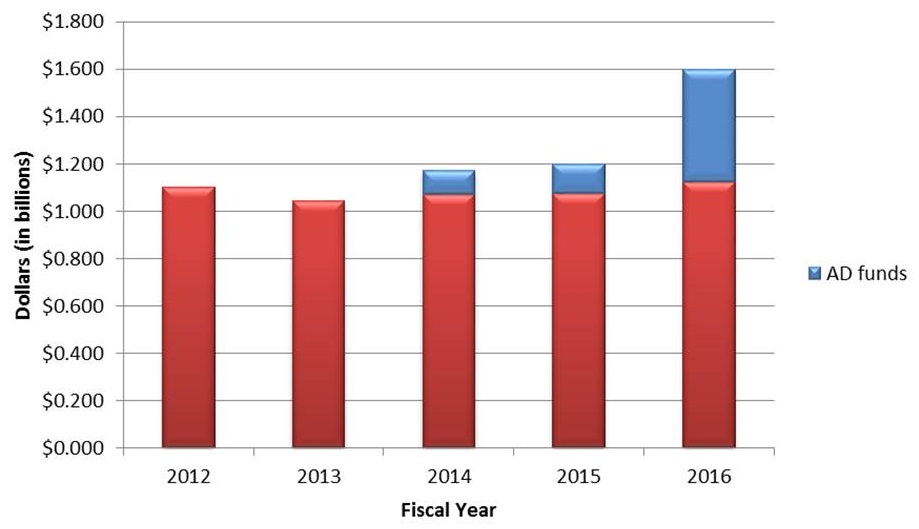
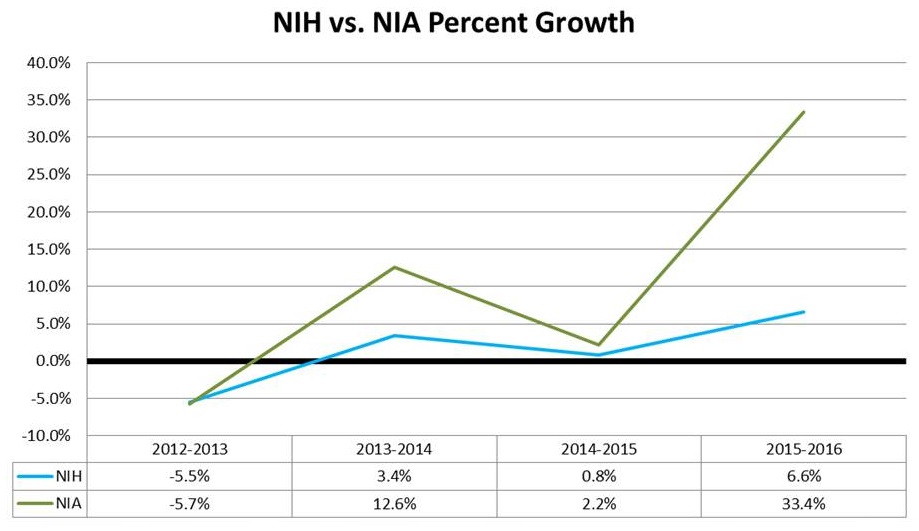
FY16 Budget Status - Funding Increases Across the Board
- $32 Billion for the NIH
- $200M for PMI
- $85M for BRAIN
- $350M for AD
- Included in the $32 Billion
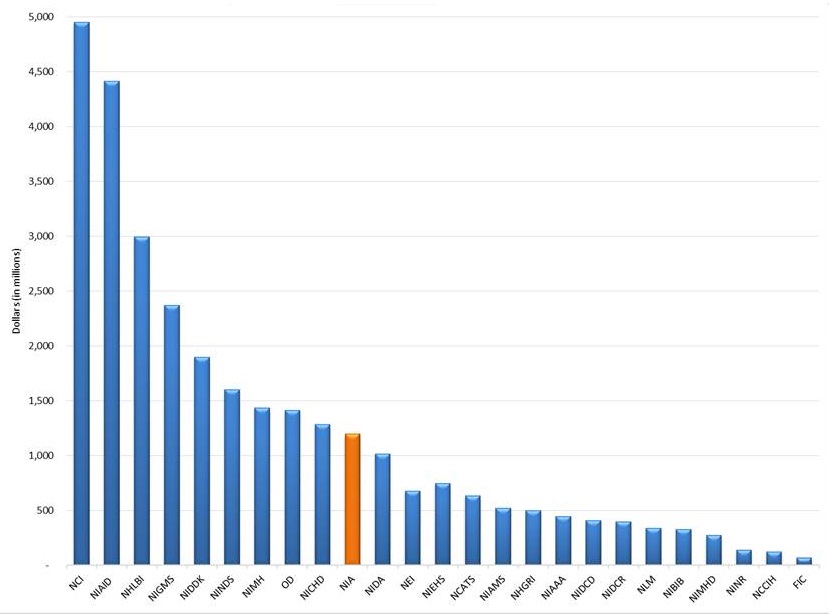
- $1.6B for the NIA
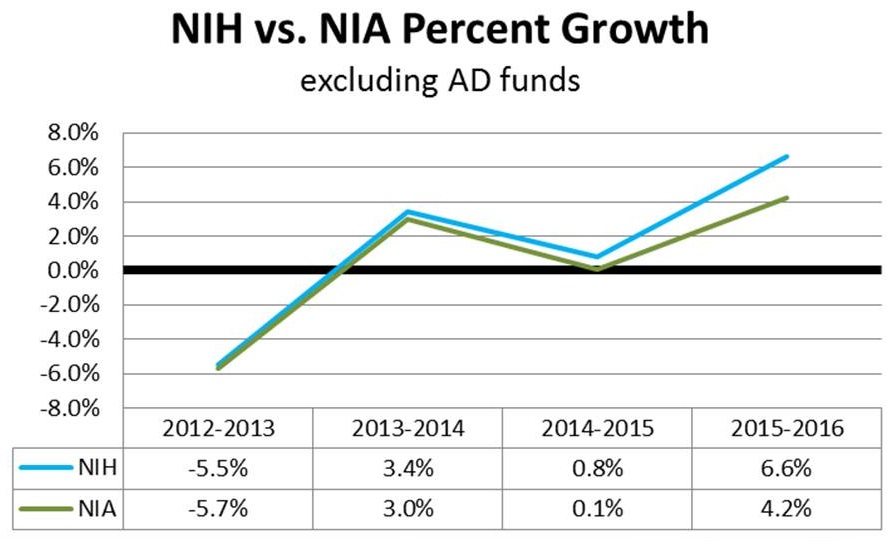
- ~4 percent increases across all ICs (not counting the $$ above) - 4.2% for the NIA
- All divisions will benefit
NIA Appropriations
NIA Appropriations
NIA Growth
NIA Growth
FY 15 NIH Operating Plan Budgets
FY 16 NIH Appropriations
Funding Policy - General Payline - FY15
| Requested Direct Costs |
<500K |
500k or greater |
| All applications except N.I or E.S.I. R01s |
8% |
5% |
| N.I. R01s |
12% |
9% |
| E.S.I. R01s |
14% |
11% |
Funding Policy - AD Payline - FY15
| Requested Direct Costs |
<500K |
500k or greater |
| All applications except N.I or E.S.I. R01s |
17% |
14% |
| N.I. R01s |
21% |
18% |
| E.S.I. R01s |
23% |
20% |
Implementation of Additional AD Funding
- Ten FOAs released in October 2015 in the event that additional funds became available in the FY16 budget
- NIA will have the ability to support relevant projects at other ICs (beyond their IC paylines)
Alzheimer's Disease Funding Opportunities
- PAR-15-349 Health Disparities and Alzheimer's Disease (R01)
- PAR-15-350 Emerging Directions for Addressing Health Disparities in Alzheimer's Disease (R03)
- PAR-15-348 Research on Informal and Formal Caregiving for Alzheimer's Disease (R01)
- PAR-15-351 Research on Informal and Formal Caregiving for Alzheimer's Disease (R21)
- PAR-15-356 - Major Opportunities for Research in Epidemiology of Alzheimer's Disease and Cognitive Resilience (R01)
- PAR-15-357 - Understanding Alzheimer's Disease in the Context of the Aging Brain (R01)
- PAR-15-358 - Capturing Complexity in the Molecular and Cellular Mechanisms Involved in the Etiology of Alzheimer's Disease (R01)
- PAR- 15-359 - Novel Approaches to Diagnosing Alzheimer's Disease & Predicting Progression (R01)
- PAR-16-365 - Pilot Clinical Trials for the Spectrum of Alzheimer's Disease and Age-related Cognitive Decline (R01)
- PAR-16-364 - Phase III Clinical Trials for the Spectrum of Alzheimer's Disease and Age-related Cognitive Decline (R01)
Application Update
- NIA has received just under 200 applications for the first of two submission deadlines eligible for 2016 funding consideration for the 10 new AD FOAs.
- Together, these applications have requested more than $145 million in single year costs.
- These new submissions are in addition to the many applications targeting AD research submitted through normal investigator-initiated routes that NIA normally receives.
New Alzheimer's Initiatives
- Impact of Aging in Human Cell Models of Alzheimer's Disease
- Alzheimer's Disease Clinical Trials Consortium
- Neurodegenerative Disease Biorepository
- From Association to Function in the Alzheimer's Disease Post Genomics Era
- Technology to Assess Everyday Functions
- Mobile Consent
Tracking and Next Steps
- RCDC will report FY2015 dollars for AD+ADRD (in addition to the individual categories) in early 2016
- This number will serve as a new baseline for tracking expenditures related to implementation of AD and related dementias research milestones
- Both the new "rolled-up" number as well as the original categories (AD, FTD, LBD, and VCI/D) will be available going forward
- NIH's FY17 plans will be revised operationally in the context of the new funding, as part of an ongoing planning and implementation process
- The FY18 bypass budget is anticipated to be released in summer 2016
Important Tracking Links
ADRD Summit 2016
- Natcher Conference Center on NIH Main Campus, Bethesda, MD
- March 29-30, 2016
- Update research recommendations:
- Frontotemporal degeneration
- Lewy body dementia
- Vascular contributions to cognitive impairment and dementia
- Multiple etiology dementias
- Health disparities
- New in 2016: NGO Session
- Registration is free and open to the public: https://meetings.ninds.nih.gov/meetings/ADRelatedDementias2016/
- Hosted by NINDS in collaboration with NIA
NINDS Funding Opportunity Announcements on AD-Related Dementias - planned for FY 2016
- Biomarkers for small vessel vascular contributions to cognitive impairment and dementia (VCID)
- Basic research on diffuse white matter disease in VCID
- Biomarkers for Lewy body dementia
- Tau biology and contribution to neurodegeneration