Monday, August 1, 2016
Development of the FY18 NIH Bypass Budget for Alzheimer's Disease and Other Dementias
Richard J. Hodes, M.D.
Director
National Institute on Aging
Bypass Budget Language
SEC. 230. Hereafter, for each fiscal year through fiscal year 2025, the Director of the National Institutes of Health shall prepare and submit directly to the President for review and transmittal to Congress, after reasonable opportunity for comment, but without change, by the Secretary of Health and Human Services and the Advisory Council on Alzheimer's Research, Care, and Services, an annual budget estimate (including an estimate of the number and type of personnel needs for the Institutes) for the initiatives of the National Institutes of Health pursuant to the National Alzheimer's Plan, as required under section 2(d)(2) of Public Law 111-375.
NIH Meetings Provided the Basis for Priorities, Milestones, and Budget Estimates
- 2012 Alzheimer's Disease Research Summit
- 2013 meeting on Alzheimer's Disease-Related Dementias: Research Challenges and Opportunities
- 2013 meeting on Advancing Treatment for Alzheimer's Disease in Individuals with Down Syndrome
- 2015 Alzheimer's Disease Research Summit
- 2016 meeting on Alzheimer's Disease-Related Dementias
Recommendations used to create milestones for developing the Alzheimer's Disease (and related dementias) Bypass Budget
Combined External and Internal Input - FY18 AD Bypass Budget
- Input at 2012-2016 meetings:
- Academic research community
- Industry
- Non-governmental organizations
- Other Input:
- 2016 Request for Information
- Development of comprehensive milestones (NIH staff) - based on recommendations and milestones from meetings
- Trans-NIH (including NINDS) staff discussion; milestones edited to ensure comprehensive inclusion of priorities for FY18
- NIH staff "price" the milestones
- Final budget estimate for FY18
Trans-NIH Input for FY18 ADBB
- Thirteen Institutes and Centers provided feedback that contributed to the development of this year's professional judgment budget:
- NIA
- NINDS
- NIMH
- NINR
- NIBIB
- NICHD
- NIEHS
- NIDDK
- NIDCR
- NHLBI
- NIAAA
- NCATS
- FIC
Using CADRO as a Framework
- The eight CADRO (Common Alzheimer's Disease Research Ontology) categories provide the overarching framework for the FY18 AD Bypass Budget and narrative.
- CADRO provides the framework for IADRP (the International Alzheimer's Disease Research Portfolio) and will allow tracking of implementation in the budget areas in future years.
FY18 Professional Judgment Budget Baseline
| Baseline Estimate, Fiscal Year 2017 (dollars in thousands) |
|
|---|---|
| Alzheimer's Disease including Alzheimer's Disease Related Dementias (AD+ADRD) | $991,000 |
- RCDC is now reporting dollars for AD+ADRD (in addition to the individual categories) and will do so going forward
- This number will serve as a new baseline for tracking expenditures related to implementation of AD and related dementias research milestones
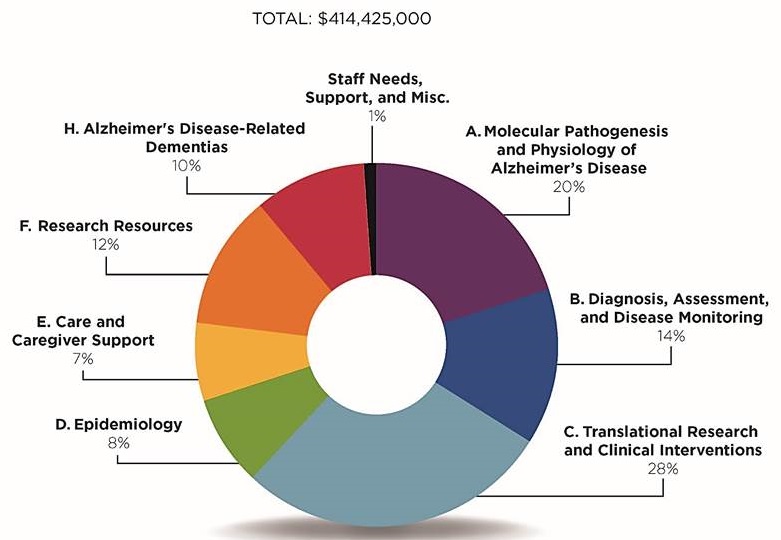
FY18 Professional Judgment Budget
| Area of Research | Amount (dollars in thousands) |
|---|---|
| A. Molecular Pathogenesis and Physiology of Alzheimer's Disease | $83,000 |
| B. Diagnosis, Assessment and Disease Monitoring | $58,100 |
| C. Translational Research and Clinical Interventions | $118,600 |
| D. Epidemiology | $33,325 |
| E. Care and Caregiver Support | $28,000 |
| F. Research Resources | $49,950 |
| H. Alzheimer's Disease-Related Dementias | $40,200 |
| Staff Needs, Support, and Misc. | $3,250 |
| Total Additional Resources Needed | $414,425 |
| TOTAL RESOURCES NEEDED (dollars in thousands) | $1,405,425 |
Distribution of Additional Budget Funding Request Across Research Areas, FY18
 |
Implementation Tracking
- IADRP: http://iadrp.nia.nih.gov/
- Will continue to offer detailed tracking of initiatives and awards with respect to our research milestones, under the CADRO categories
- Web-based tool for tracking funding initiatives and activities: https://www.nia.nih.gov/alzheimers/milestones
- Developed by the NIH and other AD funding organizations
- Aimed at addressing the research milestones associated with NAPA
VA Research Update
- The VA just completed its most recent Merit review cycle
- Five Merit awards will be funded from the 18 that were reviewed.
- The VA expanded the RFA to include FTLD, LBD, and vascular contributions to Dementia (VCID).
- 2/5 have direct relationship to FTLD
- 1/5 has a connection with VCID
Contact Lisa Opanashuk with questions
Introducing...Eliezer Masliah, M.D.
- New Director of NIA's Division of Neuroscience
- Expertise in neuroscience and neuropathology
- 24 years at the University of California, San Diego
- Served on the NIA Advisory Council, the NIA Neuroscience of Aging Study Section, and the Cellular and Molecular Biology of Neurodegeneration Study Section.
Special thanks to Dr. Tony Phelps for his support as Acting Director
 |
