Friday, April 29, 2016
NAPA Research Progress Report
Richard J. Hodes, M.D.
Director
National Institute on Aging
FY16 Budget Status -- Funding Increases Across the Board
- $32 Billion for the NIH
- $200M for PMI
- $85M for BRAIN
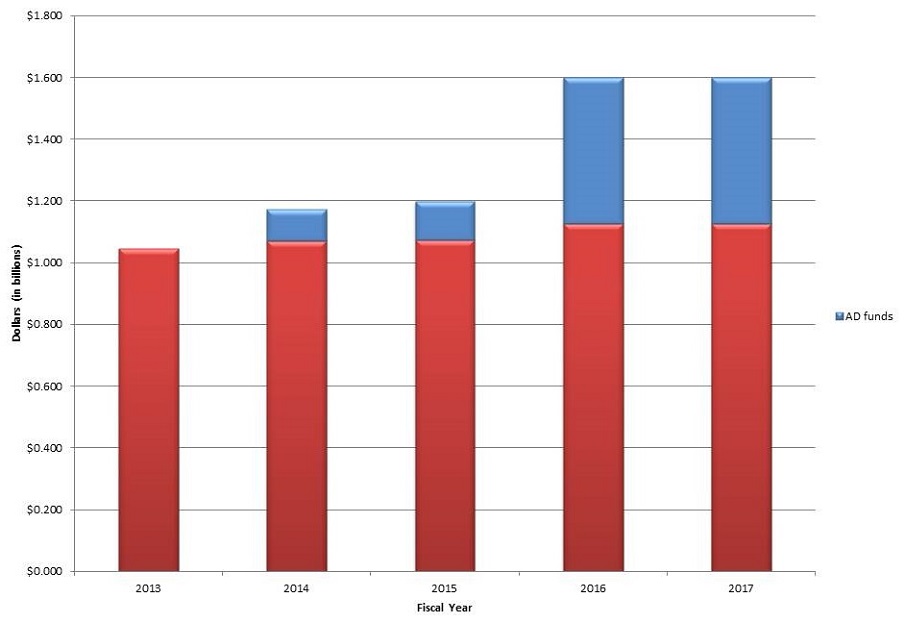
- $350M for AD
- $1.6B for the NIA
- ~4 percent increases across all ICs (not counting the $$ above) -- 4.2% for the NIA
- All divisions will benefit
- DBSR
- DGCG
- DAB
- DN
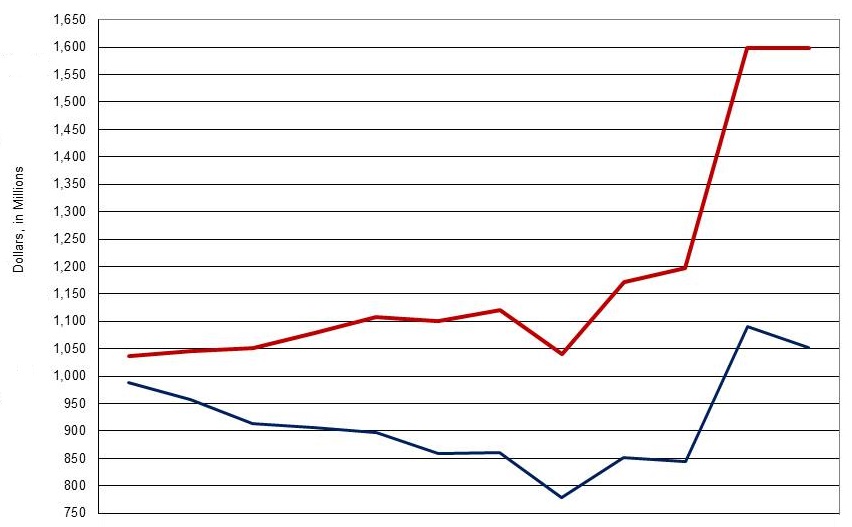
NIA Appropriations FY 2005-2017 PB
| Current versus Constant, FY05 Base Year | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
 |
||||||||||||
| 06 | 07 | 08 | 09 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | |
| Actual (red line) |
1,036.6 | 1,045.5 | 1,051.0 | 1,079.0 | 1,108.2 | 1,100.4 | 1,121.3 | 1,040.6 | 1,171.7 | 1,197.5 | 1,598.2 | 1,598.2 |
| Constant (blue line) |
988.9 | 957.7 | 913.3 | 906.4 | 897.6 | 859.4 | 861.2 | 779.4 | 851.8 | 844.2 | 1,090.0 | 1,051.6 |
|
||||||||||||
NIA Appropriations
 |
Implementation of Additional AD Funding
- Ten FOAs were released by the NIA in October 2015 for potential funding beginning in FY2016
- NINDS has begun to release additional Alzheimer's Disease Related Dementias funding announcements
- NIA is working with other NIH Institutes and Centers to help support relevant projects (beyond their IC paylines)
Application Update
- NIA has received just under 300 applications for the first two submission deadlines eligible for FY2016 funding consideration for the 10 new AD FOAs.
- These new submissions are in addition to the many applications targeting AD research submitted through normal investigator-initiated routes that NIA normally receives.
Payline Update
| Requested Direct Costs | <500k | 500k or greater | ||
|---|---|---|---|---|
| Allocation | General | AD research | General | AD research |
| All applications except N.I. or E.S.I. R01s | 9% | 22% | 6% | 19% |
| N.I. R01s | 17% | 25% | 14% | 22% |
| E.S.I. R01s | 19% | 27% | 16% | 24% |
FY17 AD Initiatives
- Alzheimer's Disease Clinical Trials Consortium
- Impact of Aging in Human Cell Models of AD
- Neurodegenerative Disease Biorepository
- From Association to Function in the AD Post-Genomics Era
- Technology to Assess Everyday Functions
Tracking and Next Steps
- RCDC has reported FY2015 dollars for AD+ADRD (in addition to the individual categories) and will do so going forward
- This number will serve as a new baseline for tracking expenditures related to implementation of AD and related dementias research milestones
- IADRP will continue to offer detailed tracking of initiatives and awards with respect to our research milestones, under the CADRO categories
- NIH's FY17 plans will be revised operationally in the context of the new funding, as part of an ongoing planning and implementation process
- The FY18 bypass budget is anticipated to be released in summer 2016
International Alzheimer's Disease Research Portfolio - UPDATE
- Joint initiative led by NIA and the Alzheimer's Association (since 2010) to capture funded AD and related dementias research -- promotes collaboration
- Currently includes 35 public, private and international funding organizations in 10 countries, including U.S., Australia, Brazil, Canada, Czech Republic, France, Italy, The Netherlands, Poland and the UK
- Recent additions -- Research Manitoba (Canada), Bright Focus Foundation (US), LECMA: Vaincre Alzheimer (France), Internationale Stichting Alzheimer Onderzoek (The Netherlands)
- Captures 7000+ unique projects conducted by 4000+ researchers across 1000+ institutions representing ~$4.7 billion in basic, translational and health services research funding
- Upcoming plans -- update CADRO categories, topics and themes; solicit funded ADRD projects from IADRP and potential new partners.
AD Research Milestone Database
- https://www.nia.nih.gov/alzheimers/milestones
- Web-based tool for tracking funding initiatives and activities developed by the NIH and other AD funding organizations aimed at addressing the research milestones associated with NAPA
- Will facilitate strategic coordination and collaborations
- Milestones tracked span the entire AD research landscape including basic, translational, clinical and health services research (the new ADRD milestones will be included once approved)
The FY2018 AD Bypass Budget -- In Progress
- 2012 Alzheimer's Disease Research Summit
- 2013 meeting on Alzheimer's Disease-Related Dementias: Research Challenges and Opportunities
- 2013 meeting on Advancing Treatment for Alzheimer's Disease in Individuals with Down Syndrome
- 2015 Alzheimer's Disease Research Summit
- 2016 meeting on Alzheimer's Disease-Related Dementias*
The recommendations and milestones that emerged from these meetings are used to create a comprehensive and up-to-date set of milestones for the budgeting process
*The preliminary report from the 2016 ADRD meeting will be used for the FY18 Bypass Budget.
Combined External and Internal Input -- FY18 AD Bypass Budget
- Input at 2012-2016 meetings:
- Academic research community
- Industry
- Non-governmental organizations
- Other Input:
- 2016 Request for Information
Development of comprehensive milestones (NIH staff) -- based on recommendations and milestones from meetings
Trans-NIH (including NINDS) staff discussion; milestones edited to ensure comprehensive inclusion of priorities for FY18
NIH staff "price" the milestones
Final budget estimate for FY18
Molecular Mechanisms of the Vascular Etiology of Alzheimer's Consortium
- NIA and NINDS launched M2OVE-AD in March 2016
- The goal: to dissect the complex molecular mechanisms by which vascular risk factors influence AD and identify new targets for treatment and prevention.
- It builds upon the open-science approach and the big-data infrastructure established by the Accelerating Medicines Partnership.
- This five-year, $30-million program brings together >12 research teams working on five complementary projects.
Incidence of Dementia over Three Decades in the Framingham Heart Study
 |
The 5-year age-/sex-adjusted cumulative hazard rates for dementia were:
|
| Relative to the incidence during the first epoch, incidence declined by 22%, 38%, and 44% during the 2nd, 3rd, and 4th epochs |
| Satizabal, CL et al. (2016) 374(6):523-32 |
VA Research Update
- VA's Research office updated the wording in its specialty Alzheimer's Disease Request for Applications for biomedical laboratory or clinical research to include FTD, LBD, and/or vascular contributions to cognitive impairment and dementia in addition to AD
- VA's Research office is discussing with NIA ways to increase collaboration on funding opportunities, including identifying opportunities to collaborate more closely around dementia research (e.g., recruitment/retention in clinical trials; data sharing; biorepository use; use of VA's electronic health records in research studies).
