by
Joseph A. DiMasi, Ph.D.
Tufts Center for the Study of Drug Development
Tufts University*
A background report prepared for the
Department of health and Human Services' Conference on
Pharmaceutical Pricing Practices, Utilization and Costs
August 8-9, 2000
Leavey Conference Center, Georgetown University
Washington, DC
Final Version
* The analyses of pricing trends contained in this paper were conducted by the author in his capacity as a consultant to the Drug Value Group at the Schneider Institute for Health Policy, Brandeis University.
INTRODUCTION
Prescription drug expenditures have risen at double-digit rates in recent years. These developments have prompted considerable commentary about the behavior of pharmaceutical firms and concern about whether consumers are getting value for money. A certain degree of confusion likely exists in the general public, and even among commentators and policy makers about the nature of the increases in pharmaceutical costs. In some instances expenditure increases are interpreted as price inflation. To shed light on the concerns, a number of studies have been conducted recently to sort through the factors that contribute to the rate at which prescription drug expenditures have been increasing (Barents Group, 1999; Dubois et al., 2000; Express Scripts, 2000; Merck-Medco, 2000; Schneider Institute for Health Policy, 2000). The contribution that price increases make to the escalating costs is determined in somewhat different ways in these studies, and so the contribution level differs somewhat. Additionally, a certain portion of the cost growth must be attributed to “new drugs.” Unfortunately, at the level at which these studies are conducted, this factor must remain something of a black box. This is so because the new drug component is the product of both price and quantity effects. To further our understanding of pharmaceutical pricing patterns for both old and new drugs, it is necessary to study prices at the narrow category level or even at the individual drug level.
The purpose of this report is to examine pricing dynamics for prescription drugs for recent years. The prices of new drugs will be analyzed over time and by factors that may be associated with launch prices. Price trends for 1995 to 1999 will also be examined by therapeutic category for a number of major categories (in terms of expenditure levels or growth) with well-defined subclasses that can be analyzed individually and compared on the basis of the age of the category. The pricing of generic substitutes in these therapeutic categories will also be provided for comparative purposes. Finally, launch prices during the study period for new entrants to existing subclasses within the therapeutic categories will be examined and compared to the prices of the incumbents in the subclass.
The results show that new drugs can vary substantially in their impact on drug expenditures. Some drugs are introduced at significant discounts to existing drugs that are highly substitutable with the new drug. In addition, while entirely new classes of compounds to treat a disease or condition are often priced at a premium relative to older classes, this is not always the case. The data also suggest that new drug prices tend to reflect the degree of price sensitivity in the market and the perceived value of the product to patients. Finally, average price increases over time for the pharmacologic and chemical classes examined varied in all directions in relation to general price inflation. However, most class price increases were similar to economy-wide inflation rates.
METHODS
Prescription drug claims data were provided to the Schneider Institute for Health Policy at Brandeis University by PCS Health Systems, Inc., a large pharmaceutical benefits manager (PBM) that currently manages the prescription drug benefits of approximately 50 million individuals in the United States on behalf of health plans, HMOs, and self-insured employers. The claims data were monthly and covered fiscal years (October to September) 1995 to 1999. Data were obtained for individuals who were continuously enrolled during a fiscal year. Over the study period the number of covered individuals ranged from approximately six to ten million. Before receiving the data, patient identifiers in the datasets were encrypted.
Price Calculations
The PCS claims data for drug products were reported at the 11-digit NDC level. This means that each record contained identifying information for the molecule dispensed, its dosage strength and form, and its package size and type. The data also identified whether the dispensed product was a brand or a generic. Information on the days of use and the number of units of the product dispensed for each claim was included in the datasets. Most drugs are available in many strengths, forms, and package sizes and types. In theory, prices can be determined for each product presentation. Although unit pricing for packs tends to vary inversely with pack size, the pricing differentials are often not large. In addition, package size is clinically irrelevant to prescribers and patients. What matters is the strength and form of the molecule. However, to account for the contributions of all pack sizes and types we calculated prices for each different strength and form of a molecule as the weighted average of the prices for each pack size and type, where the weights were the number of units dispensed for each size and type.
For purposes of this report, price trends and price comparisons were examined on the basis of the most frequently prescribed strength and form for each molecule. The prices used are based on transaction costs for insured individuals in the retail pharmacy market.1 The prices are determined on the basis of what is referred to as the ingredient cost for the claim. This is the amount paid to the pharmacy for the claim, net of dispensing fees and taxes. It includes any patient copays and deductibles. Each price is calculated as the cost of the most frequent daily dosing for the drug. When inflation-adjusted prices were required, we deflated the nominal prices by the Consumer Price Index (CPI-U) for all items.
PBMs often negotiate rebates from manufacturers on behalf of their clients. The rebates are post-transaction transfers that are shared between the PBM and the payers. Discussions with PBM industry executives indicated that rebates are given for access (formulary status) and the ability to move market share for individual products (Personal Communications, 2000). Rebates are not given for all products. One industry executive estimated that only about one-third of products are rebatable at any point in time. Generics will not be rebated. In addition, brand name products with no or only one direct competitor likely will not be rebated. The entry of generics in a therapeutic class may also lessen the inclination of manufacturers to offer rebates for the off-patent and even the on-patent drugs in the class.
Although there can be outliers, rebates generally are in the range of 2% to 4% of sales. The industry executives also indicated that after granting rebates in the early 1990s, manufacturers retrenched and substantially reduced PBM rebates in the mid-1990s because of concerns that payers were not delivering a significant benefit for the rebates.2 However, when PBMs implemented stricter access and utilization controls, rebates returned in the late 1990s to at least the levels that had existed in the early 1990s. This suggests that the degree of rebating increased over our study period. Rebate data were not available to us, but the potential impact on our results of incorporating rebates into prices will be discussed at appropriate points below.
Although our pricing data relate to retail transactions for the insured population, they likely can provide reasonable indicators of trends for the varied prices that different buyers face. We obtained publicly available data on Federal Supply Schedule (FSS) unit prices for drugs that had FSS contracts that were initiated in the last quarter of our study period.3 While prices for our data were generally higher than the FSS prices, the simple correlation coefficient of our prices for the last month of our study period and the FSS prices was 0.99, and the correlation coefficient for the logs of these prices was 0.91. Thus, our prices are likely also correlated with average manufacturer prices.
Launch prices for drugs that entered the market during the study period were obtained as the price of a usual daily dose for the first month in which the drug appeared in the dataset.4 When averaging launch prices across drugs by launch year or some other characteristic, we computed a weighted average, where the weights were the number of days of use for the first three months following the launch month.5 We found 156 molecules in the dataset that were first launched during the study period. From the medical literature and the data itself, it was evident that the dosing for a number of these compounds was too variable for a meaningful daily cost to be computed. These compounds were dropped from the launch price analyses, resulting in 100 new molecules to be examined. We classified each of the new drugs as acute, subchronic, or chronic. Acute drugs were defined as drugs that are used primarily on a very short –term basis (less than three weeks). Subchronic drugs were defined as drugs to be used typically for three weeks up to six months. Chronic drugs were those drugs that are intended usually to be used on a longer-term basis.
RESULTS
We obtained results on several significant aspects of the pricing of prescription pharmaceutical products. First we examined trends in launch price levels for all new drugs introduced during 1995 to 1999 for which we have reliable data on daily dosing. Second, we examined price changes over the study period for eight therapeutic categories and a number of subclasses. The subclasses are groups of drugs that are used to treat a given condition or set of related conditions and that either have the same mechanism of action or have chemical structures that are similar. The therapeutic categories are antiarthritics, antidepressants, antihistamines, antihyperlipidemics, antihypertensives, antiulcer, and two antibiotic classes (cephalosporins and macrolides). The compounds in these classes constitute a mix of drugs that are used on an acute, subchronic, and chronic basis. In aggregate these classes accounted for half of total retail prescription drug expenditures in 1999, and expenditures on these compounds grew at roughly the same rate as for prescription drugs as a whole.6 We also examine the pricing of new entrants to subclasses in the eight therapeutic categories.
New Drug Launch Prices
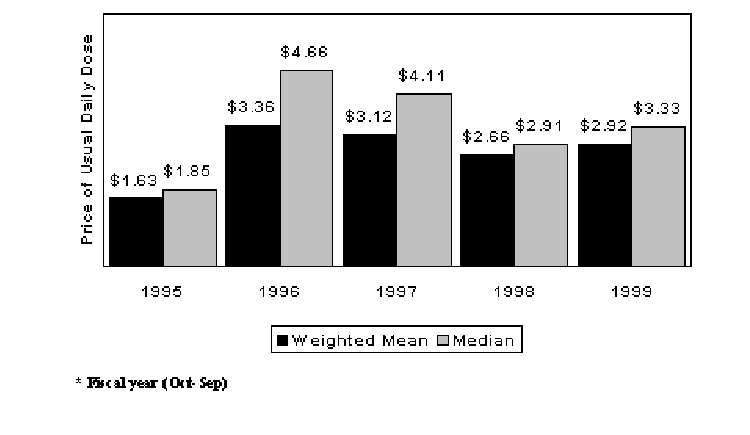
Figure 1 shows average launch prices for new drugs by launch year. Prices for the 1995 drugs were relatively low, but one cannot make much of this result since only nine compounds were used for the 1995 calculations.7 Beyond 1995, no time trend is apparent. The mix of drugs launched in a year may significantly affect average launch prices. It was not feasible to determine dosing variability for the several thousand older drugs in use. However, we should gain some insight on relative price levels by examining average cost per day for all new and all old drugs over the study period. In 1999 dollars, the weighted average cost per day for the new drugs during 1995 to 1999 was $2.20, which was one-third higher than the weighted average cost per day for all older brand name drugs ($1.66). If the new drugs had been priced at the average of all the old drugs, then drug expenditures would have been lower over the study period by 2.4%.8
Figure 1. New Drug Launch Prices (1999$) by Launch Year*
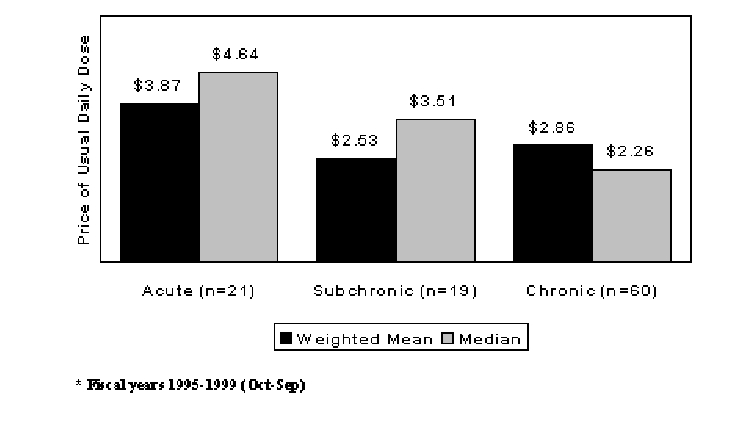
A number of drug characteristics may relate to launch prices. Among the factors that may be considered are the length of use, convenience of use, and relative safety and efficacy profiles. Uninsured, and even insured, patients may be more price sensitive the longer is the expected duration of therapy. Thus, we may hypothesize that prices will tend to be lower for drugs that are used for longer periods. Lu and Comanor (1998) found evidence in their regressions that acute drugs are launched at higher prices than chronic drugs. Here, we have divided drugs more finely by defining three duration categories. Figure 2 shows that average launch prices were higher for acute drugs. The median price for subchronic drugs was also greater than the median price for chronic drugs. The mean price is somewhat lower. However, during the study period, ten AIDS antivirals were launched. These drugs were classified as chronic and had daily dose prices that were much higher than those for most drugs. When the AIDS antivirals are excluded, the mean price for chronic drugs drops below that for subchronic drugs to $2.38 per day.
Figure 2. New Drug Launch Prices (1999$) by Duration of Use*
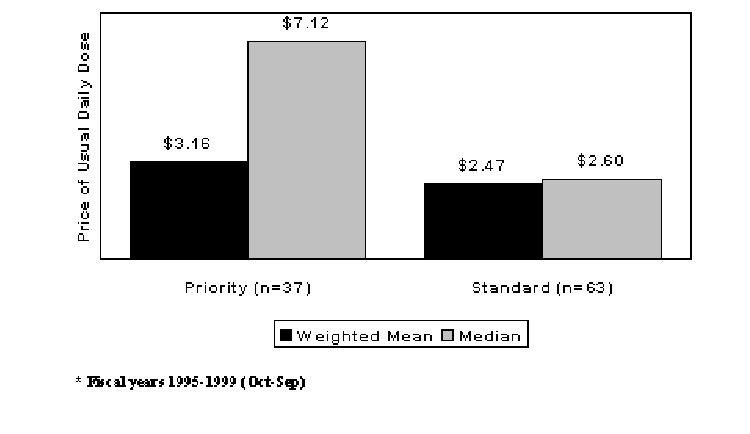
A measure of the value of a new drug introduction is the Food and Drug Administration (FDA) rating of therapeutic significance at the time of approval. The FDA divides new drugs into two rating categories for purposes of prioritizing internal application reviews. A drug that receives a priority rating is thought to represent a moderate or significant gain over existing therapy in average efficacy, convenience, or safety. A drug that receives a standard rating is thought to represent little or no gain over existing therapy. The ultimate value of some compounds may not be learned until the drugs have been used in practice for some time, but the rating system provides a reasonable measure of perceived relative value at the time of approval. Figure 3 suggests that clinical advantages for new drugs tend to be rewarded in the marketplace with higher prices. Both mean and median launch prices are higher for priority drugs.9
Figure 3. New Drug Launch Prices (1999$) by FDA Rating of Therapeutic Significance (1995-1999*)
New drugs that have the same mechanism of action or are chemically similar to other drugs already on the market must compete with the incumbents for market share. Thus, we may hypothesize that unless these follow-on products have significant clinical advantages over other drugs in their class, they will tend to be priced at parity or at a discount to at least the subclass price leader (Lu and Comanor, 1998; Kolassa, 1997; Reekie, 1978,1998; Towse and Leighton, 1999).
Table 1. New Drugs in Existing Classes Tend to be Priced at a Discounta
| Subclassb | Drug | Launch Month | Discount Relative to Weighted Mean Price | Discount Relative to Price Leader |
|---|---|---|---|---|
| ACE-inhibitors | Univasc® 7.5 mg | May-95 | 52.7% | 67.8% |
| Mavik® 2 mg | June-96 | 30.4% | 53.2% | |
| ARBs | Diovan® 80 mg | Feb-97 | 1.4% | 1.4% |
| Avapro® 150 mg | Oct-97 | -2.6% | -2.6% | |
| Atacand® 16 mg | Oct-98 | 0.3% | 0.3% | |
| Micardis® 80 mg | Dec-98 | 0.7% | 0.7% | |
| CCBs | Sular® 20 mg | Feb-96 | 37.7% | 67.9% |
| Posicor® 50 mg | Jul-97 | 8.8% | 55.0% | |
| Cox-2 | Vioxx® 25 mg | May-99 | 0% | 0% |
| Macrolides | Dynabac® 250 mg | Oct-95 | 42.6% | 49.0% |
| Non-sedating antihistamines | Allegra® 60 mg | Aug-96 | 14.1% | 15.0% |
| PPIs | Prevacid® 30 mg | May-95 | 10.1% | 10.1% |
| Aciphex® 20 mg | Sep-99 | 4.9% | 6.7% | |
| Statins | Lipitor® 10 mg | Jan-97 | 33.9% | 60.1% |
| Baycol® 0.3 mg | Jan-98 | 29.5% | 43.1% | |
| SNRIs | Serzone® 150 mg | Feb-95 | 9.7% | 9.7% |
| SSRIs | Luvox® 100 mg | Jan-95 | 8.1% | 12.7% |
| Celexa® 20 mg | Aug-98 | 17.9% | 23.0% | |
| 3rd Generation Oral Cephalosporins | Cedax® 90 mg | Feb-96 | -7.4% | 20.0% |
| Omnicef® 300 mg | Aug-98 | -3.1% | 18.2%
|
a A positive value in the table indicates a lower price for the new entrant, while a negative value indicates a higher price.
bSome of the incumbents in a few subclasses are combination drugs. These products were excluded from the analysis.
We compared the launch prices of drugs that were new entrants to existing subclasses in the eight therapeutic categories to the price for the incumbent with the highest price and to the weighted (days of use) mean price for incumbents in the subclass at the time of launch (Table 1). Of the 20 drugs examined, 13 were launched at discounts of at least 5% relative to both the price leader and to the weighted average price for the class. Five of the drugs were introduced essentially at parity with existing prices, but four of them were members of one class. The angiotensin-receptor blockers (ARBs) all entered essentially at parity with the other members of the class. The fifth drug is the Cox-2 inhibitor Vioxx, which entered at the same price as Celebrex, the first Cox-2 to be launched. Only the third-generation oral cephalosporins entered the market at a premium relative to the average price for the subclass. However, even these drugs were discounted significantly relative to the price leader.10
Price Trends by Therapeutic Category
We identified major subclasses for each of the eight therapeutic categories examined. For a given therapeutic category, we calculated annual weighted mean prices for each subclass and for generics in each of the subclasses that had experienced generic entry. Given the general trend for rebating over our study period noted above, price trends that incorporated rebates would tend to show less price inflation than is indicated here. In terms of price comparisons across subclasses, since we would expect little or no rebating for old classes with significant generic entry, the price levels of the newer subclasses will tend to be somewhat overstated relative to older subclasses. In addition, we did not attempt to quality-adjust the price levels for the various classes. If the prices in the new and the older classes were quality-adjusted for differences in efficacy, safety, and convenience, then price differentials could diminish or vanish. Further research on making quality-adjustments to prescription drug pricing data is needed before conclusions can be drawn for many classes (Berndt et al., 1999; Suslow, 1996).
The pricing data for generics will be affected by the addition over time of new molecules going off patent. The general pattern is for generic drug prices to fall over time as additional manufacturers market the generic. However, this tendency can be masked at times if other brand name drugs in the class go off patent during the study period. The results for each category are as follows.
Antiarthritics
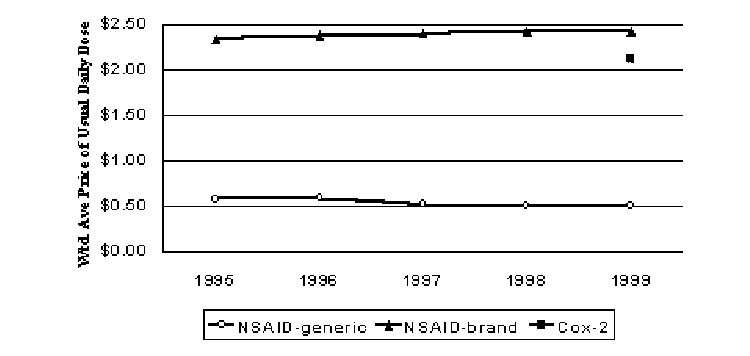
A new subclass (Cox-2 inhibitors) for the arthritis market emerged in 1999 and experienced rapid growth. The Cox-2 inhibitors are thought to have a better side effect profile than older NSAIDs, but only for those patients who risk serious gastrointestinal distress from using the older NSAIDs. Efficacy is apparently similar. Thomas and Ritter (2000) discusses the treatments available and drug utilization in this market. Figure 4 indicates that this new subclass entered the market at prices that were somewhat below the average price for other NSAIDs, but at prices that were four times that of the generic NSAIDs that had taken a large share of the market. Reports indicate that prices were discounted relative to other brands in order to reduce potential payer concerns about cost (NDA Pipeline, 1999). Real prices for the NSAIDs increased slightly over the study period. However, real prices for generic NSAIDs declined slightly. However, because the use of generics increased over the period, the average real price for the NSAIDs when generics are included as lower cost substitutes for their brand counterparts declined 28% from 1995 to 1999.
Figure 4. Pricing Trends for Antiarthritics (1999$)
Antidepressants
Figure 5. Pricing Trends for Antidepressants (1999$)
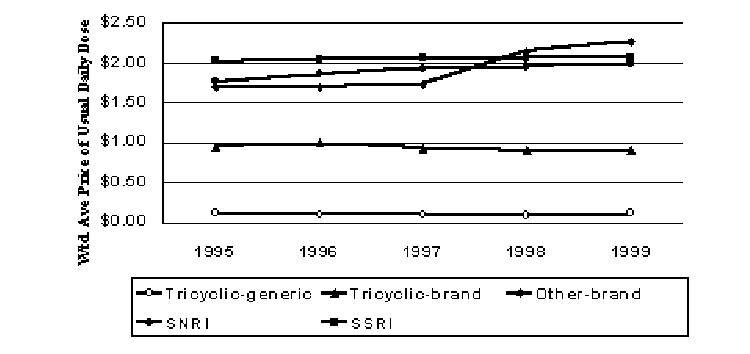
Selective serotonin reuptake inhibitors (SSRIs) have come to dominate the antidepressant market since the first entrant Prozac was approved in 1987. They are thought to have a better side effect profile than the older classes, such as the tricyclics.11 Selective norepinephrine reuptake inhibitors (SNRIs) and other miscellaneous brand name compounds are priced similarly to the SSRIs, but they have not captured large shares of the market. Since 1996, real price trends for SSRIs have been flat, while tricyclic prices have declined slightly in real terms (Figure 5). When generic tricylics are included with their brand counterparts, real prices for the tricyclics declined 34% from 1995 to 1999.
Antihistamines
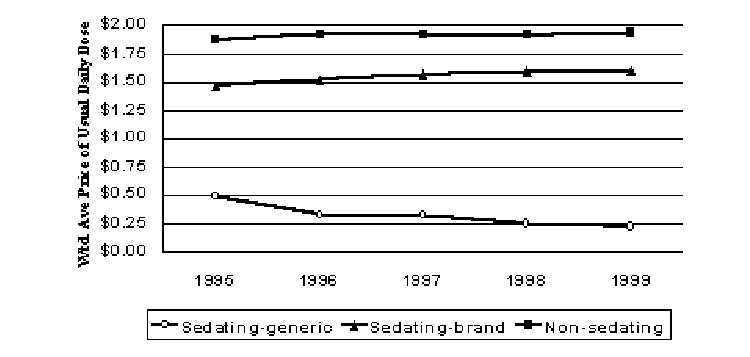
Real prices for sedating antihistamines increased steadily over the study period, rising 9% above inflation from 1995 to 1999. Real prices for generic sedating antihistamines fell by half over the study period. When generic sedating antihistamines are included with their brand counterparts real prices increased 11% faster than general inflation from 1995 to 1999 because the relative use of generics declined with the introduction of new brands. However, the newer non-sedating class has largely displaced this older class of compounds. After increasing 2.4% faster than inflation in 1996, real prices for the non-sedating antihistamines were essentially constant over the next two years and increased about 1% in 1999 (Figure 6).
Figure 6. Pricing Trends for Antihistamines (1999$)
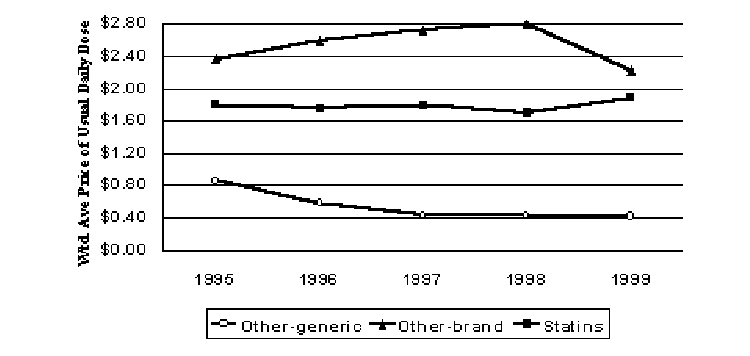
Antihyperlipidemics
The standard of care in the cholesterol-lowering market has been the use of HMG-CoA reductase inhibitors (statins). The first statin approved for marketing in the United States was Mevacor in 1987. Since then, five other statins have beem approved (Pravachol®, Zocor®, Lescol®, Lipitor®, and Baycol®). Thomas and Ritter (2000) describes utilization trends in this therapeutic area. Older classes of compounds used to lower lipid levels include bile acid sequestrants and fibric acid derivatives. While daily dose pricing was higher for brand-name drugs in these other classes, they accounted for only 3% of days of use in 1999. The real prices for the statins rose and fell somewhat for 1996 to 1998, but they increased notably by 11% for 1999 (Figure 7).
Figure 7. Pricing Trends for Antyhyperlipidemics (1999$)
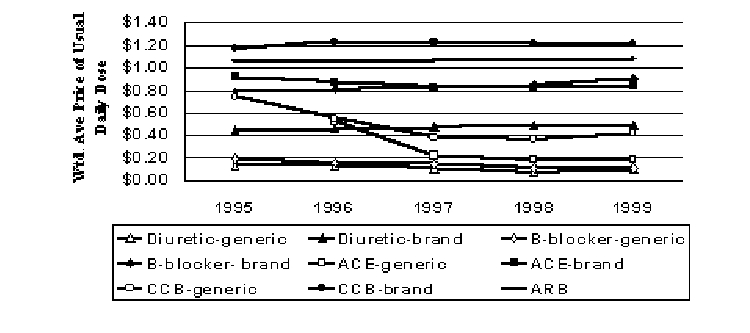
Antihypertensives
The antihypertensive market is crowded, with multiple drugs in numerous categories. Some of the subclasses are very old, but are still useful. Diuretics is the oldest subclass, followed closely by beta-blockers. ACE-inhibitors are slightly older than the calcium channel blocker (CCB) class. ARBs is the newest class. Real prices have increased for brand-name diuretics and beta-blockers, but these subclasses have many low-cost generic entrants (Figure 8). When generics are included with their brand counterparts, real prices fell 32% and 23% from 1995 to 1999 for diuretics and beta-blockers, respectively. Brand ACE-inhibitors experienced a decline of 9% in real prices over four years, but brand CCBs had a 4% increase in real prices over the same period. CCBs are the highest price class. However, both ACE-inhibitors and CCBs have some generic competition. When generics are included with their brand counterparts, real prices fell 13% and 7% from 1995 to 1999 for ACE-inhibitors and CCBs, respectively. ARBs are a new class for the study period. The first entrant in this class, Cozaar, entered the market in 1995. Real prices for ARBs have increased only slightly. Prices for the individual drugs in this class are very similar.
Figure 8. Pricing Trends for Antihypertensives (1999$)
Antiulcer
Figure 9. Pricing Trends for Antiulcer Drugs (1999$)
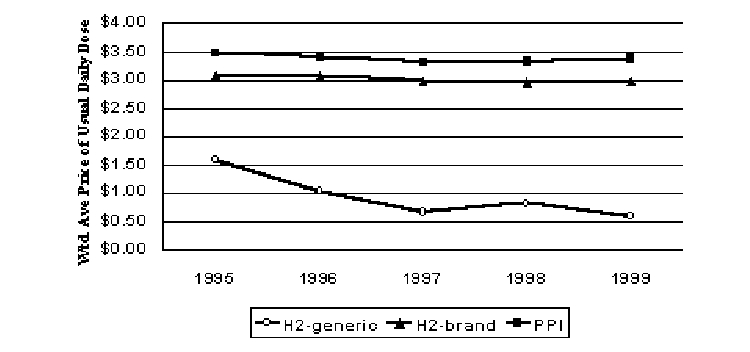
Drugs to treat ulcers and gastoesophageal reflux disease have been among the fastest growing. H2-receptor antagonists (H2RAs) had dominated the market until recently, as both brand and generic H2-blockers are being supplanted by the newer proton pump inhibitor (PPI) class of compounds. Thomas and Ritter (2000) discusses treatment and analyzes utilization trends for these drugs. Two of the four H2RAs had generic competition during the study period. The PPIs are about 13% more expensive than the brand H2RAs, but about six times more expensive than the generic H2RAs in 1999 (Figure 9). However, all segments experienced real price declines over the study period. The real prices for both PPIs and brand H2RAs fell 3% from 1995 to 1999. Real prices for the H2RA generics fell by about two-thirds over this period. When generic H2RAs are included with their brand counterparts, real prices for H2RAs fell 55% from 1995 to 1999.
Oral Cephalosporins
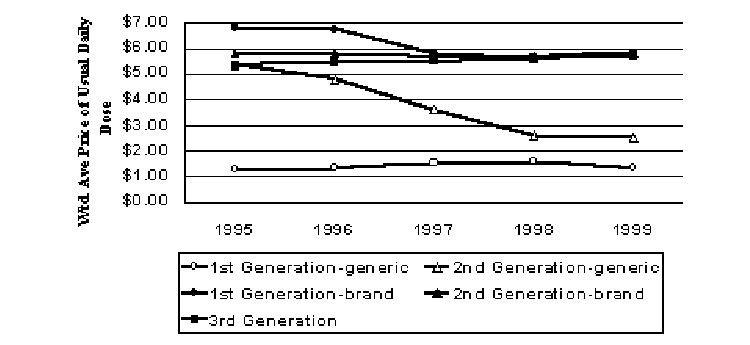
Cephalosporins are antibiotics that are divided into generations that differ in their spectrum of activity against bacterial infections. Third generation cephalosporins are newer, but second generation cephalosporins are widely used. During the early part of the study period, the third generation cephalosporins were actually priced lower on a daily basis than the earlier generations (Figure 10). However, by the end of the period they were priced similarly. Real prices fell for both first and second generation brand cephalosporins during the study period, but they rose 8% from 1995 to 1999 for the third generation products. When generics are included with their brand counterparts, real prices fell 46% and 8% from 1995 to 1999 for first and second generation oral cephalosporins, respectively.
Figure 10. Pricing Trends for Oral Cephalosporins (1999$)
Macrolides
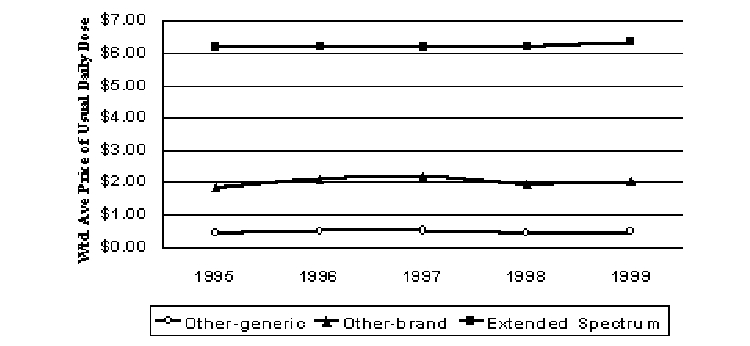
Macrolides are a different type of antibiotic. They prevent bacteria from reproducing, while the cephalosporins and many other antibiotics kill bacteria. The newer products in this class are referred to as extended spectrum macrolides (Biaxin®, Dynabac®, and Zithromax®). The extended spectrum macrolides are priced much higher than the other brand and generic products in this class (Figure 11). Real prices for the extended spectrum macrolides increased a little less than 3% over the four years from 1995 to 1999. When generics are included with their brand counterparts, real prices rose 7% from 1995 to 1999 for the other macrolide antibiotics.
Figure 11. Pricing Trends for Macrolide Antibiotics (1999$)
SUMMARY
We found higher new drug launch prices for compounds that represent a gain over existing therapy and for drugs that are indicated primarily for shorter-term use. These results suggest that the prices that are set for new drugs bear a relation to clinical value and to consumer price sensitivity. This is consistent with prior research covering an earlier period (Lu and Comanor, 1998). Price levels (as opposed to price increases) for prescription drugs therefore depend on the nature of pharmaceutical innovation, and not just on the pace of innovation. Other things being equal, the impact that new drugs will have on pharmaceutical costs will tend to be greater for more innovative products, both because higher prices can be charged for products that are more highly valued by patients and prescribers and because utilization will be greater as patients and prescribers demand these new therapies.
The evidence on launch prices for follow-on products indicates an additional source of variability in the contribution that new drugs make to prescription drug expenditure changes. Our examination of new drug introductions into existing classes of compounds revealed that the majority of these new drugs are launched at discounts (sometimes substantial) to both the class price leader and to the average price in the class. Thus, these new drugs tend to reduce pharmaceutical expenditures directly. They may also do so indirectly as their availability increases the leverage that payers have to extract rebates from manufacturers.
It has often been suggested that new classes of compounds with higher prices will tend to displace older therapies, thereby increasing pharmaceutical costs. Our data generally support the notion that new classes are priced higher than old classes, but even this outcome is not uniform. Several of the classes we examined were priced lower than the brand name drugs in some older classes.
Finally, our results for eight therapeutic categories, representing half of all prescription drug expenditures, showed that average prices for drug subclasses did not, for the most part, increase substantially in excess of general price inflation. In many cases, real prices were flat or had declined slightly. Even in those classes where real prices increased, the outcome could, in theory, be partially or completely reversed if rebates were taken into account. These results are consistent with recent studies on prescription drug spending trends that have shown that pure price inflation has not been a major contributor to rising prescription drug costs.
REFERENCES
Barents Group (1999). Factors Affecting the Growth of Prescription Drug Expenditures. National Institute of Health Care Management Research and Educational Foundation, Washington DC.
Berndt, ER, Pindyck, RS, and P Azoulay (1999). “Network Effects and Diffusion in Pharmaceutical Markets: Antiulcer Drugs,” NBER Working Paper Series: #7024, Cambridge, MA.
The Boston Consulting Group, Inc. (1993). The Changing Environment for U.S. Pharmaceuticals: The Role of Pharmaceutical Companies in a Systems Approach to Health Care, New York: The Boston Consulting Group, Inc., April 1993.
Dubois RW, AJ Chawla, CA Neslusan et al. (2000). “Explaining Drug Spending Trends: Does Perception Match Reality?” Health Affairs 19(2):231-239.
Express-Scripts (2000). 1999 Drug Trend Report. www.express-scripts.com.
Kolassa, EM (1997). Elements of Pharmaceutical Pricing, New York: Haworth.
Lu ZJ and WS Comanor (1998). “Strategic Pricing of New Pharmaceuticals,” Review of Economics and Statistics, 80(1), 108-118.
Merck-Medco (2000). Managing Pharmacy Benefit Costs www.merckmedco.com.
The NDA Pipeline-1998 (1999). “Celebrex Priced to Minimize Reimbursement Issues - $2.42 Per Day,” Chevy-Chase, MD: F-D-C Reports, 505-506.
Personal Communications. Kevin Waite and James Herrick of PCS Health Systems, Inc., April-June 2000.
Reekie WD (1978). “Price and Quality Competition in the United States Drug Industry,” Journal of Industrial Economics, 26, 223-237.
Reekie WD (1998). “How Competition Lowers the Cost of Medicines,” PharmacoEconomics, 14, Supplement 1, 107-113.
Schneider Institute for Health Policy (2000). Factors Driving Prescription Drug Expenditure Increases. Unpublished data.
Suslow VY (1996). “Measuring Quality Change in the Market for Anti-Ulcer Drugs,” in RB Helms (ed.), Competitive Strategies in the Pharmaceutical Industry, Washington DC: AEI Press, 49-72.
Thomas CP and G Ritter (2000). “Drug Utilization Trends by Therapeutic Class” [http://aspe.hhs.gov/health/reports/Drug-papers/index.htm].
Towse A and T Leighton (1999). “The Changing Nature of NCE Pricing of Second and Subsequent Entrants,” in Sussex J and N Marchant (eds), Risk and Return in the Pharmaceutical Industry, London: Office of Health Economics, 91-105.
U.S. General Accounting Office (1997). Drug Prices: Effects of Opening Federal Supply Schedule for Pharmaceuticals Are Uncertain, Washington DC: GAO/HEHS-97-60.
Footnotes
(1) Mail-order claims were excluded from these analyses. Unit prices are generally 5% lower for brand mail-order prescriptions and prescription sizes are often three times the amount supplied for comparable retail prescriptions. Mail-order claims were a small, but growing, proportion of the claims data provided. In essence, mail-order adds an additional price for each product presentation. The use of mail-order can also vary by drug. To provide cleaner price comparisons, we restricted analysis to retail prescriptions.
(2) At this time, formularies were typically open, i.e. all drugs were covered regardless of whether they were listed in the formulary.
(3) The vast majority of FSS prices are determined by the Federal Ceiling Price (FCP), a legislatively mandated discount for four Federal agencies (GAO, 1997). The FCP is 76% of the nonfederal average manufacturer price (NFAMP), adjusted by an excess inflation rebate factor if the drug’s price increased faster than the CPI in the previous year. The NFAMP is a weighted average of prices paid by wholesalers for the nonfederal class of trade, net of cash discounts and chargebacks. Thus, FSS prices are likely highly correlated with manufacturer prices.
(4) In a small number of cases the first month at which a price could be calculated resulted in an unusual value that was based on just one or two prescriptions for the month. In those cases, the launch month was taken to be the following month, and the launch price is the price for that month.
(5) Eligibility for our data was constant for a given year, but it increased over the study period. Thus, the number of days of use is dependent on the year in which the prescriptions were filled. For the purpose of computing the weights, we indexed the days of use for a month to reflect the relative number of enrollees for the fiscal year in which the month fell.
(6) The share of expenditures for these classes had declined slightly from 53% in 1995 to 50% in 1997, but was constant thereafter.
(7) A new drug may be launched any time after approval. Drugs approved late in fiscal year 1995 can be launched in 1996. However, we cannot be certain about when drugs approved in 1994 are launched. In this way, the count of launches for 1995 can be relatively low. The number of launches was 20, 25, 26, and 20 for 1996, 1997, 1998, and 1999, respectively.
(8) The new drugs accounted for only 7% of all days of use during the study period. However, the ultimate impact of new drugs is underestimated by these data both because these drugs had not been available for the whole period and because their diffusion rates may increase over time.
(9) We also regressed the launch price for a new drug on year of launch, therapeutic rating, duration of use, and dosing frequency. The evidence did not support a time trend. However, statistically significant coefficients with the expected signs were found for therapeutic rating and duration of use. Daily dosing frequency was interacted with duration of use. It was hypothesized that, other things being equal, more frequent dosing would be associated with lower prices. The coefficients for dosing frequency interacted with acute and subchronic use were significant with the expected signs. However, the coefficient for chronic use was significant with the opposite sign. This result may have been caused by the inclusion of the AIDS antivirals and other medications in this category with relatively high prices and very frequent dosing.
(10) If rebates are given in a class, then post-rebate relative prices for individual drugs in a class may differ from the relative prices shown here. However, new entrants have not established a market presence at launch. Their manufacturers should therefore be at least as likely as most others to rebate aggressively if the new entrant’s transaction price is equal to or higher than the average post-rebate price for the class. It seems unlikely then that rebating would completely eliminate or reverse the discounting of follow-on product prices relative to incumbent prices that we have observed. Rebates would also not be relevant to the uninsured market.
(11) MAO inhibitors are another well-defined old class of antidepressants, but the data indicate that their utilization is extremely low. Thus, they are not reported on separately.
