Non-Fatal Opioid Overdose and Associated Health Outcomes: Final Summary Report
Jon Zibbell, PhD, Jennifer Howard, PhD, Sarah Duhart Clarke, BS, Abigail Ferrell, BA, and Sarita L. Karon, PhD
RTI International
September 2019
Printer Friendly Version in PDF Format (33 PDF pages)
ABSTRACT
Non-fatal opioid overdose (NFOO) is a significant cause of opioid-related morbidity in the United States. As the number of NFOOs continues to grow, it is important to understand the short- and long-term consequences of NFOO. This report examines the existing literature on the acute and chronic health and functional outcomes of individuals who experience NFOO, and also identifies differences in outcomes for NFOOs involving illicitly-manufactured fentanyl.
This report was prepared under contract #HHSP233201600021I between HHS’s ASPE/DALTCP and Research Triangle Institute. For additional information about this subject, you can visit the DALTCP home page at https://aspe.hhs.gov/office-disability-aging-and-long-term-care-policy-daltcp or contact the ASPE Project Officers, Daniel Schwartz, DEB Potter and Helen Lamont, at HHS/ASPE/DALTCP, Room 424E, H.H. Humphrey Building, 200 Independence Avenue, S.W., Washington, D.C. 20201; Daniel.Schwartz@hhs.gov.
DISCLAIMER: The opinions and views expressed in this report are those of the authors. They do not reflect the views of the Department of Health and Human Services, the contractor or any other funding organization. This report was completed and submitted on March 4, 2019.
TABLE OF CONTENTS
1. BACKGROUND
2.1. Scoping Review Framework
2.2. Identification of Relevant Publications
2.3. Selection of Publications
2.4. Data Extraction and Synthesis
3. RESULTS
3.1. Effects of Non-fatal, Opioid-Involved Overdoses
3.2. Differing Health Outcomes between Illicitly-Manufactured Fentanyl, Heroin, and Prescription Opioids
3.3. Interaction between Pre-existing Conditions and Subsequent Overdose
3.4. Health Service Utilization
4. DISCUSSION
4.1. Discussion of Results
4.2. Gaps in the Literature
4.3. Importance of Information
4.4. Study Limitations
5. CONCLUSION
APPENDIX A: Example Search Strategy--PubMed
LIST OF FIGURES
- FIGURE 1: Publication Selection Process
LIST OF TABLES
- TABLE 1: Search Terms
ACRONYMS
The following acronyms are mentioned in this report and/or appendix.
| ASPE | Office of the Assistant Secretary for Planning and Evaluation |
|---|---|
| CDC | Centers for Disease Control and Prevention |
| CNS | Central Nervous System |
| DEA | Drug Enforcement Administration |
| IMF | Illicitly-Manufactured Fentanyl |
| MMWR | Morbidity and Mortality Weekly Report |
| MOR | µ-Opioid Receptor |
| MPTP | Illicitly-Manufactured Meperidine (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) |
| MRI | Magnetic Resonance Imaging |
| NFOO | Non-Fatal Opioid-involved Overdose |
| NIH | National Institutes of Health |
| OIRD | Opioid-Induced Respiratory Depression |
| RTI | Research Triangle Institute |
| SAMHSA | Substance Abuse and Mental Health Services Administration |
EXECUTIVE SUMMARY
The Office of the Assistant Secretary for Planning and Evaluation (ASPE) contracted with RTI International to conduct a scoping review of the scientific literature on non-fatal opioid-involved overdose (NFOO) to better understand the health outcomes and health care implications associated with opioid epidemic. NFOO is a significant cause of opioid-related morbidity in the United States, and is of heightened interest to ASPE given the sharp increase in fatal and non-fatal opioid overdoses as a result of the current fentanyl crisis. Compared to the first and second phases of the opioid epidemic, when the majority of opioid-related morbidity and mortality was associated first with prescription opioids and then with heroin, illicitly-manufactured fentanyl (IMF) is now the leading cause of fatal opioid overdoses (CDC, 2017; O'Donnell, Gladden, & Seth, 2017). People who consume IMF, either wittingly or unwittingly, are at higher risk for accidental overdose, experience comparatively atypical overdose-related side effects, have unique risk profiles, and are at higher risk for poorer health outcomes (Somerville, 2017). To our knowledge, no investigation has been conducted to examine whether the sharp increase in NFOOs over the last several decades is negatively impacting overdose victims' chronic physical and mental health outcomes. To investigate the status of the scientific literature regarding the consequences of NFOO on victims' physical and mental health, we conducted a scoping review of the existing knowledge in multiple stages: (1) publication identification; (2) selection of publications; and (3) data extraction, charting, and synthesis. Key findings from our review are presented below.
Key Findings
Findings from this scoping review confirm that NFOO is an important cause of morbidity among persons who misuse and abuse prescription and/or illicitly-manufactured opioids (e.g., heroin). The findings are summarily presented below and organized by three topics: (1) health outcomes associated with NFOO; (2) differing health outcomes for NFOOs that involve prescription opioids vs. heroin vs. IMF; and (3) the interaction between overdose victims' pre-existing health conditions and the health outcomes associated with NFOO.
Health Consequences of Non-Fatal Opioid-involved Overdoses
Opioid-Induced Respiratory Depression
Opioid-induced respiratory depression (OIRD) was identified as a primary complication and the most dangerous source of adverse health outcomes associated with NFOO (King, Morris & Schahmann, 2015). The health consequences associated with OIRD include a range of acute complications that develop during or immediately after a NFOO event. OIRD most often presents as acute respiratory compromise with victims reverting to normal breathing activity either on their own or with the use of an opioid overdose reversal drug like naloxone (Wermeling, 2015). Most NFOO victims recover from OIRD and present only minor health consequences (e.g., dizziness, confusion, ineptness) lasting minutes to hours (Sporer & Dorn, 2001; Buajordet et al., 2004; Olfson et al., 2018).
Brain Injury
Most literature identifying chronic complications associated with NFOO concerns the development of hypoxia-related brain injuries and subsequent complications caused by a prolonged version of OIRD that contributes to a protracted deficiency of oxygen reaching the body's tissues (Ueno et al., 2018; Somerville et al., 2017; Betts, Ritter, & Kubal, 2012; Salazar & Dubow, 2012). The range of health complications associated with opioid-induced hypoxia includes kidney failure, heart complications, neurologic consequences, seizures, nerve damage, temporary motor paralysis, fluid buildup in the lungs, stroke, and pneumonia from inhaling vomitus (Marino et al., 2018; Betts, Ritter & Kubal, 2012; O'Brien & Todd, 2009; Fitzgerald et al., 2000).
Several studies have employed brain scans and demonstrate susceptibility of certain regions of the brain to hypoxic injury, with short-term memory loss and changes in cognitive and physical functioning documented. Stroke has also been observed in rare NFOO cases. Opioid-induced hypoxic brain injury has also been shown to contribute to the development of severe disability, including mental disorientation, the loss of body movement, gait changes, incontinence, paralysis of lower extremities, and changes in behavior due to altered mental states, among a range of other presentations (Buttner et al., 2000). Slower reaction time, impaired memory (amnestic syndrome), reduced motor skills, and diminished physical functioning have all been documented as hypoxic-related brain injuries associated with NFOO (Barash, Somerville, & DeMaria, 2017; Ueno, et al. 2018; Somerville et al., 2017; Betts, Ritter, & Kubal, 2012; Salazar & Dubow, 2012).
Differences in Effects of Overdoses between Single, Multiple, and Recurring Opioid Overdose
There is relatively scarce literature detailing with health outcomes that develop among victims who experience more than one NFOO. However, one study demonstrated increased risk for hypoxic brain damage among persons who experience multiple or recurring opioid overdose events due to repeated and prolonged hypoxia (Zamora et al., 2015).
Differing Health Outcomes between Illicitly-Manufactured Fentanyl, Heroin, and Prescription Opioids
Fentanyl, a synthetic opioid 100 times more potent than morphine, stands apart from heroin and other semi-synthetic opioids in these findings by its relatively high potency and rapid onset of action when smoked, sniffed, or injected. These characteristics are the result of fentanyl's high lipid profile, which allows its molecules to dissolve quickly in fats and rapidly cross the blood-brain barrier (Burns et al., 2016). A rapidly growing supply of IMF in existing heroin markets has substantially increased the number of fentanyl overdose deaths over the last 5 years, with fentanyl deaths surpassing the number of deaths from both heroin and prescription opioids in 2016 and 2017. Even so, few studies have examined the specific health complications associated with fentanyl overdose compared to overdoses involving prescription opioids or heroin. The medical research literature does identify several side effects resulting from the clinical use of intravenous fentanyl in hospital settings, with skeletal muscle rigidity and chest wall rigidity the most commonly reported (Burns et al., 2016). Chest wall rigidity, colloquially referred to as "wooden chest syndrome," is understood as the acute inability to ventilate or move the chest wall; this effect poses serious complications for ventilating overdose victims suffering from OIRD in non-hospital settings.
Interaction between Pre-existing Conditions and Subsequent Overdose
Few studies included in our review examined the interplay between pre-existing physical or mental health conditions among people consuming opioids and subsequent NFOO among this population. Some mental health conditions, such as depression and suicidality, have been strongly associated with opioid use disorder and drug overdose. Comorbid mental health conditions are identified as risk factors for drug overdose, but it is difficult to distinguish the degree to which they are influenced or exacerbated by NFOO in retrospective studies (Neale, 2000). Beyond depression, suicide attempts are shown to be associated with unintentional overdose (Neale, 2000; Bradvick et al., 2007), and suicide risk is correlated with higher numbers of drug overdoses among victims (Bradvick et al., 2007). It is unclear, however, to what degree suicidality is exacerbated by drug overdose and vice versa. Symptoms resulting from opioid-induced hypoxia described above may exacerbate pre-existing disabilities and conditions among NFOO victims. For example, declines in a person's cognitive and physical functioning due to having one or more NFOO may present difficulties for managing their existing chronic illnesses, such as diabetes, addiction, or clinical depression (King, 2015).
Discussion and Conclusion
As the number of NFOO continues to grow, it is paramount that health care and treatment providers as well as policymakers have information on how NFOO can impact overdose victims' mental and physical functioning in both the short-term and long-term. This is especially true for the population of people who consume street opioids, given their increased risk for fentanyl consumption. Without this information, providers and policymakers will be hindered in their ability to provide appropriate and high-quality health care and supportive services for overdose victims. Further, they will be providing care to this population with no real understanding or guidance for how to identify the longer-term health consequences that may derive from recurring hypoxic overdose events, especially those associated with IMF use.
The purpose of this scoping review was to better understand the health and functional outcomes associated with NFOO. After reviewing 48 peer-reviewed and grey publications identified and selected for review, our analysis concluded that numerous clinical complications are associated with NFOO. Among these, most of the concentration focuses on acute outcomes stemming from respiratory depression and hypoxia. The current lack of longitudinal studies examining the chronic complications resulting from non-fatal opioid overdose events restricts our ability to understand the longer-term effects of opioid overdose on victims' physical, mental, and cognitive functioning. Most critical are the downstream effects of a fentanyl overdose crisis that, between 2016 and 2017, killed males aged 25-44 at an age-adjusted rate of 27 per 100,000. The fentanyl crisis has contributed to a population of fentanyl consumers who overdose with more frequency than they did with prescription opioids or heroin because of fentanyl's higher potency and more rapid duration of action.
Potentially fruitful avenues for future research include an additional review of the literature with expanded search strategies, the collection of primary data through interviews with key subject matter experts and NFOO victims, and longitudinal studies utilizing secondary data. A longer-term recommendation for clinical research may include expanding current brain injury studies, such as the Chronic Traumatic Encephalopathy program at Boston University, to include fatal overdose victims who experienced recurring non-fatal hypoxic overdose events prior to death.
1. BACKGROUND
The opioid epidemic in the United States continues to generate unprecedented levels of morbidity and mortality. More than 70,237 people died from a drug overdose in 2017, and over two-thirds of those deaths involved an opioid (CDC, 2017). The rise in overdose deaths since 2013 has been largely attributable to the introduction of fentanyl (a synthetic opioid about 50 times more potent by weight than heroin) within illicit drug supplies. Illicitly-manufactured fentanyls (IMFs)[1] typically enter the country via illicit drug channels alongside heroin and other illicit drugs, before being sold either by itself or mixed with heroin (DEA, 2017). IMFs have also been distributed in the form of counterfeit prescription drugs, such as OxyContin or Xanax, which have been linked to overdose outbreaks in multiple states (Green & Gilbert, 2016).Since 2016, prescription opioid-involved overdose deaths have been declining, and deaths from heroin and IMFs are now the main drivers of opioid-related mortality (O'Donnell, Gladden, & Seth, 2017), with IMFs responsible for a majority of opioid overdose deaths since 2016 (CDC, 2017). According to the Centers for Disease Control and Prevention's National Syndromic Surveillance Program, opioid overdose emergency department visits increased roughly 30% from third quarter 2016 (July-September) to third quarter 2017 in all five United States regions--the Midwest showed the greatest increase (69.7%), followed by the West (40.3%), Northeast (21.3%), Southwest (20.2%), and Southeast (14.0%) (CDC, 2018). The increases in opioid overdose rates are consistent with previous reports indicating that non-fatal overdose hospitalizations and emergency department visits associated with heroin and IMF poisonings are on the rise (CDC, 2017).
Opioid abuse and non-fatal opioid overdose have been linked to concurrent upsurges in overdose-related health complications. For example, a 2014 study in Journal of the American Board of Family Medicine documented increases in adverse health outcomes associated with increased duration of opioid use (Paulozzi et al., 2014), and the National Institutes of Health (NIH) have linked opioid poisoning with hypoxia-related complications such as organ system damage, cardiac arrest, stroke, psychosis, and death (NIH, n.d.). The current IMF crisis has contributed substantially to the sharp increase in fatal and non-fatal opioid overdose, with IMF contributing to the development of unusual non-fatal overdose presentations. For example, a recent case report from Annals of Internal Medicine identified increases in unusual amnestic syndrome associated with the combined use of IMF and cocaine (Duru et al., 2018).
Similarly, increases in anecdotal reports in the United States have identified increases in previously unseen side effects associated with IMF-involved overdoses, such as rigid chest syndrome, acute rigor mortis, and memory loss (Burns, 2016). People who use heroin and IMF are at higher risk for recurring overdose, they experience unusual overdose-related side effects, and they have unique health concerns or are at higher risk of poorer health outcomes (Somerville, 2017). Taken together, the increase in both known and unfamiliar/unusual health outcomes associated with opioid poisoning, and specifically IMF-involved overdose, suggests the need for a rigorous review of the literature to investigate existing scientific knowledge on the short and long-term effects of non-fatal opioid overdose, with a specific focus on IMF and heroin.
The Office of the Assistant Secretary for Planning and Evaluation (ASPE) contracted with RTI International to conduct a scoping review to better understand the health and functional outcomes implications associated with non-fatal opioid-involved overdose (NFOO). This study addressed the following research questions:
-
What effects do non-fatal, opioid-involved overdoses have on overdose victims' physical and mental health, including but not limited to functional and cognitive impairment?
-
How do physical and mental health outcomes, including but not limited to functional and cognitive impairment, associated with non-fatal, opioid overdoses that involve IMF and fentanyl analogs (e.g., acetylfentanyl, furanylfentanyl, carfentanil, etc.) differ from the physical and mental health outcomes of non-fatal overdoses involving heroin and other semi-synthetic opioids (e.g., oxycodone)?
-
How do non-fatal overdoses interconnect with or exacerbate other physical conditions and health outcomes among overdose victims, such as memory loss, cognitive impairment, mental illness, and increased physical disability?
2. DATA AND METHODS
2.1. Scoping Review Framework
Our methods were guided by a framework for conducting scoping reviews originally proposed by Arksey & O'Malley (2005) and later expanded by Levac & colleagues (2010; Colquhoun et al., 2014). This section describes the steps we undertook to ensure that we explored the full breadth of the literature and obtained comprehensive information about the short-term and long-term outcomes of non-fatal overdoses.
2.2. Identification of Relevant Publications
We began this scoping review by creating a data charting tool to capture and organize the information identified during the course of the review. We designed our tool (i.e., an Excel database) to capture:
-
Publication information, including author(s) names, study title, publishing journal or organization/agency, publication year, and abstract.
-
Reviewer initials, reviewers' determinations for including or excluding a publication during each step of the review, and reasons for exclusion at each step.
-
Study data extracted during the review process, such as study drug of focus, methodology, and findings.
The data charting tool allowed reviewers to synthesize information by research question and subquestion/theme and provide additional notations that may be pertinent as, "additional notes/noteworthy findings." In addition, the tool allowed reviewers to indicate key articles and additional databases noted in the reviewed publication that were further investigated for possible inclusion in the review.
After creating a charting tool, we took a two-part approach to identify relevant studies for inclusion in the review: we conducted a structured search of academic literature and a thorough review of several grey literature sources. The two searches were composed of multiple steps that included developing preliminary lists of search terms, exclusion and inclusion criteria, and databases and organizational and agency websites for conducting the searches. Table 1 shows the organizations, agencies, and publications searched as well as the search terms used.
| TABLE 1. Search Terms | |
|---|---|
| Peer-Reviewed Literature | Grey Literature |
| 1998-present English language Databases: PubMed, Embase, Web of Science, PsycINFO, Toxline, ScienceDirect, JSTOR, Google Scholar Search Terms (for first search): opioid* OR fentanyl OR "illicitly-manufactured fentanyl" OR "illicitly-made fentanyl" OR acetylfentanyl OR furanylfentanyl OR carfentanil OR heroin OR oxycodone OR oxymorphone OR Opana OR hydromorphone OR Dilaudid OR OxyContin OR hydrocodone OR Vicodin OR "novel psychoactive substance*" OR methadone AND overdos* AND ("non-fatal" OR nonfatal OR unintentional* OR accidental* OR accident) Search Terms (for second search): opioid* OR fentanyl OR "illicitly-manufactured fentanyl" OR "illicitly-made fentanyl" OR acetylfentanyl OR furanylfentanyl OR carfentanil OR heroin OR oxycodone OR oxymorphone OR Opana OR hydromorphone OR Dilaudid OR OxyContin OR hydrocodone OR Vicodin OR "novel psychoactive substance*" OR methadone AND overdos* OR postoverdos* AND hypoxic hypoxia OR arterial hypoxia Search Terms (for third search): opioid* OR fentanyl OR "illicitly-manufactured fentanyl" OR "illicitly-made fentanyl" OR acetylfentanyl OR furanylfentanyl OR carfentanil OR heroin OR oxycodone OR oxymorphone OR Opana OR hydromorphone OR Dilaudid OR OxyContin OR hydrocodone OR Vicodin OR "novel psychoactive substance*" OR methadone AND overdos* OR postoverdos* AND (longitudinal OR follow-up) AND (intervention* OR medication assisted treatment*) | 1998-present English language Organizations/Agencies:
Research Institutes:
Search terms: opioid OR fentanyl OR acetylfentanyl OR furanylfentanyl OR carfentanil OR heroin OR oxycodone OR oxymorphone OR Opana OR hydromorphone OR Dilaudid OR OxyContin OR hydrocodone OR Vicodin OR methadone AND Overdose OR Nonfatal |
Our team worked with an experienced librarian to refine the approach to identifying relevant studies at multiple points in the process. An example search strategy can be found in Appendix A.
Once we identified a list of initial studies, we downloaded the publications into an Endnote database, removed duplicate publications, and completed a high-level review of the resulting publication titles. Our team met with the librarian to discuss the initial results and refined the exclusion and inclusion criteria to focus the scope to identify relevant studies. We paid special attention to ensure that all relevant systematic reviews were adequately captured. This step was completed three times to ensure all relevant peer-reviewed publications were captured. Once we agreed that all relevant studies were identified from all relevant sources, a team member imported the publication information and abstracts from selected relevant publications into the data charting tool.
Concurrent to this review, members of the research team completed a search of the grey literature. They identified major organizations and governmental agencies and used key search terms to identify relevant resources from the website of each organization and agency. All resources identified from the search of the grey literature were included in the data charting tool for additional review.
2.3. Selection of Publications
After independent review by two members of the research team, we selected studies for inclusion. The purpose of the abstract review was two-fold: (a) to further refine the inclusion/exclusion criteria; and (b) to begin the process of eliminating studies from further analysis. A third, senior reviewer was included as a "tie breaker" in the instances where there was disagreement between the reviewers. Only those publications that both reviewers indicated should be included were reviewed further. A reason for exclusion was included for all publications excluded from further analysis.
Two independent reviewers from the research team then assessed each article selected for inclusion. Like the abstract review, this assessment served two purposes: to further narrow the studies included in the review (i.e., remove studies that were not relevant to the three research questions) and to extract data using a qualitative thematic analysis approach. We focused on the findings of the publications, but also summarized content from other sections of the publications that were helpful in answering the research questions or providing information on other relevant studies.
During each level of the review, we evaluated the quality of the resources identified and excluded resources that did not have the necessary methodological rigor for the review. We evaluated quality based on the number or type of citations (e.g., publications without citations were excluded) included in the publication as well as a minimum level of detail in the publication (e.g., conference proceedings without detailed notes and citations were excluded).
We identified 254 articles through a search of the peer-reviewed and grey literature. One hundred and eight of these were reviewed in full for relevance; 60 of that 108 were culled based on determination that the publication type as unsuitable (six), the study focused on a substance not included in the research questions (four), or they were otherwise not relevant (50). Data was extracted and synthesized from the 48 publications that met our inclusion/exclusion criteria. See Figure 1.
| FIGURE 1. Publication Selection Process |
|---|
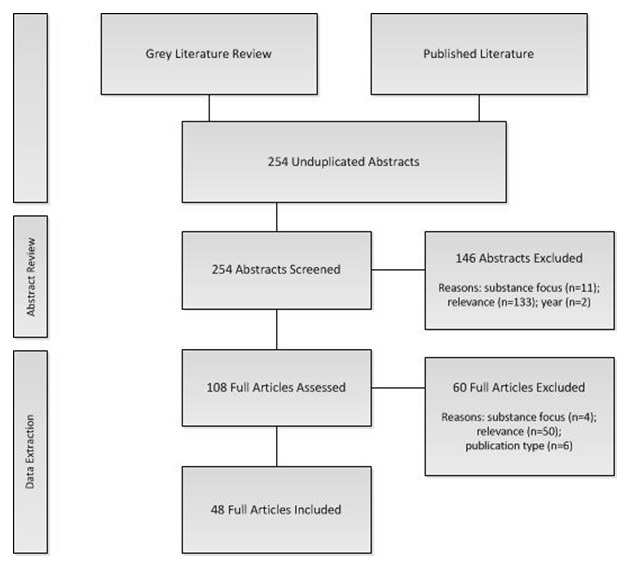 |
2.4. Data Extraction, Charting and Synthesis
Once relevant publications were selected for inclusion, we began extracting content relevant to each of the research questions and charting the information in the data charting tool. We then synthesized the data. To synthesize the data collected from the publications, we conducted a qualitative thematic analysis. This was done by identifying themes and subthemes within the content extracted by research question. Themes and subthemes identified were then documented in the data collection tool for further analysis and synthesis. We completed an additional analysis of the themes and subthemes identified across the three research questions to contextualize the implications of the results for health care practice. Finally, we identified gaps in the literature examined in order to highlight future research needs.
3. RESULTS
3.1. Effects of Non-Fatal Opioid-Involved Overdoses
Many of the studies included in this review focus on the health effects associated with NFOO. Findings confirm that non-fatal overdoses involving µ-receptor opioids (including natural, semi-synthetic and synthetic opioids) is an important cause of morbidity among persons who misuse and abuse prescription and/or illicitly-manufactured opioids (e.g., heroin). NFOO is of clinical consequence due to its association with a wide range of acute and chronic medical complications, including aspiration pneumonia,[2] pulmonary edema,[3] bronchopneumonia,[4] rhabdomyolysis,[5] muscle tissue breakdown, hypoxic brain injury, peripheral neuropathy,[6] renal failure, cognitive impairment, and traumatic injuries sustained during overdose (Darke & Hall, 2003; Ridgway & Pountney, 2007). One study found more than three-quarters of overdose victims reported at least one post-overdose morbidity symptom, including pneumonia, palsy, rhabdomyolysis, pulmonary edema, seizure, or cardiac arrhythmia (Warner-Smith, Darke, & Day, 2002). Studies have also documented depression among opioid overdose victims, but the lack of baseline assessments limit identification of significant associations between depression and overdose (Dassanyake et al., 2012). Corrigan & colleagues (2013) nevertheless suggest that diminished performance in several cognitive domains can increase depressive symptoms and negatively impact everyday functioning and the likelihood of poorer social and vocational outcomes.
Opioid-Induced Respiratory Depression
Non-fatal overdose is associated with a number of post-overdose complications. Opioid-induced respiratory depression (OIRD) is identified as a primary complication and the most dangerous source of adverse health outcomes associated with NFOO (King, Morris, & Schahmann, 2015). OIRD is caused by the activation of the µ-opioid receptor (MOR) expressed on the surface of neurons in brainstem respiratory centers that control breathing (Dahan et al., 2018). In most NFOO cases, OIRD appears in the form of acute respiratory compromise with the victim reverting to normal breathing activity, either on their own or with the use of an opioid antagonist like naloxone (Wermeling, 2015). Most NFOO victims recover from acute OIRD and present minor health consequences lasting minutes to hours (Sporer & Dorn, 2001; Buajordet et al., 2004; Olfson et al., 2018).
In some cases, such as when higher dose opioids are consumed or when opioids are taken concurrently with other central nervous system (CNS) depressants (e.g., benzodiazepine), acute respiratory compromise can progress into respiratory distress, cessation of breathing (i.e., apnea), cardiorespiratory collapse, respiratory arrest, and death (Solis et al., 2017; Dassanayake & Michie, 2012). Research indicates that enduring cognitive and physical consequences are associated with NFOO as OIRD progresses from irregular breathing to apnea to the protracted loss of oxygen (Quinn & Abbot, 2014). Hypoxia-related outcomes associated with opioid poisoning include kidney failure, heart complications, neurologic consequences, seizures, nerve damage, temporary motor paralysis, fluid buildup in the lungs, stroke, and pneumonia from inhaling vomitus (Marino et al., 2018; Betts, Ritter, & Kubal, 2012; O'Brien & Todd, 2009; Fitzgerald et al., 2000).
Brain Injury
The health consequences of OIRD associated with NFOO include a range of acute complications that develop during and/or immediately after an overdose event. Relatively scarce is literature detailing the more chronic health outcomes that develop among victims experiencing one or more NFOO. The majority of chronic literature concerns the development of hypoxic-related brain injuries associated with NFOO and subsequent complications (Ueno et al., 2018; Somerville et al., 2017; Betts, Ritter, & Kubal, 2012; Salazar & Dubow, 2012). The hippocampus, basal ganglia, and globous pallidus seem particularly susceptible to injury, with short-term memory loss and changes in cognitive and physical functioning indicated as symptomatic outcomes. Stroke has been observed in rare NFOO cases and secondary infections from injecting opioids (e.g., endocarditis) can lead to brain abscesses or meningitis, further extending the injury. Opioid-induced, hypoxic brain injury can also lead to the development of sever disability, including mental disorientation, ataxia, gait disturbances, incontinence, paraplegia, and catatonia, among a range of other presentations (Buttner et al., 2000). Slower reaction time, impaired memory (amnestic syndrome), reduced motor skills, and diminished physical functioning have all been documented with hypoxic brain injuries associated with NFOO (Barash, Somerville, & DeMaria, 2017; Ueno et al., 2018; Somerville et al., 2017; Betts, Ritter, & Kubal, 2012; Salazar & Dubow, 2012). More notable is one study indicating the increased risk for hypoxic brain damage among persons who experience multiple or recurring opioid overdose events due to repeated and prolonged hypoxia (Zamora et al., 2015).
Episodes of delayed post-hypoxic leukoencephalopathy--a rare type of neuron damage caused by a prolonged period of oxygen deficiency--are documented among overdose victims and indicated via MRI by slight deterioration of the periventricular regions of the brain. A recent Morbidity and Mortality Weekly Report (MMWR) documented a cluster of cases of sudden onset amnesia, a rare cause of memory loss often associated with toxic exposure (e.g., carbon monoxide poisoning), in Massachusetts between 2012 and 2016 (Barash, Somerville, & DeMaria, 2017). These cases were similarly confirmed by MRI and involved acute and complete ischemia (i.e., a restriction in blood supply to tissues that cause a shortage of oxygen) of both hippocampi. The cause of the acute amnesia among these patients is unknown to researchers, but the toxicological presence of opioids across cases suggests associations with substance use.
Differences in Effects of Overdoses between Single, Multiple, and Recurring Opioid Overdose
Differential health complications associated with single, multiple, and recurring opioid overdose were identified. Most notable is the identification of a strong correlation between prior and future NFOO events, as persons experiencing a single NFOO event were found more likely to experience a future overdose event in their lifetime; ergo, a person's overdose risk increases with every overdose they experience (Olfson et al., 2018; Darke et al., 2014). NFOO victims who experience multiple/recurring opioid overdose are shown to be at greater risk for long-term physical and cognitive consequences resulting from OIRD than persons experiencing a single overdose. (Darke et al., 2007). Repeatedly subjecting the brain to a deficiency of oxygen is shown to cause damage to the white matter of the brain (King, Morris, & Schahmann, 2015), and the risk for neurophysiological consequences increases with the both number of times a person experiences a hypoxic/anoxic event and the duration of each event (Beeskow et al., 2018).
3.2. Differing Health Outcomes between Illicitly-Manufactured Fentanyl, Heroin, and Prescription Opioids
Studies included in this review have identified the overdose risks associated with opioid analgesics via their interaction with the MOR and the depressive effect on the CNS. The synthetic opioid fentanyl stands apart from heroin and other semi-synthetic opioids in these findings by its high potency relative to heroin--fentanyl is 50x more potent by weight than heroin--and its rapid onset of action (5-10 seconds when injected), both of which are made possible because of fentanyl's high lipophilicity and its ability to rapidly cross the blood-brain barrier (Burns et al., 2016). The number of fatal IMF-involved overdoses has increased substantially since 2013, surpassing the number of deaths from heroin and prescription opioids in 2016 and 2017, with a 45.2% change in IMF-involved overdose deaths from 2016-2017 (19,413-28,466, respectively; Scholl et al., 2019). Few studies, however, have examined whether there are specific health complications associated with IMF-involved overdose compared to overdoses involving prescription opioids or heroin.
While the epidemiological literature on overdose-related health outcomes linked to IMF are negligible (Somerville et al., 2017), the medical research literature does identify several iatrogenic side effects (i.e., side effects resulting from medical treatment) associated with the clinical use of intravenous fentanyl in hospital settings, specifically skeletal muscle rigidity and chest wall rigidity (Burns et al., 2016). Chest wall rigidity, colloquially referred to as "wooden chest syndrome," is understood as the acute inability to ventilate or move the chest wall. Wooden chest syndrome presents obstacles for reversing respiratory depression associated with fentanyl toxicity, due to the hardening of the chest wall and the inability to ventilate victims, but the syndrome is shown to dissipate with naloxone administration. Rigid chest is not commonly associated with natural and semi-synthetic opioids and mainly observed in the synthetic lipid soluble compounds (e.g., fentanyl, acetyl fentanyl and sufentanil).[7] Because fentanyl is known to produce sudden respiratory consequences at toxic levels, the potential for rapid respiratory depression and arrest may illustrate potential risks for reversing overdoses involving IMF in street settings given reports of sudden unresponsiveness and rapid onset hypoxia associated with IMF (Somerville et al., 2017; MMWR).
Solis, Cameron-Burr, & Kiyatkin (2017) also studied differences between heroin, fentanyl, and fentanyl-adulterated heroin and each substance's influence on factors associated with respiratory depression leading to acute hypoxia in rats. They found that while all three heroin/heroin+fentanyl configurations led to decreased oxygen concentrations in rats, fentanyl caused a faster drop in oxygen levels than heroin, and heroin-containing fentanyl led to more sustained hypoxia and greater decreases in body temperature than both heroin and fentanyl alone. Two additional case studies also examined differences in health outcomes for NFOO involving fentanyl compared to heroin and other opioids, with both studies describing episodes of delayed post-hypoxic leukoencephalopathy[8] after a NFOO involving fentanyl (Beeskow et al., 2018; Betts, Ritter, & Kubal, 2012). In both case studies, patients experienced symptoms of post-hypoxic leukoencephalopathy about 3 weeks after a non-fatal overdose; for both patients, these symptoms included odd behavior, agitation, and decreased psychomotor or cognitive activity, as well as memory impairment in one of the patients (Beeskow et al., 2018; Betts et al., 2012). While neither case study described the full recovery of patients, one study did demonstrate patient improvement at 6-months (Betts et al., 2012).
3.3. Interaction between Pre-existing Conditions and Subsequent Overdose
As with the differences in outcomes between IMF and other opioid-related non-fatal overdoses, few studies have examined the interplay between already-existing physical or mental conditions and subsequent NFOO. Some mental health conditions, such as depression and suicidality, have been strongly associated with opioid use disorder and drug overdose. Comorbid mental health conditions are identified as risk factors for drug overdose but it is difficult to distinguish the degree to which they are influenced or exasperated by NFOOs in retrospective studies (Neale, 2000). Tobin & Latkin (2003), for example, found that individuals who had experienced an overdose within 12 months prior to being interviewed scored significantly higher on depression indexes compared to those that had not. In this case, depression may have acted as a catalyst for the overdose event, been exasperated by it, or both. Beyond depression, intentional suicide attempts are shown to be associated with unintentional overdose (Neale, 2000; Bradvick et al., 2007), with intentional suicide risk correlated with a higher number of drug overdoses (Bradvick et al., 2007). It is unclear, however, to what degree suicidality is exasperated by drug overdose.
A similar interplay is described with respect to prefrontal cortex functioning and opioid misuse and abuse. According to the cognitive deficits model, physical abnormalities in the structure of the prefrontal cortex can make it more difficult for individuals to control their impulses and drug use behavior. As individuals succumb to their impulses and use opioids and overdose, hypoxia can result in damage to the prefrontal cortex, worsening their addiction and increasing negative cognitive and mental health outcomes (Kosten & George, 2002).
Symptoms associated with the opioid overdose-induced hypoxic injuries described above may exasperate already-existing disabilities and conditions. Barash & colleagues (2017), for example, documented three cases of seizures associated with opioid overdose: one had previously experienced seizures, one experienced seizure during their acute overdose, and one developed a seizure disorder and post-overdose amnesia. While researchers did not document a history of worsening epilepsy, they were able to show an interaction between seizures and opioid overdose at each stage. Accordingly, declines in cognitive and physical functioning instigated by NFOO may also make it difficult for individuals to manage their existing chronic illnesses, such as diabetes and clinical depression (King, 2015).
Finally, it is possible that the interaction between disability and opioid overdose has not been well examined simply because some of the health outcomes are rare and difficult for physicians and researchers to identify through standard medical practice. For example, in one case study, physicians described the challenge of diagnosing a young woman with a history of seizures and cri du chat, a developmental disability. The medical team focused heavily on characterizing the woman's altered level of consciousness and ran extensive tests before taking a full history with her caregiver and discovering her recent codeine use (Kumar, 2007).
3.4. Health Service Utilization
Research containing information about utilization of health services after a non-fatal overdose is also scarce. One study meeting our inclusion/exclusion criteria conducted in Australia approximately 20 years ago found that, within their sample of current heroin users, about one-half had experienced a non-fatal overdose in their lifetime; following these non-fatal overdoses, 54% of respondents were administered naloxone, 59% reported that an ambulance attended the overdose, and 45% of respondents were hospitalized because of the overdose (McGregor et al., 1998).
4. DISCUSSION
4.1. Discussion of Results
Our findings suggest NFOO is associated with the current opioid epidemic and represents an increasing proportion of morbidity in the United States. The current and past research on non-fatal opioid overdose included in this review describe a number of physiological complications stemming from an NFOO event. It is notable that the vast majority of clinical presentations associated with non-fatal opioid overdose identified in the literature are acute complications of the opioid overdose itself, with findings indicating that most consequences appear during or immediately after the overdose event, or in days to weeks afterward.
Evidence indicates that people who experience a single opioid overdose are more likely to have subsequent overdoses (Dassanyake et al., 2012), and multiple and repeated opioid overdoses are correlated with decreasing cognitive performance (Darke et al., 2007), increases in depression symptoms (Tobin & Latkin, 2003), and suicidal ideation (Bradvick et al., 2007). The findings related to IMF, specifically its effect on increasing the cumulative number of overdoses individuals will experience in a lifetime, raise the possibility that people who repeatedly experience a NFOO may be undergoing recurrent hypoxic events and experiencing "minor" brain injuries whose negative effects are compounded by repetition. Beyond studies identifying opioid overdose-induced brain injuries via MRI, the recurring onset of hypoxia characteristic of "serial overdosers" may generate no immediate symptoms, while others may be so subtle that it becomes difficult for people to recognize, or symptoms may not appear for weeks, months, or years after. Consequently, these findings illustrate a plausible parallel between concussions as a traumatic brain injury on the one hand and opioid overdose-related hypoxic events as a "milder" brain injury on the other: both events affect the brain and both are known to be associated with physical, mental, and cognitive complications.
Sharp increases in IMF supply and consumption since 2013 have been linked to the sharp rise in fentanyl-involved overdoses, with the number of fentanyl overdoses surpassing the number of prescription opioid and heroin overdoses for 2016 and 2017 (DEA, 2018). Fentanyl is a highly potent synthetic opioid that is roughly 50 times more potent by weight than heroin (Wade et al., 2015). Increased fentanyl consumption has given rise to the panoply of acute health complications described above, but numerous reports also indicate that IMF is engendering unique and heretofore uncommon overdose presentations not seen with natural and semi-synthetic opioids (Somerville et al., 2017). A recent study in rats detected rapidly strong but transient decreases in extracellular oxygen levels when human-relevant doses (40 mcg/kg) of fentanyl was administered, with brain hypoxic effects becoming significantly enhanced when the fentanyl dose increased by 10% (Solis, Cameron-Burr, & Kiyatkin, 2017). As with similar pharmacokinetic studies on fentanyl toxicity, these findings indicate that illicit fentanyl consumption, even in nominal amounts, poses heightened risks for acute brain complications, including enhanced brain hypoxia and induced brain hypothermia. Additional symptoms associated with fentanyl toxicity have also been documented, including sudden respiratory distress, rigor mortis, rigid chest, comatose, immediate cyanosis, among others (Somerville et al., 2017; Burns et al., 2016).
The surge in IMF consumption has not only contributed to concurrent increases in IMF-involved overdoses (as noted by a recent CDC report indicating a 45% increase in fentanyl-involved deaths from 2016-2017) but the cumulative number of overdoses experienced by individuals are also increasing (Scholl et al., 2019). An increase in the number of lifetime overdoses experienced by people using IMF is especially disconcerting given the damage that recurring hypoxic[9] and anoxic events[10] can have on the brain. At present, there are effective, evidence-based interventions that can reduce the negative health effects associated with NFOO, such as training and equipping opioid consumers, emergency responders, bystanders, family members, correctional officers, and drug treatment and health care providers in the use of naloxone (Beletsky, 2012). Naloxone, a drug that counters the effects of opioids, is effective in temporarily reversing the depressive effects of an opioid overdose (U.S. Surgeon General, 2018) and reducing opioid overdose death rates (McClellan et al., 2018). The rapid onset of respiratory depression and arrest associated with acute fentanyl toxicity makes the risk of hypoxic and anoxic events more likely, suggesting that naloxone may need to be administered more quickly than overdoses involving other opioids, in addition to potentially needing more than one dose per overdose reversal.
4.2. Gaps in the Literature
Our review of the literature identified several knowledge gaps. Specifically, while acute complications that transpire during or shortly after an opioid overdose are detailed and robust, the paucity of findings on the chronic complications resulting from recurring non-fatal opioid overdose events are alarming given the evidence we do have on hypoxic brain injuries stemming from opioid-induced toxicity. In addition, given what we know about fentanyl's potency and the surge in IMF-involved overdoses, the limited number of publications examining differences in health outcomes between heroin, IMF, and prescription opioids was surprising. The current literature suggests that non-fatal overdoses involving synthetic opioids (e.g., fentanyl) can lead to more serious or longer-term health outcomes, but the specific discrepancies between health outcomes involving these types of drugs has yet to be examined more in-depth. A gap also appears to exist on whether existing physical and mental health outcomes among persons who misuse and abuse opioids become exacerbated when said consumers experience (>1) non-fatal opioid overdose. This lack of information is compounded by the retrospective design of many studies, which makes rigorously evaluating the functional status of individuals prior to overdose exceptionally challenging. The current dearth of longitudinal studies examining these topics restricts our ability to understand the myriad complications of opioid overdose on victims' physical, mental, and cognitive functioning, and specifically the downstream effects of a fentanyl overdose crisis that, at present, involves thousands of people.
4.3. Importance of Information
As the number of non-fatal opioid-involved opioids continues to grow, it is paramount that health care and treatment providers as well as policymakers have information on the health and functional outcomes of individuals who have experienced singular, multiple, or reoccurring overdose events. This is especially true of IMF-involved overdoses. Without this information, providers and policymakers will be hindered in their ability to provide appropriate and high-quality health care and supportive services for individuals who have experienced one or multiple overdose events. However, our review demonstrated that there is a paucity of peer-reviewed and grey publications leaving gaps in our understanding of the health and functional outcomes of non-fatal opioid overdoses. Further research that includes an additional review of the literature with expanded search strategies, the collection of primary data through interviews, and longitudinal studies utilizing secondary data, may address these knowledge gaps.
4.4. Study Limitations
Although our review yielded many studies presenting acute health consequences of an overdose, few focused on the chronic health and functional outcomes of non-fatal overdoses. Likewise, there appears to be little information in the peer-reviewed or grey literature regarding the differences in short-term and long-term health and functional outcomes between IMF and semi-synthetic opioids. Similarly, our review found very few publications that examined the interplay between pre-existing conditions and non-fatal overdose.
It is unclear if the limited number of publications examining these topics is truly a reflection of the amount of research currently being conducted in this area or a reflection of the limitation in our search criteria. Although our team included a senior librarian and multiple (three) separate searches of the peer-reviewed literature and a thorough review of the grey literature, our review may have been limited by our search terms. For example, several publications were not identified in the first attempt in the peer-reviewed literature because they did not include the terms (or variations) of "opioid," "overdose," or "non-fatal" in their titles or keywords. Future reviews should build upon our search strategy and expand the list of search terms to include specific names of medications and health outcomes known to be associated with non-fatal overdose.
5. CONCLUSION
The purpose of this scoping review was to better understand health and functional outcomes associated with NFOO. After reviewing 48 peer-reviewed and grey publications identified and selected for review, our analysis concluded that numerous clinical complications are associated with non-fatal opioid overdose. Among these, most focus on acute outcomes stemming from respiratory depression and related hypoxia.
Epidemiological research suggests that the increased saturation of IMF in existing heroin markets may be serving as an overdose risk modifier due to fentanyl's pharmacokinetics as a synthetic opioid and its higher potency compared to heroin (Dragoovic, Tamburro & Hadidi, 2016; Marinetti & Ehlers, 2014). IMF is contributing to a rise in severe and uncommon opioid overdose presentations and increasing the cumulative number of overdose events experienced by individual fentanyl consumers. The combination of atypical overdose complications on the one hand and cumulative increases in the number of overdose events on the other raises concerns that recurring hypoxic brain injuries experienced by overdose victims may be contributing to chronic brain damage and associated physical, mental, and cognitive disability among this population.
Taken together, these findings raise the possibility that people who repeatedly experience an opioid overdose may be undergoing recurring hypoxic events and experiencing "minor" brain injuries whose effects may be compounded by repetition. This hypothesis is based on aforementioned findings whose evidence points to a plausible parallel between recurring overdose events and repeat concussions, with opioid overdose-induced hypoxia understood as a "milder" form of brain injury: concussions and hypoxic overdose events both affect the brain, and both are associated with diseases that affect the function or structure of the brain.
The current dearth of longitudinal studies examining the more chronic complications resulting from multiple or recurring non-fatal opioid overdose events restricts our ability to understand the longer-term effects of opioid overdose on victims' physical, mental, and cognitive functioning, which is compounded by the unknown downstream effects of a fentanyl overdose crisis that, at present, involves hundreds of thousands of people. Although the studies examined as part of this scoping review do offer insight into the myriad consequences of non-fatal opioid overdose on victims' physical and mental health, large gaps in our knowledge of NFOO still exist. Potentially fruitful avenues for future research include an additional review of the NFOO literature with expanded search strategies, the collection of primary data through interviews with key subject matter experts and NFOO victims, and longitudinal cohort studies that follow overdose victims for decades. A longer-term recommendation for clinical research may even include expanding current brain injury studies, such as the Chronic Traumatic Encephalopathy Program at Boston University, to include brains of fatal overdose victims who are known to have experienced--and/or been negatively affected by--repeated non-fatal hypoxic overdose events prior to death.
REFERENCES
Arksey, H., & O'Malley, L. (2005). Scoping studies: towards a methodological framework. International Journal of Social Research Methodology, 8, 19-32.
Baca, C.T., & Grant, K.J. (2005). Take-home naloxone to reduce heroin death. Addiction, 100(12), 1823-1831.
Bachi, K., Sierra, S., Volkow, N.D., Goldstein, R.Z., & Alia-Klein, N. (2017). Is biological aging accelerated in drug addiction? Curr Opin Behav Sci, 13, 34-39.
Barash, J.A., Somerville, N., & DeMaria, A. Jr. (2017). Cluster of an unusual amnestic syndrome--Massachusetts, 2012-2016. MMWR, 66(3), 76. Retrieved from https://www.cdc.gov/mmwr/volumes/66/wr/mm6603a2.htm.
Beeskow, A.B., Oberstadt, M., Saur, D., Hoffman, K.T., & Lobsien, D. (2018). Delayed post-hypoxic leukoencephalopathy (DPHL)--An uncommon variant of hypoxic brain damage in adults. Frontiers in Neurology, 9.
Beletsky, L., Rich, J.D., & Walley, A.Y. (2012). Prevention of fatal opioid overdose. JAMA, 308(18), 1863-1864.
Betts, A.M., Ritter, J.L., & Kubal, W.S. (2012). Reversible delayed posthypoxic leukoencephalopathy after drug overdose: MRI findings in a collection of patients. Emergency Radiology, 19(2), 165-173.
Bond, G.R., Ho, M., & Woodward, R.W. (2012). Trends in hepatic injury associated with unintentional overdose of paracetamol (acetaminophen) in products with and without opioid: An analysis using the National Poison Data System of the American Association of Poison Control Centers, 2000-7. Drug Safety, 35(2), 149-157.
Bradvik, L., Frank, A., Hulenvik, P., Medvedeo, A., & Berglund, M. (2007). Heroin addicts reporting previous heroin overdoses also report suicide attempts. Suicide Life Threat Behav, 37(4), 475-81.
Buajordet, I., Naess, A.C., Jacobsen, D., & Brørs, O. (2004). Adverse events after naloxone treatment of episodes of suspected acute opioid overdose. Eur J Emerg Med, 11(1), 19-23.
Burns G., DeRienz R.T., Baker D.D., Casavant M., & Spiller H.A. (2016). Could chest wall rigidity be a factor in rapid death from illicit fentanyl abuse? Clin Toxicol (Phila), 54(5), 420-423.
Buttner, A., Mall, G., Penning, R., & Weis, S. (2000). The neuropathy of heroin abuse. Forensic Science International, 113, 435-442.
CDC. (2018). Vital signs: Trends in emergency department visits for suspected opioid overdoses--United States, July 2016-September 2017. MMWR, 67(9), 279-285.
CDC. (2017). Annual surveillance report of drug-related risks and outcomes--United States, 2017. Atlanta, GA: U.S. Department of Health and Human Services, CDC. Retrieved from: https://www.cdc.gov/drugoverdose/pdf/pubs/2017-cdc-drug-surveillance-report.pdf.
CDC. (2016). Wide-ranging online data for epidemiologic research (WONDER). Atlanta, GA: U.S. Department of Health and Human Services, CDC, National Center for Health Statistics. Retrieved from: https://wonder.cdc.gov.
Coffin, P.O., Tracy, M., Bucciarelli, A., Ompad, D., Vlahov, D., & Galea, S. (2007). Identifying injection drug users at risk of nonfatal overdose. Acad Emerg Med, 14(7), 616-623.
Colquhoun, H.L., Level, D., O'Brien, K.K., Straus, S., Tricco, A.C., Perrier, L., Kastner, M., & Moher, D. (2014), Scoping reviews: Time for clarity in definition, methods, and reporting. Journal of Clinical Epidemiology, 67(12), 1291-1294.
Conner, K.R., Britton, P.C., Sworts, L.M., & Joiner, T.E. Jr. (2007). Suicide attempts among individuals with opiate dependence: The critical role of belonging. Addict Behav, 32(7), 1395-1404.
Darke, S., Marel, C., Mills, K.L., Ross, J., Slade, T., Burns, L. & Tesson, M. (2014). Patterns and correlates of non-fatal heroin overdose at 11-year follow-up: Findings from the Australian Treatment Outcome Study. Drug Alcohol Depend, 144, 148-152.
Darke, S., Sims, J., McDonald, S., & Wickes, W. (2000). Cognitive impairment among methadone maintenance patients. Addiction, 95(5), 687-695.
Darke, S., Williamson, A., Ross, J., Mills, K.L., Harvard, A., & Teesson, M. (2007). Patterns of nonfatal heroin overdose over a 3-year period: Findings from the Australian treatment outcome study. J Urban Health, 84(2), 283-291.
Darke, S., Williamson, A., Ross, J., & Teesson, M. (2005). Non-fatal heroin overdose, treatment exposure and client characteristics: Findings from the Australian treatment outcome study (ATOS). Drug Alcohol Rev, 24(5), 425-432.
Dassanayake, T.L., Michie, P.T., Jones, A., Carter, G., Mallard, T., & Whyte, I. (2012). Cognitive impairment in patients clinically recovered from central nervous system depressant drug overdose. Journal of Clinical Psychopharmacology, 32(4), 503-510.
Dragovic, L., Tamburro, L., & Al-Hadidi, J. (2016). Resurgence of fentanyl as a drug of abuse. Journal of Forensic Science & Medicine, 2(2), 111-114.
Duru, U.B., Pawar, G., Barash, J.A., Miller, L.E., Thiruselvam, I.K., & Haut, M.W. (2018). An unusual amnestic syndrome associated with combined fentanyl and cocaine use. Ann Inter Med, 168(10), 747-748
Elmer, J., Flickinger, K.L., Anderson, M.W., Koller, A.C., Sundermann, M.L., Dezfulian, C., Okonkwo, D.O., Shutter, L.A., Salcido, D.D., Callaway, C.W. & Menegazzi, J.J. (2018). Effect of neuromonitor-guided titrated care on brain tissue hypoxia after opioid overdose cardiac arrest. Resuscitation, 129, 121-126.
Ersche, K.D., & Sahakian, B.J. (2007). The neuropsychology of amphetamine and opiate dependence: Implications for treatment. Neuropsychology Review, 17(3), pp. 317-336.
Fitzgerald, J., Hamilton, M., & Dietze, P. (2000). Walking overdoses: A re-appraisal of non-fatal illicit drug overdose. Addiction Research, 8(4), 327-355.
Hantson, P., Vanbinst, R., & Wallemacq, P. (2003). Accidental methadone overdose in an opiate-naive elderly patient. Intensive Care Medicine, 29(11), 2105.
Havens, J.R., Oscer, C.B., Knudsen, H.K., Lofwall, M., Stoops, W.W., Walsh, S.L., Leukefeld, C.G., & Kral, A. (2011). Individual and network factors associated with non-fatal overdose among rural Appalachian drug users. Drug & Alcohol Dependence, 115, 107-112.
Heale, P., Dietze, P., & Fry, C. (2003). Intentional overdose among heroin overdose survivors. J Urban Health, 80(2), 230-237.
Hedegaard, H., Warner, M., & Miniño, A.M. (2017). Drug overdose deaths in the United States, 1999-2016. NCHS data brief, no 294. Hyattsville, MD: U.S. Department of Health and Human Services, CDC, National Center for Health Statistics. Retrieved from: https://www.cdc.gov/nchs/data/databriefs/db294.pdf.
King, F., Morris, N.A., & Schahmann, J.D. (2015). Delayed post-hypoxic leukoencephalopathy: Improvement with antioxidant therapy. Case Reports in Neurology, 7, 242-246.
Kumar, A. (2007). Accidental iatrogenic opioid overdose: A difficult diagnosis. British Journal of Anaesthetic & Recovery Nursing, 8(1), 5-6.
Kumar, N., Bhalla, M.C., Frey, J.A., & Southern, A. (2015). Intraparenchymal hemorrhage after heroin use. American Journal of Emergency Medicine, 33(8), 1109.e3-1109.e4.
Levac, D., Colquhoun, H., & O'Brien, K.K. (2010). Scoping studies: Advancing the methodology. Implementation Science, 5(69), 1-9.
Marinetti, L.J., & Ehlers, B.J. (2014). A series of forensic toxicology and drug seizure cases involving illicit fentanyl alone and in combination with heroin, cocaine or heroin and cocaine. Journal of Analytical Toxicology, 38(8), 592-598.
McClellan, C., Lambdin, B.H., Ali, M.M., Mutter, R., Davis, C.S., Wheeler, E., Pemberton, M., & Kral, A.H. (2018). Opioid-overdose laws association with opioid use and overdose mortality. Addictive Behaviors, 86, 90-95.
McGregor, C., Darke, S., Ali, R., & Christie, P. (1998). Experience of non-fatal overdose among heroin users in Adelaide, Australia: Circumstances and risk perceptions. Addiction, 93(5), 701-711.
Mulpur, A.K., Mirsadraee, S., Hassan, T.B., McKeague, H., & Kaul, P. (2004). Refractory ventricular fibrillation in accidental hypothermia: Salvage with cardiopulmonary bypass. Perfusion, 19(5), 311-314.
Neale, J. (2000). Suicidal intent in non-fatal illicit drug overdose. Addiction, 95(1), 85-93.
O'Donnell, J.K., Gladden, R.M., & Seth, P. (2017). Trends in deaths involving heroin and synthetic opioids excluding methadone, and law enforcement drug product reports, by census region--United States, 2006-2015. MMWR, 66(34), 897-903. Retrieved from: https://www.cdc.gov/mmwr/volumes/66/wr/mm6634a2.htm.
Olfson, M., Crystal, S., Wall, M.M., Wang, S., Liu, S.M., & Blanco, C. (2018). Causes of death after nonfatal opioid overdose. JAMA Psychiatry, 75(8), 820-827.
National Institutes of Health (NIH). (n.d.). Opioid overdose crisis, Retrieved from: https://www.drugabuse.gov/drugs-abuse/opioids/opioid-overdose-crisis.
Paulozzi, L.J., Zhang, K., Jones, C.M., & Mack, K.A. (2014). Risk of adverse health outcomes with increasing duration and regularity of opioid therapy. JABFM, 27(3), 329-338.
Pfab, R., Eyer, F., Jetzinger, E., & Zilker, T. (2006). Cause and motivation in cases of non-fatal drug overdoses in opiate addicts. Clin Toxicol (Phila), 44(3), 255-259.
Quinn, D.K., & Abbott, C.C. (2014). Catatonia after cerebral hypoxia: Do the usual treatments apply? Psychosomatics, 55(6), 525-535.
Reece, A., & Hulse, G. (2013). Lifetime opiate exposure as an independent and interactive cardiovascular risk factor in males: A cross-sectional clinical study. Vasc Health Risk Manag, 9, 551-561.
Ridgeway, Z.A., & Pountney, A.J. (2007). Acute respiratory distress syndrome induced by oral methadone managed with non-invasive ventilation. Emerg Med J, 24(9), 681.
Salazar, R., & Dubow, J. (2012). Delayed pothypoxic leukoencephalopathy following a morphine overdose. Journal of Clinical Neuroscience, 19, pp. 1060-1062.
Scholl, L., Seth, P., Kariisa, M., Wilson, N., & Baldwin, G. (2019). Drug and opioid-involved overdose deaths--United States, 2013-2017. MMWR, 67(5152), 1419-1427. Retrieved from: https://www.cdc.gov/mmwr/volumes/67/wr/mm675152e1.htm.
Solis, E. Jr., Cameron-Burr, K.T., & Kiyatkin, E.A. (2017). Heroin contaminated with fentanyl dramatically enhances brain hypoxia and induces brain hypothermia. eNeuro, 4(5), pp. 1-10.
Somerville N.J., O'Donnell, J., Gladden, R.M., Zibbell, J.E., Green, T.C., Younkin, M., Ruiz, S., Babakhanlou-Chase, H., Chan, M., Callis, B.P., Kuramoto-Crawford, J. Nields, H.M., & Walley, A.Y. (2017). Characteristics of fentanyl overdose--Massachusetts, 2014-2016. MMWR, 66, 382-386
Sporer, K.A., & Dorn, E. (2001). Heroin-related noncardiogenic pulmonary edema--A case series. Chest, 120(5), 1628-1632.
Stewart, D., Gossop, M., & Marsden, J. (2002). Reductions in non-fatal overdose after drug misuse treatment: Results from the National Treatment Outcome Research Study (NTORS). Journal of Substance Abuse Treatment, 22(1), 1-9.
Tobin, K.E., & Latkin, C.A. (2003). The relationship between depressive symptoms and nonfatal overdose among a sample of drug users in Baltimore, Maryland. J Urban Health, 80(2), 220-229.
U.S. Department of Justice, Drug Enforcement Administration (DEA). (2018). 2018 National Drug Threat Assessment. Retrieved from: https://www.dea.gov/sites/default/files/2018-11/DIR-032-18%202018%20NDTA%20final%20low%20resolution.pdf.
U.S. Surgeon General. (2018). Surgeon General's Advisory on Naloxone and Opioid Overdose. Retrieved from: https://www.surgeongeneral.gov/priorities/opioid-overdose-prevention/naloxone-advisory.html.
Warner-Smith, M., Darke, S., & Day, C. (2002). Morbidity associated with nonfatal heroin overdose. Addiction, 97(8), 963-967.
Warner-Smith, M., Darke, S., Lynskey, M., & Hall, W. (2001). Heroin overdose: Causes and consequences. Addiction, 96(8), 1113-1125.
Wermeling, D.P. (2015). Review of naloxone safety for opioid overdose: Practical considerations for new technology and expanded public access. Ther Adv Drug Saf, 6(1), 20-31.
Wines, J.D. Jr., Saitz, R., Horton, N.J., Lloyd-Travaglini, C., & Samet, J.H. (2007). Overdose after detoxification: A prospective study. Drug Alcohol Depend, 89(2-3), 161-169.
Yen, Y.F., Chou, P., Lin, Y.S., & Deng, C.Y. (2015). Factors associated with health-related quality of life among injection drug users at methadone clinics in Taipei, Taiwan. Journal of the Chinese Medical Association, 78(5), 292-298.
APPENDIX A. EXAMPLE SEARCH STRATEGY--PubMed
PubMed--Search 1
#3 Add Search ((#1 OR #2) NOT ("Comment"[Publication Type] OR "Letter"[Publication Type] OR "Editorial"[Publication Type])) Filters: Publication date from 1998/01/01; English 238
#2 Add Search (opioid*[Title] OR fentanyl[Title] OR "illicitly-manufactured fentanyl"[Text Word] OR "illicitly-made fentanyl"[Text Word] OR acetylfentanyl[Title] OR furanylfentanyl[Title] OR carfentanil[Title] OR heroin[Title] OR oxycodone[Title] OR oxymorphone[Title] OR Opana[Title] OR hydromorphone[Title] OR Dilaudid[Title] OR OxyContin[Title] OR hydrocodone[Title] OR Vicodin[Title] OR novel psychoactive substance*[Text Word] OR methadone[Title] OR "Analgesics, Opioid"[Mesh] OR "Analgesics, Opioid"[Pharmacological Action] OR "Opioid-Related Disorders"[Mesh] OR "Fentanyl"[Mesh] OR "carfentanil"[Supplementary Concept] OR "N-(1-phenethylpiperidin-4-yl)-N-phenylacetamide"[Supplementary Concept] OR "Heroin"[Mesh] OR "Heroin Dependence"[Mesh] OR "Oxycodone"[Mesh] OR "Oxymorphone"[Mesh] OR "Hydromorphone"[Mesh] OR "Methadone"[Mesh]) AND (non fatal overdos*[Text Word] OR nonfatal overdos*[Text Word] OR unintentional overdos*[Text Word] OR accidental overdos*[Text Word]) Filters: Publication date from 1998/01/01; English 202
#1 Add Search (opioid*[Title] OR fentanyl[Title] OR "illicitly-manufactured fentanyl"[Text Word] OR "illicitly-made fentanyl"[Text Word] OR acetylfentanyl[Title] OR furanylfentanyl[Title] OR carfentanil[Title] OR heroin[Title] OR oxycodone[Title] OR oxymorphone[Title] OR Opana[Title] OR hydromorphone[Title] OR Dilaudid[Title] OR OxyContin[Title] OR hydrocodone[Title] OR Vicodin[Title] OR novel psychoactive substance*[Text Word] OR methadone[Title] OR "Analgesics, Opioid"[Mesh] OR "Analgesics, Opioid"[Pharmacological Action] OR "Opioid-Related Disorders"[Mesh] OR "Fentanyl"[Mesh] OR "carfentanil"[Supplementary Concept] OR "N-(1-phenethylpiperidin-4-yl)-N-phenylacetamide"[Supplementary Concept] OR "Heroin"[Mesh] OR "Heroin Dependence"[Mesh] OR "Oxycodone"[Mesh] OR "Oxymorphone"[Mesh] OR "Hydromorphone"[Mesh] OR "Methadone"[Mesh]) AND (overdos*[Title] OR "Drug Overdose"[Mesh]) AND ("non-fatal"[Title] OR nonfatal[Title] OR unintentional*[Title] OR accidental*[Title] OR accident[Title]) Filters: Publication date from 1998/01/01; English 122
PubMed--Search 2
#1 Add Search ((opioid*[Title] OR fentanyl[Title] OR "illicitly-manufactured fentanyl"[Text Word] OR "illicitly-made fentanyl"[Text Word] OR acetylfentanyl[Title] OR furanylfentanyl[Title] OR carfentanil[Title] OR heroin[Title] OR oxycodone[Title] OR oxymorphone[Title] OR Opana[Title] OR hydromorphone[Title] OR Dilaudid[Title] OR OxyContin[Title] OR hydrocodone[Title] OR Vicodin[Title] OR novel psychoactive substance*[Text Word] OR methadone[Title] OR "Analgesics, Opioid"[Mesh] OR "Analgesics, Opioid"[Pharmacological Action] OR "Opioid-Related Disorders"[Mesh] OR "Fentanyl"[Mesh] OR "carfentanil"[Supplementary Concept] OR "N-(1-phenethylpiperidin-4-yl)-N-phenylacetamide"[Supplementary Concept] OR "Heroin"[Mesh] OR "Heroin Dependence"[Mesh] OR "Oxycodone"[Mesh] OR "Oxymorphone"[Mesh] OR "Hydromorphone"[Mesh] OR "Methadone"[Mesh]) AND (overdos*[ti] OR "Drug Overdose"[Mesh]) AND ("Hypoxia"[Mesh] OR "Hypoxia, Brain"[Mesh] OR "hypoxic hypoxia"[tw] OR "arterial hypoxia" OR "hypoxia"[tw]) Filters: Publication date from 1998/01/01; English 16
PubMed--Search 3
#2 Add Search (("Longitudinal Studies"[Mesh] AND ("Drug Overdose/therapy"[Majr] OR "Opioid-Related Disorders/therapy"[Majr]) NOT #1)) Filters: Publication date from 1998/01/01; English 120
#1 Add Search ((opioid*[Title] OR fentanyl[Title] OR "illicitly-manufactured fentanyl"[Text Word] OR "illicitly-made fentanyl"[Text Word] OR acetylfentanyl[Title] OR furanylfentanyl[Title] OR carfentanil[Title] OR heroin[Title] OR oxycodone[Title] OR oxymorphone[Title] OR Opana[Title] OR hydromorphone[Title] OR Dilaudid[Title] OR OxyContin[Title] OR hydrocodone[Title] OR Vicodin[Title] OR novel psychoactive substance*[Text Word] OR methadone[Title] OR "Analgesics, Opioid"[Mesh] OR "Analgesics, Opioid"[Pharmacological Action] OR "Opioid-Related Disorders"[Mesh] OR "Fentanyl"[Mesh] OR "carfentanil"[Supplementary Concept] OR "N-(1-phenethylpiperidin-4-yl)-N-phenylacetamide"[Supplementary Concept] OR "Heroin"[Mesh] OR "Heroin Dependence"[Mesh] OR "Oxycodone"[Mesh] OR "Oxymorphone"[Mesh] OR "Hydromorphone"[Mesh] OR "Methadone"[Mesh]) AND (overdos*[ti] OR "Drug Overdose"[Mesh]) AND ("Longitudinal Studies"[Mesh] OR "Follow-Up Studies"[Mesh] OR longitudinal[ti] OR follow-up[ti])) Filters: Publication date from 1998/01/01; English 70
NOTES
-
IMFs include both fentanyl hydrochloride and fentanyl analogs (e.g., acetylfentanyl, furanylfentanyl, and carfentanil, etc.).
-
Swelling or an infection of the lungs or large airways.
-
Lung congestion.
-
Inflammation of the lungs.
-
The breakdown of damaged skeletal muscle.
-
Damage to the peripheral nerves, such as the hands or feet.
-
See Fermaglich (1996) [https://jamanetwork.com/journals/jama/article-abstract/395849] for a description of the Parkinsonian effects associated with the consumption of illicitly-manufactured meperidine (MPTP [1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine], also a synthetic opioid. Several substance users were rendered immobile and speechless following use of the drug and were colloquial refer to in the press as "frozen addicts." Clinical observations persuade doctors they are suffering from a pernicious form of Parkinson's disease. Confirmation arrives when they are treated with levodopa but have side effects that limit therapy, returning them to their debilitating frozen states.
-
A neurological relapse after initial recovery from an acute event resulting in the loss of oxygen to the brain.
-
Partial supply of oxygen to the brain, but at low levels that are inadequate to maintain normal brain function.
-
Complete interruption of the supply of oxygen to the brain.
