Through the HHS Office of the Assistant Secretary for Planning and Evaluation (ASPE), the Personalized Health Care Initiative sponsored a workshop in Washington, D.C., on July 7, 2008 entitled “Understanding the Needs of Consumers in the Use of Genome-based Health Information Services. This workshop was aimed at enabling various stakeholders to share differing perspectives and discuss alternatives for helping consumers make informed use of genome-based health information, both now and in the future.
Disclaimer: The findings and conclusions of these presentations are those of the authors and do not necessarily represent the views of ASPE or HHS.
"Agenda
12:30 – 12:45 pm
Welcome, Meeting Objectives and Overview
Richard Campanelli, JD
Counselor to the Secretary for Science and Public Health,
US Department of Health and Human Services
12:45 – 1:05 pm
Medical decision-making empowered by genomic information
Eric Topol, MD
Director, Scripps Translational Science Institute
Dean, Scripps School of Medicine
- How can validated genomic information empower consumers in medical decision-making?
- Compare and contrast what is currently available in the marketplace.
- What is your vision for the future genomic services?
1:05 – 1:30 pm
Consumer interest in health and genomic information
Steve Bodhaine, MBA
Group President, Research/Product Development, Yankelovich
1:30 – 2:30 pm
Panel 1: What is the consumer interest in genome-based health information?
1:30 – 2:10 pm
Panel Discussion
2:10 – 2:30 pm
Q&A
Linda Avey
Co-founder, 23andMe, Inc.
Esther Dyson
EDventure Holdings
Rebecca Fisher, MLIS
Patient Advocate
Matthew Holt
Co-founder Health 2.0 Conference
- What drives the consumer’s interest in health information for predicting health outcomes?
- What are key factors for motivating interest in genetic testing?
- For what information are consumers ready?
2:30 – 2:45 pm
Break
Panel 2: Is the testing process reliable, and is the information’s privacy maintained?
2:45 – 3:25 pm
Panel Discussion
3:25 – 4:00 pm
Q&A
Deven McGraw, JD, MPH
Director, Health Privacy Project, Center for Democracy and Technology
Ryan Phelan
Founder and CEO, DNA Direct
Jeffrey Gulcher, MD, PhD
Chief Scientific Officer, deCODE genetics
Reed Tuckson, MD, FACP
Executive Vice President and Chief of Medical Affairs, UnitedHealth Group
- What are potential ways to inform consumers that quality control standards for testing are being met?
- How are risk assessments determined?
- Will (should) the genomic data be utilized for public health benefit or other population-based research in the future?
- Are there cautions that the consumer should consider when sharing their genomic information with others, such as family members, members of social network, or clinicians?
- What protections are currently or should be employed to protect the security and privacy of consumer genomic information?
4:00 – 5:15 pm
Panel 3: what is currently useful to consumers, and what can they expect in the future?
4:00 – 4:50 pm
Panel Discussion
4:50 – 5:15 pm
Q&A
Mari Baker
President and CEO, Navigenics
Katherine Johansen, PhD
Senior Scientist, American Medical Association Program in Genetics and Molecular Medicine
Nancy Johnson
Senior Public Policy Advisor, Baker Donelson
Ronni Sandroff, MA
Director/Editor, Health and Family Information, Consumer Reports
Angela Trepanier MS, CGC
President, National Society of Genetic Counselors
Assistant Professor, Clinical Center for Molecular Medicine and Genetics, Wayne State University
- How can genomic information and explanatory resources deepen health literacy and support consumer empowerment for prevention?
- What checklist of resources or information should consumers have to assign value to genome-based health information services?
- How valuable is the information currently available relative to other health information, such as family history, blood pressure, BMI etc.?
- What can/should consumers expect in the future?
5:15 – 5:30 pm
Looking to the future of consumer services (moderated discussion)
Michael Cowan, MD
Chief Medical Officer, BearingPoint
Transcript
Horizon Ballroom
Ronald Reagan Building and International Trade
1300 Pennsylvania Ave. NW
Washington D.C. 20004
Monday, July 7, 2008
LIST OF PARTICIPANTS
RICHARD CAMPANELLI, JD
Counselor to the Secretary for Science and Public
Health, U.S. Dept. of Health and Human Services
ERIC TOPOL, M.D.
Director, Scripps Translational Science Institute
Dean, Scripps School of Medicine
STEVE BODHAINE, MBA
Group President, Research/Product Development
Yankelovich
LINDA AVEY
Co-founder
23andMe, Inc.
ESTHER DYSON
EDventure Holdings
REBECCA FISHER, MLIS
Patient Advocate
MATTHEW HOLT
Co-founder Health 2.0 Conference
Senior Consultant
Professional Services Solutions, Inc.
DEVEN MCGRAW, JD, MPH
Director, Health Privacy Project
Center for Democracy and Technology
RYAN PHELAN
Founder and CEO
DNA Direct
JEFFREY GULCHER, M.D.
Chief Scientific Officer
deCODE genetics
REED TUCKSON, M.D., FACP
Executive Vice President and Chief of Medical Affairs
UnitedHealth Group
MARI BAKER
President and CEO
Navigenics
KATHERINE JOHANSEN, PhD
Senior Scientist
American Medical Association Program in Genetics and
Molecular Medicine
NANCY JOHNSON
Senior Public Policy Advisor
Baker Donelson
RONNI SANDROFF, MA
Director/Editor, Health and Family Information
Consumer Reports
ANGELA TREPANIER, MS, CGC
President, National Society of Genetic Counselors
Assistant Professor, Clinical Center for Molecular
Medicine and Genetics, Wayne State University
MICHAEL COWAN, MD
Chief Medical Officer
BearingPoint
Disclaimer: The findings and conclusions of this presentation are those of the authors and do not necessarily represent the views of ASPE or HHS.
Introduction
DR. DOWNING: -- behalf of Secretary Leavitt’s Personalized Health Care Initiative and the Office of the Assistant Secretary for Planning and Evaluation, we welcome you here to the Ronald Reagan Building and the International Trade Center. And thank you for joining us for this afternoon’s workshop on consumer genomic information services. Now, before we start the conversations for this afternoon, we want to encourage you to engage in this conversation, and we ask (inaudible) that your questions today, you’ll use the microphone; it’ll be circulating at the various time points during the afternoon. This workshop is being broadcast via the web, and there are a good number of those that are joining us remotely and will be submitting questions, as well, so those will be picked up. If you’re listening in now, if you visit the registration website, you’ll be able to access the email address necessary to submit your questions. There’ll be several periods during the meeting today in which emailed questions will be entertained.
I’d like to turn now to Mr. Richard Campanelli from the Office of the Secretary and the Secretary’s Liaison to the Secretary Advisory Committee and a number of HHS Agencies. Mr. Campanelli was here today to offer the welcome from the Secretary this afternoon. He is the counselor for the Secretary in science and public health, and this is where services that policy advisor in representing CDC, NIH, FDA, and the Agency for Healthcare Research and Quality. And prior to his service as counselor, he served for nearly five years as a director for Office of Civil Rights where he oversaw the implementation of the HIPPA Privacy Rule, and now works as our advisor in the role of Liaison to the Secretary on the Personalized Health Care Initiative. Rick has an anniversary coming up this month that I just recall reading last night, and so we’re very pleased that he’s had such a leading role in helping us facilitate many of the aspects of the Personalized Health Care Initiative.
He has been a very important supporter and advocate for personalized health care and joins us today in setting the stage for this afternoon’s workshop. So, Rick, we’ll hand it off to you at this point.
Welcome, Meeting Objectives and Overview
The Secretary charged this Personalized Health Care Initiative with laying the groundwork here at the federal level, and then partnership with the private sector for a better future with a new kind of health care, truly individualized, personalized health care. We can all see the prospects of much more individualized care, much more effective medical therapies, earlier detection of disease, new powers of prediction and prevention of disease. We all want those things to happen, and rightly we’re quite anxious and a bit impatient for them to come across. Each of us in this room from where we sit know we are blazing a new trail, that’s partly why it’s so exciting, and we want to bring in that future as effectively as possible.
We’re here today to talk about an essential aspect of that future, namely, the interests and needs of consumers as this new realm of knowledge comes online.
This intersection of genomics and consumers has fundamental importance for personalized health care, especially because of new opportunities for consumer
engagement and for prevention that it presents. In recent months, we’ve seen that the traffic at this intersection between genomics and consumer engagement has become quite accelerated and there’s been a lot of public awareness about it. That’s going to continue as it should. So it’s great that we’re meeting today, that the Secretary’s Advisor Committee on Genetics, Health, and Society, SACGHS, is meeting today, and tomorrow also, and to focus on many of these related issues. And I’m glad to see many of the SACGHS members here.
As we stand here today at this intersection of consumers and genomic information, we’re actually looking at several different highways or roads that are converging right to this space where we are. The first one is genomic science. The completion of the Human Genome Project marked a huge scientific accomplishment, but as much as that was an accomplishment, that was just the beginning. That was just a starting point. And we’re all hearing about new genetic findings almost every week. As usual, new discoveries raise new questions, even as they’re providing new answers. And nothing about this field is standing still, and there’s no reason to think that things are going to slow down anytime soon. That’s a good thing. And as we should expect in any new field, how we communicate about these developments and what people hear is going to make a huge difference in whether consumers, providers, and payers will quickly and with confidence come to embrace the real potential that the advances in genomic health and personalized health care have the potential to provide.
The second highway that’s converging on us in this new and rapidly evolving -- the second highway that’s converging on us are the new and rapidly evolving
technologies that are being brought to (inaudible) in this area. That includes technologies that were nurtured by the Human Genome Project itself, like DNA microarrays. It also includes model information technologies, including both the rapid movement and exchange of information that we now take for granted on the web, as well as new kinds of information sharing and new powers of informatics. Unlike many advances in the past, these technologies are not just putting information into the hands of researchers. It’s not just specialists who are experiencing the information explosion, it’s all of us. That makes us ask new questions, questions that are changing all the time so we can better understand how the end-users will be able to use that information to its highest benefit in improving their health care and the public’s health care. So this brings us to the third major highway that’s converging here on this intersection -- the increased engagement of consumers themselves. Of course, in almost every field, web-savvy consumers are not waiting to be shown how the world is changing; they are leading the change in creative ways that could hardly have been imagined only a decade ago. This weekend I was up seeing my mom in New Jersey. She is 81-years-old, don’t tell her I told you to say that, she thinks that’s what the HIPPA Privacy Rule is about. Anyway, she’s 81, and her mom, my grandmother, was a classic Italian lady from the old country. When you said to her, “What’s the recipe for,” any given dish, you know, she would say -- you would say, “Well, how much of a particular ingredient,” you know, “how much bread crumbs should I put in this thing?” and her answer to every question no matter what you asked was, “This much.” She’d put out her hand and cup her hand, and that would be the answer to everything. She just knew and you’d have to be around her to get the information. But this weekend an interesting thing happened where -- as I was thinking about this talk -- is when I asked my mom for one of her recipes and, you know, my mom just said -- well, she started to tell us, then she said, “You know, it’s really much easier than that. Just go on the web, there’s a lot of great recipes available.” I thought, you know, this is a sea change. And of course, she’s also -- we talked this weekend about looking to the web to help her make some choices in health care about a drug benefit program that she’s thinking about changing. There are so many things that are changing, and my 81-year-old mom who, you know, not a great fan of technological changes, she knows about it and she’s excited about it. And she knows about changes that are being -- she has read up on possibilities for genomic health, and she asked questions about this and wonders where is it going and what does it mean? It’s very interesting that we’re having that conversation, and that’s a really good thing. In health care we’re encouraging consumers to take a more active role in their care. Their ability to do so is based in large part on the information they can access and use to make better health care choices. As that happens, all of us in this space owe them the support they need to make the best information and choices they can that are there before them.
So we all stand together at this busy intersection of genomics and consumer health today where these three roads converge -- advances in genomic health, new technologies that are being brought to bear in applying that science, new opportunities and access for consumers that take an active role in their own health care. That’s quite a busy intersection. And in this context, we need to find ways to encourage the traffic to move effectively and safely. That’s challenging, but we should expect these challenges whenever we get to new spaces like this.
As Greg mentioned, I was the Director of the Office for Civil Rights when the HIPPA Privacy Rule rolled out, and I was there for its initial implementation for a few years. There were two goals in that context that I remember we talked about. That -- I have some analogy that strike me as somewhat similar here. And we talked about -- and sometimes they were talked about as competing goals. There was the goal, of course, of protecting health information; that’s an essential goal. And at the same time, there was a goal of making sure that the information could still be both accessed and shared by individuals so that that information would be helpful to them. We didn’t want to -- we wanted to protect privacy and do it in a way that wouldn’t impede access to health care. And some -- a lot of folks talked about those as balancing between those two goals, but we recognized -- and I think all of you recognized that we needed to accomplish both of those goals. And it’s similar I think in some ways for the issues that we’re thinking about now where we have much more information, and we’ll have much more information available to all of us and especially to consumers.
In the personalized health care environment, we want to provide access to -- we want to help people be able to be good consumers of health care. We want to help them understand how they can improve their lives in so many different ways. And we want to do that in a way that’s accurate, rightly communicated, and rightly understood. These are challenges, but they are -- there is great potential in the improvements in individualized and public health that can occur if we accomplish both of those purposes.
Today we’re coming together to share our experience and perspectives on how in this intersection to put consumers first in personalized health care. We’re
all working in different areas, but do have goals in common for better health care and healthier population.
So let me just say a few words about the workshop. We’ve given it the catchy title, “Understanding the Needs of Consumers in the Use of Genome-based Health Care Information Services.” And we’ve got to work on the marketing of that, but that is a mouthful. The key word, though, is consumer and our key focus today. Our purpose here is to look at the ways that genomic information is going to reach consumers and then ask some basic questions. What are the opportunities here for consumers?
What are the cautions that need to be exercised? What tools do consumers need, and who can provide them? What are our different roles and how can we work together? I also want to keep our sights -- I hope that today all of you will help work to keep our sights set on the future. We were only a few years out from completion of the Human Genome Project, we’ve arrived at a time when some of the science and technology that was developed as the result of that project is being made available. But we are just at the beginning of the beginning. Among those represented among us today are some who are already
providing those services directly to consumers. Thank you for coming. We want to learn from you what we can so that we can all learn from your experience thus far. We want to learn from everyone in this room today so we can all be better at forward thinking in this arena. As the science and technology in this space continue to evolve rapidly, we need to ask ourselves what information will be available to consumers, in what ways and under what conditions can it help consumers achieve better health? And most of all, what can we do now to help achieve the best possible outcomes as these new capabilities and new opportunities come online? That’s the basic question for us today. In this area where consumers meet genomic information, and where new consumer knowledge is so important, what can we do now to make a better future? We have a half day, and that’s a tall order.
Mike Cowan is our facilitator. Mike is an Admiral and former Surgeon General of the Navy, so he’ll be using all his command skills to help us stay on course. We’ve already seen the good work you’ve done that way. Eric Topol from Scripps in San Diego will lead off with a view of what’s happening now and what we may expect in the future. Steve Bodhaine from the Yankelovich Public Opinion Survey Firm will provide us with a short portrait of consumer understanding an attributes in this space today. Then we’ll have our three panels with Q&A opportunities after each. And we’ll wind up with the discussion moderated by Mike Cowan, and Mike will be coming up here in a minute or so to introduce our first speaker.
Let me thank you all again for coming today. The Secretary and the Department share with you a strong interest and desire to see the day when consumers can confidently rely on every increasing array of genomic and technological advances to target preventative therapies, prevention therapies, and so much more. Thank you very much. [Applause]
DR. COWAN: Well this is an exciting afternoon. Again, thank you for being here. My role today will be kind of the traffic cop. Those of you who have -- and everybody’s looked at the schedule and you see we have an exciting topic, we have exciting speakers and panels -- and the audience -- I’ve looked through the credentials of the people who have come here to represent the entire professional spectrum of people who are interested in this topic. And there’s another 20 or more people who have joined us virtually, and we will work to get them into the discussion. So we have a big subject, lots of ramifications, lots of people with passionate interest in it. This is all good news, and Greg Downing, who was the introducer, the gentleman in the yellow tie; Dr. Downing is the Director of the Personalized Health Care Initiative. I don’t think I mentioned your name Greg, but he’s the leader of this whole effort today and has been working to put this all together. So I will try to keep us on track. We’ve all been to conferences and know that there are riffs on the theme on that we can take, and we shall. And I will talk some more about the ground rules and how we will handle that in a moment, but what I’d like to do is get us started right into the meat of things after I make just a couple of quick announcements.
There are bathrooms that are real close to us, but they’re not for us. They are under restoration and so restrooms are down the hall, down the elevators, bottom of the elevators take a left, and they’re sort of tucked up under the elevators.
If you have cell phones and have not turned them off or put them on (inaudible) already, would you do so? Everybody’s done so? Oops. I’ll do mine in a minute. And I think that’s all of the housekeeping we need right now. We have two exciting sort of keynote talks to get out our thinking juices flowing, and then we will go into a first panel followed by a break, and two following panels. And I get the privilege of introducing Dr. Eric Topol. He’s the Director at the Scripps Translational Science Institute. He has about ten other titles there, but if we read his titles he wouldn’t have as much speaking time and we’d be asking him to shorten it up. He is also the Dean of the Scripps School of Medicine.
Anybody know a graduate of the Scripps School of Medicine? Nobody’s graduated there yet. It’s a new medical school. Eric is in the process of putting it together, and he’s putting it together with the future of genomics as being an integral part of the future of medicine. I think it’s a very exciting project and I think you planned on saying a word about. So with no further -- Eric, please. [Applause]
Medical Decision-making Empowered by Genomic Information
And it’s been really eight years, so that was June 26 of year 2000, before we finally have seen what has been termed by science the breakthrough of the year. In fact, that’s not only the breakthrough the year for 2007 as it was announced in December, it will be the breakthrough of the year for the next few years because so much is happening so quickly in this space. The two major reasons for this, as I think most people here know, is that ultra high throughput genotyping became possible. In 1997, just over ten years ago, we could only measure one base-pair substitution at a time, assay it, and defined Moore’s Law, in fact, where there would be about 256 by 2007. We’re at a million or more SNPs per individual that can be assessed.
And the other major thing that happened in this space was that the genome, which has relatively unmanageable information, 6.4 billion base pairs in the diploid genome, was now managed by projects such as Perlegen Science and International HapMap breaking the genome into bins and being able to tag those bins, and having only about 250,000 to 500,000 being able to represent a window into the genome. And these two things -- the convergence of the technology, along with the breaking down of the genome into information bins allowed a remarkable state in advancement of human genomic knowledge.
Unlike any other field in science and biomedical research where there’s a hypothesis, this is one in which the genome talks because there is no a priori hypothesis. And the result of that has been a genomics gold rush, which we labeled as such last summer, and it hasn’t stopped at all since a year ago. In fact, I want to just briefly give you a table which shows on a weekly basis since April 2007 -- just about a weekly basis -- over 40 diseases have been approached via these genome-wide association studies relying on the high throughput SNP- typing and the haplotype map information. And you can see this transcends all different disciplines in medicines: cancer, metabolic diseases included obesity and diabetes, immune diseases such as Chrohn’s and lupus and rheumatoid arthritis; cardiovascular diseases such as heart attack, atrial fibrillation. And this goes on -- even Restless Leg Syndrome, which we didn’t accept as a medical condition until we knew the gene markers for this showed up, and you can see that this goes to gallstone disease, macular degeneration, and so on. And in fact, it’s virtually -- all the major cancers have been approached.
And just to take us up to date as of today, yesterday Nature Genetics had another third major gene for obesity, PCSK1. So this type of avalanche of new knowledge has set a template which has never been replicated in the last several decades, all in just a year-and-a-half time because of these breakthroughs. Let me use a few examples to hopefully demonstrate that there is actionable information today for consumers. So, for example, macular degeneration affecting 9 million Americans; blindness, the leading cause of blindness in our society -- we had no idea what was the pathogenesis of this disease. We knew there was this -- on the macula -- there was an inflammation, an accumulation of this inflammatory material known as drusen, and it led to eventual (inaudible) blindness. We also knew that there was a series of environmental factors, like smoking, high-fat diet, sedentary lifestyle, obesity, hypertension that were correlated with macular degeneration. But now we know the principle genes. The principle genes of compliment factors, which are the underpinnings of this disease, and this is what occurs in the inflammation pathway to be the root cause of macular degeneration. Well, why is this important? Now we can take a baby and say that that individual has 0 percent change of ever developing macular degeneration, or we can take an individual and find that they have a 400-fold.
And by the way, if that individual with the very high risk smokes, that risk could go up to 10,000-fold. And indeed, the environmental gene interactions have been assessed in this condition. This was the first genome-wide association study back in 2005, where we have the most knowledge about those sorts of important interactions. So already today we can give people who have compliment factor risk variance a choice. If they smoke, for example, they may have a much higher risk of going on to blindness, whereas a cessation of smoking is an important actionable item.
The chromosome 9p21 marker is a particularly important one in the cardiovascular arena because it not only catches the risk for heart attack, but also abdominal aortic aneurysm and intracranial aneurysm. These are all events that are very hard to predict with all the things that we have today. When do arteries crack or rupture?
Such as occurred in the case of Tim Russert just weeks ago. So this is a remarkable marker, 9p21, which shows a risk with one copy of 70 percent -- 35, 40 percent increase with two copies over 70 to a doubling of risk. And it is of many different conditions, which I mentioned are very difficult to diagnose.
What about diabetes? With over 20 million Americans having diagnosed diabetes, no less; many more in which this diagnosis is likely in the future or not yet diagnosed. We now have over 20 genomic markers of bins in the genome which correlate, each individually, somewhere between 20 to 30 to 40 percent increased risk for one copy, and this of course in many cases is additive. And some work has been done to integrate the risk of these different markers to show risk that ranges from 2-fold all the way up to 20-fold on the basis of an additive phenomena of different genomic markers.
What about breast cancer? It’s the guidelines that all women over age 40 are supposed to have a mammogram every year. Is that really necessary when most women carry no risk variance for breast cancer? And so now we have over 20 different variants that have been delineated, we can assess and partition the risk in women whether they’ll have breast cancer in their lifetime. And indeed, a New England Journal paper just two weeks ago modeled on this and talked about how what percent of the population was really at risk when we use the rudimentary genomic markers, no less the ones of the future.
The same is for prostate cancer -- just five different SNP markers in prostate cancer has in an additive way in this particular study published earlier this year. One can find a population of men who have a 10-fold risk of prostate cancer, and this of course overrides the knowledge of the PSA level in the blood or other known clinical risk factors.
Now even beyond that study that was published in January, we have 20 different markers in the genome for prostate cancer, so our knowledge base has been greatly expanded. So basically what is so remarkable about this time in medicine is that our understanding has been enhanced like no other and we have defined new genes and new pathways that are truly the underpinnings of disease.
And so the human disease (inaudible) which is represented here, and in fact that we now know certain pathways are responsible for multiple diseases which we would never have forecasted. In fact, none of these pathways were the ones that had been theorized before genome-wide association studies were performed. So this is quite remarkable in itself. And basically, as Andy Pollack reviewed in a recent Science Times, the textbooks of medicine are being rewritten. The only problem is that they can't be written fast enough because our whole appreciation of diseases and health is being turned over because of this vast avalanche of new information. I don’t want to submit to you that we know so much; in fact, there are lots of inconvenient truths. We still don’t have complete cover of the genome, we haven’t really focused on insertions, deletions, copy numbers to a great extent; there are many repletory elements and smaller (inaudible) that we have little knowledge as is the case for epigenomics and diplomics as well. But nonetheless, we are now into the consumer era, the consumer empowerment if you will. And this was forecasted in a very interesting Forbes piece a year ago when this fellow wrote that you can post on Craigslist, “Single, white male, HNPCC free seeks single, white female, no BRCA1/BARD1.”
And what he also wrote was kind of, you’re going to end up searching for genes on Google. Now this is of course an area I’m particularly interested in and I thought the guy was a little ahead of his time. Well, it wasn’t very long when I started thinking about this whole Google searching your SNP variance, and then I found out that of course like Wikipedia, there’s SNPedia, and any consumer can go to SNPedia and find every information that’s ever been published or resented about any particular SNP, which is quite remarkable.
And so many different articles have focused on this, such as the feature article in Wired, and of course those in the New York Times that were associated with a Pulitzer Prize in the past year about this whole interesting phenomenon. And in fact, three companies: deCODEme, deCODE genetics, 23andME, and avigenics are offering the genome-wide scans with either saliva or a cheek swab up to a million SNPs, continual updating through their internet browser setup at a cost, for some consumers, is affordable.
And also DNA Direct is involved in this, not only by offering special tests like the TCF7L2 in diabetes or the 9p21 marker for heart attack, but also in helping individuals interpret their genome-wide scan.
Now, there has been a reaction in the medical community that we’re not ready for this, and there have been several articles such as “Risky Business” in Nature Genetics, “Ready or not” in Nature, and “Letting the Genome out of the Bottle” in the New England Journal.
These are just representative of the naysayers, if you will. But actually, I tend to disagree with some of these editorialists. In fact, I’ve had my genome scanned through two different of these entities and I learned a lot. So I present to you, for example, I had no risk factors in my family of heart attack. It’s an area that I’ve worked on for the past 25 years. I knew had a risk of cancer. When I got my genome-wide scan, I found that I had two copies of 9p21, that was a big and important step of knowledge just for me, no less to know at least I was protected from some other diseases like obesity and some immune (inaudible) diseases. And the ability to interpret these data by these companies is actually quite remarkable. What they offer for the consumer is a terrific foundation for those who are not savvy, to understand what this means, that it’s probabilistic not deterministic and many other things are still wanting in terms of our knowledge base.
This is an example of the deCODEme to help me interpret what is having two copies of 9p21 variant, a risk factor for heart attack, what does it really mean? Very graphic and very simple in all of the companies in this space are remarkably consumer oriented.
So when I put this (inaudible) together at the end of last year about what you can learn from a gene scan, I thought (inaudible) this is a great movement. And the reason it’s great movement is it will help the physician community that are so reluctant to any change.
And in fact, the concern here is that patients now are coming to their doctor’s office to get help and interpreting their genomic data. And the doctor says, “What’s a SNP?” And this is a significant problem. And what’s going to change the medical community if not the consumer movement? And in fact, that’s paradoxical because we look at this survey -- it’ll be interesting to see Steve’s remarks -- this survey says, “Who do you trust with your genomic data?” Thousands of individuals responded; they don’t trust their employer, they don’t trust their health insurer, as you might expect; they trust the most, their doctor, interestingly who has very little if not any knowledge of this field. They trust their doctor more than their spouse and even researchers studying genetics, which is quite remarkable. And of course, in California, which is where I’m from and the recent cease and desist order by the state was quite surprising because this is, I think, represents a great advance in medicine, and oriented and advocating the rights of consumers. And this sense from the Department of Public Health in California that we are no longer tolerating direct-to-consumer genetic testing in California is so amazing to me, in fact.
So as I close, I just want to leave you with some examples of actionable information, why this is so important today for those who are interested. One, for example, the risk of diabetes or a heart attack, to know that risk, to know that awareness -- those symptoms that could be representing, for example, heart attack or heart disease is quite important, no less the change in lifestyle; the avoidance of 250,000 false positive prostate biopsies a year, for example; the use of ultrasound or MRI in those women who have significant increased risk of genomic markers for breast cancer. And the diagnosis of many elusive things, like abdominal aortic aneurysm, Chron’s disease, and atrial fibrillation as the cause of stroke of unknown ideology. All these things come out of a genome-wide scan. The benefit to consumers, I believe, is quite extraordinary. First of all, this is research-grade data. These are the same platforms, the same ways that data were obtained for all the genome-wide association studies that were published in the leading peer review journals like Nature, Science, and Nature Genetics. Secondly, it’s optional. It’s a right to know, and it’s a potential benefit of course in those individuals who use the information in a guided way. And the sad part is that physicians are uninformed, totally for the most part resistant to change, but hopefully can be prodded like the direct-to-consumer advertising model with respect to learning more, and motivated to learn about genomic medicine.
So I leave you with this representation of where I think the field has been and where it’s going. Would you consider this hockey-stick plot, and this was alluded to by Rick in his opening remarks. There was of course this draft human sequence in 2000, and many people including the public, have been disenchanted, no less the medical community, that it has taken eight years to get to the point where there’s relevant information coming out of studies to effect the practice of medicine, prevention, preemption for the first time. And so in fact we are now in 2008 well into this with consumer genomics, gene specific clinical trials, which we’re coordinating and other centers as well. Over the next few years, the ability to sequence the human genome -- whole genome sequencing, finding those wherever (inaudible) and those other inconvenient truths in 15 minutes is going to be possible. Soon enough, over the next eight-year span, we’ll have a million people fully sequenced, and some aspects of medicine, perhaps not all, will be routine, individualized practice. So in that -- with that framework, we set up a new medical school, Scripps School of Medicine, where every student who enters not only faces a five-year rather than a four-year curriculum, but has deep exposure to sequencing, genotyping, and all the ohmics including mass spec for metabolimics, and hopefully will be a group of physician leaders in the future to advance this field that needs leadership in the years ahead. So I just want to thank my colleagues at our program who have worked together to try to have a unique program that’s using the information of genomics today to advance the field of medicine, and hopefully this conference will achieve that laudable goal as well. Thanks very much for your attention. [Applause]
DR. COWAN: We did not rehearse Eric’s and Rick’s comments, though they said many similar things. We will pile metaphors up -- you get a hockey stick and converging rivers, but I think those all help give us visual images of -- a clarifying picture of a complexifying field that’s very early in its development.
Our next keynoter is Steve Bodhaine. Steve is the Group President for Research and Product Development at Yankelovich. This is an organization that’s been around since 1958 and specializes in collecting and understanding consumer attitudes, beliefs, and aspirations. They do interviews, they do surveys; and he is going to share with us some insights on consumers’ interests in health and consumers’ interest in genomic information. So, Steve.
Consumer Interest in Health and Genomic Information
Now, I have the ability to deliver a one-hour presentation in 25 minutes, which means that I will speak faster and faster as I watch Keisha (phonetic) tell me that my time is running out, so if you are translating today, get your lips in overdrive because this is going to be fast.
Let’s break into this. At Yankelovich we have been engaged in health and understanding consumer health for some time. And I just put this up here to give you a sense that this is not just coming from our back pocket, we really have spent a lot of time and energy to understand where the consumers heart and mind is relative to health. And we’re careful about the terminology we use because words like health care and health mean two very different things to the consumer. Wellness and well-being are two different things to the consumer, and so we have to be very careful with the words that we choose because the consumer is going to react in a very different way.
And please note that I’m referring to them as the consumer and not the patient. The day of the patient is gone; this is the day of the consumer. In fact, it’s the day of the health collaborator. And so we’re tracking this on a continuous basis and we want to make sure that you leave here today with a better insight of who these people are and what’s driving their (inaudible). I’m going to touch on a few key things. One, we want to introduce you to several different voices that exist in the marketplace. When it comes to consumer health we’re going to address maybe four of the dozen or so key health trends that we’ve been tracking. We want to then dive into a little bit of research that we did around personalized medicine and the consumers’ level of interest and understanding and engagement with genomic medicine. And then we’re going to get down to where the role of the physician might be in the future.
So the key thing that we want to emphasize here is that relevance is critical. We live in a day when we’re way beyond clutter in the marketplace. A good marketer, when it comes to clutter, adopts two strategies. I will speak loud and more frequently, which essentially just adds more clutter to the marketplace. We live in a time where the consumer (inaudible) active engagement we call marketing resistance. They’re taking active measures to avoid our communication. Health has been notorious for filling the airways with really lousy information from a consumer point of view. I spoke at a conference not long ago where one of my esteemed colleagues got up and was pointing fingers at the marketers and saying that these guys practice things like guerilla marketing and stealth marketing and viral marketing. And I got up afterward and I changed my comment. I said, “You’re right, Kelly (sp).” I said, “We do. In fact, the challenge with health is that we’re guilty of practicing confusing marketing and confounding marketing and conflicting marketing. And we’ve done a pretty good job of disengaging the consumer in much of what we have to say.” And so if we’re going to deal with this marketing resistance, we have to adopt some new strategies.
And now, just out of curiosity how many of you have signed up for the Do Not Call Registry? All righty then. Just a brief moment. You do know that market research is exempt from that, so when we call we’d appreciate your candid responses. What you’re really signed up for is not to avoid research, but what you’re signed up for is to avoid being called at dinnertime about something that you don’t care anything about. And so consumers today we understand that with TiVo and satellite radio and Do Not Call Registries and anti-spam legislation, we’re taking active measures to avoid the very things you’re trying to communicate with us. And so we have to make sure that in today’s marketplace we are more precise in defining who the consumer is and is not and more (inaudible) we deliver to them than we’ve ever been before. And further, we have to seek power to the consumer and change the rules of engagement so that the consumer begins to dictate how he or she plays in this space. And when it comes to health, we’re seeing that happen in a very real way.
Well, let me talk to you a little bit about relevance today and some of the voice of the consumer. We did a study in 2007 in 17 countries with tens of thousands of consumers. And what we were looking for is a way to take a very heterogeneous population and put them into homogenous buckets so that we could better understand how to engage the consumer in health and in health care. And so let me share with you six segments of the population.
Segment number one is a group we called “Leading the Way.” This is a group of people who get it. They organize their whole life around health. Now, they may have some chronic conditions, but they have a normal BMI, they exercise on a regular basis, they are avid information-seekers, they get their screenings as they should. These people organize their life around health.
Maybe you know one of these people because there are not very many of them in our country. In fact, they comprise about 10 percent of the population. They tend to be a little bit older, but the key thing with this group is that they have an inter-locus of control and they have a future orientation, which means you communicate into this group that the reason that you’d want to get genomic testing would be to help you avoid the future risk of disease; that would work for this group.
Group number two is a group that we call “In it for Fun.” This group is otherwise healthy, but not because health matters. They’re healthy and they exercise because they enjoy the competition. They want to look good, they want to feel good, they want to have the energy to compete. This is how they organize their lives. They do practice good healthy behaviors, but this is not a strong health mindset and orientation. And so if we’re going to reach out to them, delivering a message that avoids the future instances of health risk is probably not terribly important. We need to talk to them in terms of what it means to their social life and how that might impact their ability to compete and be aggressive in the marketplace in which they operate. So this group actually is good; we like them, but they’re not going to resonate very powerfully with health messages per sé.
The third group is the “Value Independence.” This is a fun group; we call them the do-it-yourselfers. This group is so tired of science and medicine creating confusion in their lives that they’ve determined that they can figure it out on their own. This is the do-it- yourself diet club. They mix and match until they find that works right for them. Unfortunately, they continue to get gain weight; they’ve not been very successful with their do-it-yourself technologies and have created a whole host of challenges for them and for their families. They don’t necessarily trust the voice of the physician. They think in many cases that medicine and science are overrated. And this is a group that’s turning more and more to alternative medicine and looking across the pond for new kinds of remedies and interventions that may prove to be a more positive intervention for them than traditional medicine. Very interesting group, hard to reach, they don’t want to hear your voice. This is a group that’s going to pay an awful lot of attention to social networks. These are bloggers-extraordinaire; they’re going all over the place looking for information from people other than the scientist because they don’t know that truth is found necessarily in science. I’ve been guilty of this -- well, I won't go into this story because I don’t have time, but another time. The next group is a group we called the “I Need a Plan.” We lovingly refer to them as the undisciplined. This is a group whose heart and mind know what to do but whose body simply will not obey. They know that they need to lose weight and they will start a diet, and then they will stop a diet. They will begin to exercise and then they will stop exercising. They need structure. They have a very extra low locus of control. They need the health care professional to intervene and help them to get with the plan and stick with it so that it can have success. These guys spend a lot of money on health; they are actually very well informed, but they are looking for partners who can help them start and finish something successfully over time. We like these people a lot because they are willing to engage. But this is a group that doesn’t need one more piece of information; they simply need help in applying the information in their life.
The next group is “Not Right Now.” We refer to these folks as disinterested. This is a group that is relatively healthy, but keep in mind, the disease is what happens to somebody else. They are a bit younger; they are generation invincible and are not likely to engage with health or health-related information at all. This is a group in the world of food where we get all excited about organic food and natural food; this is the group that when Hardees rolls out says 940 calorie breakfast burrito, they were in line four days a week because it tastes good. This is a group that’s going to do what they want because it helps them feel good about themselves.
This is a group that has Aunt Sally. You know Aunt Sally; she’s 97-years-old, she started smoking when she was 3, she drinks like crazy, but she is still ornery and full of vigor and we’re going to be just like Aunt Sally. This is a group that is very difficult to reach because they’re simply not listening to health information. They’re potentially a train wreck in the future because they are gaining weight and they are engaged in very unhealthy behaviors for the most part.
The last group is a troublesome group. This is a group that we call “Get Through the Day,” often referred to as given up. They have been afflicted with poor health for the majority of their life; nothing they’ve tried has produced a meaningful result. They are frustrated and basically have resigned themselves to poor health for the rest of their life. Unfortunately, they tend to be a very expensive consumer in the health space; they have many chronic conditions and they present themselves often in the most expensive health care delivery venues possible.
And so they’re a group that we have to pay a lot of attention to. This is the group that disease management companies focus a lot of energy and attention on. But we understand that this group will never get anywhere on their own; self-help tools will be completely unsuccessful. This is a group that’s very dependent upon professionals to help them experience any kind of benefit.
Now, I throw these six out very quickly. We have a ton of data behind each of these people. We’ve looked at 40 different chronic conditions, we’ve looked at weight management, smoking cessation, exercise, sleep management, stress management, all kinds of things. As we look at these kinds of people to understand how and where and why then engage or disengage in the health debate.
And what I want you to take away from this is that one message will not fit all, nor will one solution fit all. And we have to make sure that we’re reaching out to these people in a very targeted fashion if we hope to engage them in improving their overall health and wellness.
And when I speak of wellness, I want to get into some specific trends and some definitions. Number one, we’ve been measuring for the last four or five years, the evolving health mindset. What you need to be aware of and what you’re already probably very well aware of is that health today is a holistic view. It is a combination of mental, emotional, spiritual, and physical wellness. My concern with this trend right now is that the mental, emotional, and spiritual dimensions of wellness are actually masking the physical reality of disease. We asked people to tell us how many chronic diseases they suffer from, with which they’ve been diagnosed by a medical physician or professional, and what we’re finding is that people who have even more than three chronic diseases are listing their overall health as being good or very good. Now, why in the world is that? It’s because they have a positive outlook on life. It’s because they have a sense of purpose. It’s because they have people who love them. And besides, I don’t feel any different whether I take my hypertension medication or not. And so what we’re finding is that there’s a huge emphasis on that. In fact, we ask people, “What do you do to improve your health?” And what do they tell us? “Oh, I need to stop smoking. Need to lose weight. Need to exercise more. Need to eat better. Need to get a little more sleep.” Very physical in its orientation. When we ask them, “What’s the most important things you can do to maintain your health and wellness in the year to come?”
Number one on the list is to make sure I have good insurance. Right behind that is to practice good hygiene and personal cleanliness. We’re glad that we’re washing our hands more. What’s interesting is that exercise doesn’t even make the top ten. The diet barely cracked the top ten this year; it was number 12 in 2005.
The physical reality is not nearly as important to the consumer as the mental, emotional, and spiritual dimension. If we’re trying to change physical behavior, the take-home message is that we’ve got to couch it into the context of this mental, emotional, spiritual dimension or we will not cut through the clutter or the resistance in the marketplace. So keep that in mind. And, oh by the way, I am not a patient and I am not a disease. And I refuse to be defined that way. I am not a diabetic, I am not a hypertensive, I am not dyslipidemic; I am Steve.
And, oh, by the way, I’ve got these things that interfere with what I’m trying to accomplish in my life. If you can help me with that, that’s great. So keep that in mind as we’re engaging the consumer in today’s health marketplace, it is a holistic view. And we know that even those individuals who are mentally, emotionally, and spiritually engaged with health, the physical dimension factors in very nicely because physical health -- a crisis actually will disrupt or trump these other dimensions of health and wellness.
Number two, the thing to keep in mind, in terms of particularly genomic health and medicine and where we’re moving today is that home is becoming the center of health. We are living in the world of the Baby Boomer. Maybe you know one; they may be seated next to you. They are kind and nice; be gentle with them. One thing we know about Boomers is that we will never grow old. We refuse. We are redefining retirement, we are redefining age. And we are absolutely confident that we will never need to go into long-term care. In fact, we don’t want to. We’ve been in to long-term care before and we don’t like how it smells. And we don’t ever envision ourselves in that environment. And so what we’re seeing more and more is that the harm is becoming the center of health. I started a hospice company some time ago, and what was interesting is before World War II, people would pass away in their own home. Post-World War II, the single leading indicators where people would die was the availability of the hospital bed. Medicine changed. Well, I think it’s going to re-evolve, that the home is going to become a place where much of health is actually delivered. And so we’re watching that carefully. One of the reasons behind that is because we live in a world of the multi- generational caregiver. And perhaps you know them too, they tend to be female. They’re caring for an aging parent, they may have an ailing spouse, they may have children who are experiencing chronic disease earlier and earlier, but they have their hands full. And the market is recognizing that and is quickly coming to their aid and looking for everything they can do to empower that individual to maintain his or her own health and wellness, as well as to be good custodians of the health of others with whom they’ve been entrusted. So keep in mind that the home is going to be the center.
We know that more and more of health care will be delivered in the home. We’re seeing advances in telemedicine. The whole rise in in-home diagnostics is very impressive and will continue to be there because: one, it caters to the fundamental need of convenience; and that’s an important thing in the mind of the consumer.
Which leads me to the next thing, and that is the idea of diagnosed need. In a very cynical world, which we’ve trained the consumer to live in, we have determined that we can trust ourselves as much as we can trust anybody else. And so I want to be able to get a firsthand glimpse of my own symptoms. I’ll show you a slide here at some point if I get to it, that shows us where the consumer is going for health information and why they’re going there.
There’s a massive generational differential. The mature generation still is a bit of the Marcus Welby, M.D. generation where doctor knows best. The rising generation may go to the doctor as the third or fourth voice in the health continuum. That ought to cause a bit of fear, and it’s given how well some of the younger generation takes care of themselves. But nonetheless, we are looking for not just information but tools. And if I could just submit one thing, we don’t need probably another website with health information; we can find cancer information now I think on 200 million websites. Okay. It’s enormous. The consumer came to get a drink of water; we turn on the fire hydrant. I went from a period of complete disengagement to opening the internet and creating a floodgate that makes it virtually impossible for the consumer to differentiate what is truth and fiction. What the consumer needs is accurate information delivered through a credible source with passion over and over again. But more importantly, I need the tools that help me interpret that information in a personally relevant fashion and give me the power to actually do something with it. And our idea is not the BMI calculator. Anybody gone on to do the BMI calculator?
Anybody ever play with that? Only two honest people in the group. Yeah. What’s interesting is that you take it once and you’ll find out that you’re maybe not within the normal range; it’s amazing how fast you grow. You know, you’re now 6’, now you’re 6’4, and it feels a lot better in that range. That’s not going to cut it in today’s marketplace. The consumer needs to be endowed with tools that empower them to take action and to monitor that action. But it’s not just the tools, we need improved access to competent health care professionals who can help us interpret and manage that information and help us monitor our progress so that we really do achieve the (inaudible) that we’re seeking. And all of that needs to sit in the world of personal health accountability, a term that has not been introduced to the consumer but is coming very quickly. We’re tired of waiting for the federal government to drive change, we’re tired of waiting for the state government to drive change, we’re tired of waiting for the municipal government to drive change. The employer is now firmly engaged in this. I work with a lot of employers who have launched massive health and wellness programs to engage the consumers to change behavior, and they’re holding them accountable. And we may not like their tactics, but nonetheless it’s coming.
The next stage will be the individual themselves because employers will look for ways to lessen the liability of health care and responsibility for that.
The last piece is self invention, which is interesting. We’re figuring out new ways to invent who we are, what we’re all about, and there’s a lot of interesting medical information there that I won't touch on because I’ve already had my ten minute warning.
We’re going online because we wanted to research specific diseases and illness, but interestingly, we want to diagnose the symptoms that I have. Now again, a scary thing, but we’re going on and finding diseases that we never had before.
Let me get into specifically some of the research that we did around consumer genomic medicine. We did fundamentally qualitative information for the purpose of this to provide a snippet and insight into where the consumer’s head is. And what we find is that when we talk about genomics, that the consumer has some degree of familiarity, but very limited understanding. And so we say that the familiarity with genetic testing is pretty limited. And what we find is that we know a little bit about what it is, but we don’t necessarily know how it will be used and how we can apply it to our own health and wellness to our personal success. We are not aware that there are companies out there who actually are doing this.
We think this is being done by lots of other people and other institutions, and we don’t really have a clue about what the cost associated with genetic testing is. So again, a very good indication, those of you who are living in this space have probably done a lot more extensive research, but we know that this is the beginning days for genetic testing and the consumers are interested, their curiosity is certainly piqued, but they don’t know a lot about it and necessarily how to take action. What’s interesting is that they give us a lot of the right answers. You know, it’s a procedure used to find out the makeup of a person. It’s completing a series of tests to determine various things such as health concerns. They can articulate at least the surface level of what this is about, but again, don’t know a lot about how to use it.
Who is it for? Well, everyone, some say. Children, parents, and grandparents, people who are overweight, babies and children, there’s a whole range of potential users of this kind of information. Why do they get it? Some think it’s a preventative measure, some want to know how much time you have -- which I thought is a little bit of a morbid thought, but -- should tell you to use your time any differently. But they certainly see advances in medicine. When we ask where do they go to have it done, some are going to go to the doctor’s office, some to the hospital, some to the university hospital, some to the specialist, some are going overseas, and some in an approved facility. Not too many people are going to a DNA lab, and certainly people are not thinking about doing this in a third-party remote kind of fashion.
Am I willing to consider it? I’m interested, but I’ve got to admit, I’m a bit skeptical right now as the consumer. I don’t know -- really, if I got a negative result back I would probably still go talk to a physician anyway just to be sure. And so what we’re saying is that, yeah, I’d be interested in considering this thing if I have an increased element of risk. They want to know more about the information. The biggest concern is about accuracy. If I go and get this done, how do I know for sure that it’s me you’re talking about, particularly if I don’t necessarily agree with the results. They’re not as concerned about privacy; they assume that’s a given and would expect you to take good care of that information.
But they are skeptical of getting something that doesn’t come directly from a physician. And again, we talked earlier that the physician is my most trusted source of health information, (inaudible) accessible, and there may be a misplaced sense of trust there. But the law of proximity is very much alive when it comes to health. And consumers are going to trust that individual which is closest to them and whom they believe is objective and has their best interest at heart. The (inaudible) genetic testing means, we don’t know how it’s used. We hope that it’s going to give us a better understanding of my risk for disease, and that it will help provide a blueprint for me to take more poor health and to plan more effectively for my future, but I’m worried that I won't be able to understand what comes back to me. I don’t know that I will be free from any kind of discrimination if others find out that I may be predisposed to a certain type of condition. And so the hopes are there, the challenges are there, but they don’t weigh heavy on the mind of the consumer.
What does my doctor say? Well, again, we’ve mentioned this before. The doctor’s voice is very important. But again, the rising generation is turning more and more to the web. What’s amazing to me is we look at the consumer today, they are actually looking more and more to the blog for an empathetic ear and they are trusting people in these social networks as much if not more than their physician when it comes to certain types of conditions. They’re looking for approbation around a certain type of a new type of medical device or drug or intervention based on what other people in the market who are like them have to say, even if they’ve never met them before.
So it’s a great day for this. What we’re finding is that there’s a market out there that consumers are very interested, that they’re excited about the prospect, but it’s a great unknown and there’s still a lot of learning to be had for them to take advantage of genetic testing. The key is, give me the tools so that I can interpret the information that I get and take the appropriate kind of action. Again, many voices, many consumers. Not every one of them is going to jump on this and take advantage of it, and we have to recognize that and make sure that we’re targeting our efforts to communicate with them and engage in the process going forward. So if you want to know more about the consumer, we have a lot to say. Appreciate your time today, and we’ll turn it back. Am I on time? Good. [APPLAUSE]
DR. COWAN: I did not know you could talk that fast. I have a mint if your mouth is kind of on fire. Thank you so much. Those are our three presentations. I think you’ve probably all noticed the same thing I did, there was a great deal of convergence between the three. And sometimes when speakers get up and say so much of the same thing from their different perspectives, it can seem redundant, but I would counter that some things are worth redunding because that has set a tone that will then, I think, generate a conversation that we are going to try to bring out in the three panels.
So would Esther Dyson and your panel come on up? And we’ll go ahead and we’ll shift the panels out as I introduce them. And again, one more for our speakers. [APPLAUSE]
I hope that we have set a perception, and I would be surprised if many of you would not give many of the same points of view. You’re all professionals in this field from one aspect or another, that there is going to be a very different role of the consumer going forward in this particular aspect of medicine, others too certainly, but certainly this one; and that there’s going to have to be a different professional approach to genomics-based medicine than we have used in our traditional past.
I hope you have a feeling that we are early in the game and that we are going to try to spend the rest of our time looking through the eyes of the consumer. You can go ahead and sit there. Yeah, Yeah, yeah. We won't consider this a success -- this day a success -- I’m speaking for Greg and his team planning this -- we will consider the degree of success the richness of the conversation we have. And this is not just occupational therapy for us to (inaudible) away an afternoon. You know, the history books of the Manhattan Project have just recently come out. Enough time has gone by and the historians asked the scientists, “You invented a whole new field of science and then you invented practical applications to it and then you (inaudible) practical applications and you made them work. And you did all that in about 18 months; how in the world did you do that?” And they universally came back to the historians and said it was the discussion, it was the dialogue, it was the conversation. This -- I don’t think it’s too much of a stretch to make at least an analogy to the Manhattan Project. This is a huge sea change in medicine. We are at the verge of it, and you are the ones who will create it and you are the ones who will have the discussions and have the dialogues. The scientist said, you know, “We’d have a problem and we didn’t know what it meant. And then we would have these discussions and then there would be a solution, and nobody really claimed to know where it came from. It was all in the dialogue.” So I think this is a very important day.
We’re going to go into the panels now so I want to set the rules. So that’s the expectation -- that you’re engaged, we have a conversation. Here’s the rules. One, of course, a pesky rule, but no hitting. Well, no more than necessary. Second, I already asked, please participate. When you have a question, we have microphones that I don’t see, but we will. Raise your hand; we’ll get a microphone to you. It’s being recorded; we want to keep this, we want to save it. And get a microphone in your hand, tell us who you are and why you fight -- who you are, who you work for, and then ask the question. And I’ll help moderate the questions, or feel free to ask a particular panelist or the panel head.
Please be concise for the sake of time. We’re doing okay, but try to keep it in mind. We will -- and now, here’s my job. That’s a parking lot; this room is full of passionate people who have a lot of opinions about a lot of things. Some of them directly bear on other doings, some don’t; all are important and we want to capture everything, but we cannot take the discussions down every lane. And so I will be very arbitrary and capricious, I admit it right now, that if we’re going off in a direction or something seems irresolvable or for whatever reason, we’ll put things in parking lots. And the reason we’ll put it in the parking lot is so that we don’t lose it. We’re early in this process, we’re early in the development of this branch of science and medicine, and so nothing will be lost.
Are there any questions about the rules or the engagement? What we’re trying to do at this point where we are? I see no dissent. This panel -- I heard a dissent, what was that?
Panel 1: What is the Consumer Interest in Genome-based Health Information?
MS. DYSON: That’s okay. (Inaudible).
DR. COWAN: Okay. Her gene -- well, and she knows that her genome was sequenced and published as one of ten volunteers on a personal genome project, so she’s got it both for personal and professional interest in this. And Esther will then introduce the other members of this panel. Each panelist will have an opportunity to make some comments. These are a little bit scripted just because we wanted to focus on the topics at hand, and then we will open this back up for questions and answers.Okay. Ms. Dyson, it’s all yours.
MS. DYSON: Great. Good afternoon. I’m not going to give a long talk, but what I am going to do is stand up here so that I can keep order. And I do want to keep order, not just for the panel, but for everybody. I really would like this to be interactive. So I want to start -- I know you’re not representative, but how many of you have had your genome sequenced in some form or other? Okay. How many of you would do it if it were free? And how many would never do it? Okay. If anybody changes their mind during the course of this panel, let us know because that would be interesting.
What we’re doing here today is having three panels, and ours is pretty much what the consumers want. The second panel is what the consumers are actually getting, and the third panel is what the consumers are going to get. So we’re trying not to step on each other’s toes too much, so any panel could talk about all these things. And what I’m going to do is have each panelist introduce him or herself -- you can read the bios, but there’s probably a little color or subtlety that’s missing -- and answer the questions that are in the book. They can spend maybe three, four minutes, I’m going to ask some follow-up questions, then we’re going to talk among ourselves, and then we’re going to bring in audience questions. If somebody can't restrain themselves out there, you can ask questions anytime because I want this to, as Michael said, the value comes from the dialogue so that’s what we’re going to try and do.
We’re going to start with Rebecca Fisher, who is what has been missing in many of the public discussions, which is the actual voice of the consumer, the person concerned; and then Matt Holt, a well-known health blogger and (inaudible); and finally, Linda Avey, who is a co- founder of 23andMe. I’m not going to talk about my own bio except sort of by way of disclosure; I’m a member of the Board of 23 and Me so I’m going to be especially vicious. Rebecca.
MS. FISHER: I don’t remember what I gave to you for the bio, so I’ll recap by saying that I’m a 47-year- old breast cancer survivor, BRCA1 positive, diagnosed at the age 31 in the early ‘90s.My two points today are meant to temper the rhetoric about the excitement about all that we are learning, which is not to say that I think it’s a bad thing; I think it’s a wonderful thing, but I see the naysayers that one of our presenters spoke about before as being more proceed-with-caution-sayers, and I think I agree with them. The reason that I agree with them is that most consumers are not familiar with the methods or even the vernacular surrounding genetic testing. The methods that are used and the clinical utility, the clinical validity, even the reagents that are being used, are words that belong to something very foreign to most people.
I’m a medical librarian by training, so most of the terms come, you know, with difficulty but I can figure out what they mean and I can also figure out where to find out more about what they mean. But in this emerging world of genomic information, there’s a real gap between the information that someone can download and the information that someone actually needs to use to make valid decisions about his or her health.
When my family became involved with linkage analysis in the early ‘90s, there was no BRCA1. BRCA1 was discovered in August of 1994. At that point, my family entered a research program at the University of Michigan which later moved to the University of Pennsylvania. I have two sisters, one older, one younger. They both were involved in the research and couldn’t wait for the results to be returned. As a result of their impatience, having seen me go through bilateral mastectomies, a bone marrow transplant, and two months of radiation, they went ahead and had prophylactic mastectomy, both of them. When the information came back from Myriad that our notation was on an intron, which, you know, that’s very odd for BRCA1 -- it was on an intron -- and it was not found in the research setting, so not all research methods are the same, which was news to us. But they were testing our mRNA, they were not testing our genomic DNA. Most consumers don’t get that difference. My sisters are still a little tiny bit upset that they don’t have any breasts, and I don’t blame them.
The second issue that I’ll talk about briefly because I know Esther wants to move us on, is a friend that I have who is a banker. She’s a very bright woman, very capable, 49-years-old. Recently -- very recently, two weeks ago diagnosed with breast cancer -- Stage 1, but they didn’t get the margin so they were saying to her, “What do you want to do? Do you want to go back and get more surgery? Do you want to do another lumpectomy? Do you want a mastectomy?” We had a conversation at a Starbucks at which I was able to tell her about BRCA1 -- hadn’t heard about it -- and didn’t realize that this might be a risk factor for her. "Well, Joann (phonetic), what’s your family history?” I asked her. “Well, my sister had a glioblastoma when she was 18, my brother had lymphoma at 22.” I said, “Did you tell your doctor that?” She said, “Yeah, and he just moved on.” So what I’m suggesting today is that there is a gigantic gap between what someone can download, even what someone can find on OMIM -- even what someone can find in gene reviews or the new collaborations that are coming up. There is no person standing at the point of decision for that patient. The only person is going to be their genetic counselor or maybe a medical librarian or, God love them, the physician who took the time to learn that this is a subtle and nuanced world, and we should proceed with caution. We have no deadline. And those are my comments for you. Thanks. [APPLAUSE]
MS. DYSON: Those are compelling stories, but the message I actually get from them is -- has very little to do with direct-to-consumer genetic testing and probably more with the overall level of knowledge not just among consumers, but among doctors and other people. And so what would your constructive advice to this room be about how to help solve some of these problems?
MS. FISHER: Well, I guess I’d kind of disagree that it doesn’t have to do with it because no matter how you get the information, whether it’s direct-to-consumer or through a research setting like we did or from your physician, you are going to have information. What concerns me is the commoditization of human life. That concerns me greatly. And when a kit comes in the mail for you to turn in a cheek swab and there’s no human being there, oh, yes, “We have people on call 24 hours,” whatever -- that person is -- I’m just thinking that person is not going to be equipped. If your own doctor is not equipped, I have major concerns. And so I guess I side with the proceed-with-caution-sayers.
MS. DYSON: But how do you get the doctor to be equipped?
MS. FISHER: Well, that’s the dialogue. That’s what the problem is. Doctors, a lot of them, get their information and I see Father Fitzgerald out here -- he knows it as well as I -- at Georgetown University School of Medicine, in the cafeteria, that’s where they get their information. And that is something a medical librarian will rip her hair out over, but that is the reality.
MS. DYSON: Okay. Well, we’ll definitely come back to that. Matthew, your turn.
MR. HOLT: Sure. So let me in two-and-a-half minutes, if I can, say three things. I’m Matthew Holt, I write the health care blog, I run the Health 2.0 Conference, and I would be running a genomics direct-to- consumer genomics company in California; unfortunately, I’m not a blonde female which is a major requirement as we’ll find out later.
First, a couple of things. People are going online to the web to get information because they want action and results out of what they’re getting. They want information which gets them to do something. And my major concern at the moment about direct-to-consumer genetic testing is it doesn’t necessarily give you something you can actually do out of it, but that’s a question I think that will evolve. And I think Eric Topol’s talk was very instructive about what’s going to be coming. But if we’re going to be waiting for the wider point, which is doctors to adopt all these new information technology and deliver it in a human and humane fashion to patients, we’re going to be waiting a long while. In fact, for all of Eric’s new graduates to graduate and come through the system in about 25 years, and by then we’ll be dead or close to it.
So I believe that there is a lot that can be done online in terms of tools and advocacy, which will be emerging as either a market-based or maybe as a social insurance base to technology to come. So watch that. And to my mind, direct-to-consumer genomic testing is a big part of that.
Second -- two other things that are worth saying very quickly. The first is that there’s been a lot of fuss about privacy online in general, and genomics in particular. And the major fuss that I can see is about the impact of disclosure of information. Unfortunately, we live in a world -- or live in a country and society in which the impact of information that you are not, you know, involuntarily disclosing but forced to disclose by insurance companies and others, can dramatically impact your life. If you apply for individual insurance coverage in most states in this country and you say you’re a particular disease, that either means you will pay a lot more for that insurance or you won't be able to get it at all. And that is out in the open and irrelevant to the current discussion. Now, my view is that we need to fix that first, and then work about genomics and privacy second.
Secondly, there’s obviously a lot controversy in California and New York about the impact of, should consumers be able to go out and order these tests directly. So I am talking out of both sides of my mouth here. I’m a good Marxist -- chemist-trained Marxist and I believe in socialism and social insurance. And I also believe in understanding what’s cost effective in medicine and what’s not cost effective. I don’t think there should be a blank check but for the government to pay for all medical care, but I think that stuff that has been proven to be cost effective should be covered and it shouldn’t be impacted to the point of care by your -- the size of your wallet.
So I believe in social insurance, and I don’t think it’s clear yet as to whether most genomic testing actually is cost effective, and I hope that the work that Eric and others do, will figure that out. But having said that, I don’t believe in trade protection. And, you know, if you are using the state and regulations as an attempt to protect a profession or your economic interest, you shouldn’t be able to do that if there is a better, cheaper way of getting things done. And I think that most of what we’re hearing at the moment in terms of restricting by state licensure and other types of regulations to restrict this kind of activity, as well as much other activity in health care falls into that bucket. So I think in the end, if consumers are going to be adopting genetic testing in a large-scale format, it’ll be done because it’s done in conjunction with the health care system and with their current relationships with physicians. And I think that all the direct-to-consumer testing companies here are either adopting that position or will adopt that position.
But nonetheless, it doesn’t mean it should have to be that way. So with that, I’ll shut up.
MS. DYSON: Okay. And how would you solve Rebecca’s problem of under-educated doctors, even if they don’t want anyone else doing it -- they’re not capable of doing it themselves?
MR. HOLT: Well, I mean, the first thing is you have to introduce some level of competition into that, and that could be competition from other doctors because there are doctors who will get educated and medical groups and organizations. And I will actually solve her problem a different way. I think there is a huge need in this country for medical advocates, and that’s a -- in my mind -- a perfectly fair commercial organization. There are enough Americans, you know, who have the money -- if you have the money to pay $1000 or $2000 for a genomic test, you certainly have the money to pay $50 or $100 a month for -- to handle advocacy issues for you. And I think that that market will develop. And this is one of the areas they’re going to develop it for.
MS. DYSON: And as a good Marxist, what do you think about the people who don’t have the money for that?
MR. HOLT: I think if they need it and its cost- effective, the government should pay for it.
MS. DYSON: If we can prove that it’s cost effective.
MR. HOLT: Well, I think, you know, at the moment, this is an entirely different debate.
MS. DYSON: Yes.
MR. HOLT: In the moment, we pay for an awful lot of stuff that isn’t cost-effective and everybody knows that, and Medicare writes the check every month. And I think that should change, but that’s not what we’re here to discuss --
MS. DYSON: Okay. Fair enough.
MR. HOLT: -- (inaudible) on that, I can give you one, too.
MS. DYSON: You’re right. Let’s move on to Linda Avey.
MS. AVEY: Thanks, Esther. And thanks everyone for coming. This is a great group, it looks like. I’m excited to hear your questions. I come at this from a completely different direction, I guess. From Rebecca having worked in the research community for over 20 years and working very closely with people like Eric and people who are really trying to discover these genetic markers that hopefully someday could lead us to personalized medicine and personalized care. And it was while I was with technology companies like Affymetrix and Perlegen that we kept banging our head against the same wall of trying to identify enough people who could be part of large-scale studies so that we could make these discoveries very quickly and utilize all these great tools that are being developed. And it was because of that frustration that I was sitting around talking with colleagues at Affymetrix one day and, you know, how do we change this paradigm?
How do we move this beyond our current infrastructure of typically NIH grants that get funded to a very few PhD’s typically who put in applications for them, and a lot of times their budgets might get cut back so that they have to cut back the number of people they enroll in their studies. And it’s all about statistical power, and if you don’t have that, you don’t get to the endpoint you really need.
So I’m really sympathetic to Rebecca’s situation. What I feel we’re doing at 23andMe, is we’re really arming individuals with the information of their genomes, but we’re not really focusing so much on the specific test. But what we’re doing is giving our customers information about what’s coming out of the research community. And as Eric demonstrated, there’s just a flood of data coming out right now, but it’s research results. It’s not clinically validated yet. And that’s where we see what we’re doing now with 23andMe is providing a mechanism for taking these results and giving them back to our customers but then asking them -- let them be participants in a big part of this move from research into the clinic and let them tell us what diseases do you have? What problems are you having taking drugs? Did you have a severe reaction? And once we can compile all this information together, then hopefully we’ll get to the endpoints where people can start understanding it better, understand their own genomes, and then hopefully at the same time be working with the medical community. It’s going to take a very holistic effort, as was mentioned before. We need to work together as a community. No one player in this space is going to make this happen. So we’re very hopeful -- I myself personally, I wasn’t diagnosed with my WPW until I was 31.
I’ve had severe reactions to two different antibiotics, to a point where I had drug-induced lupus. This has got to stop happening. I don’t want my kids to have to go through the same problems that I’ve been going through all through my adult life. So it’s really a vision we have for the future, and we’re hoping that 23andMe will be a platform to really gather up this information and put it into the hands of the people it matters the most to.
MS. DYSON: Thanks. That was actually an answer to the third panel, which was, what do people get eventually? So let me ask you, what is it that -- because you’re the one on the panel who actually offers such a service; what is it that people want when the sign up for 23andMe? Why do they do it?
MS. AVEY: Well, we’re just starting to get information back now, and the early things we heard back were that they wanted more information. We started out with the section of our website called the Gene Journal, and this is where we do take these research results and we translate them to our customers -- what does this mean?
What were the SNPs that were found in these genes to be either an increased risk or a decreased risk for whatever that phenotype is? And when they saw this, they wanted more information. And so what we did is we broadened the categories for what information we’re reporting back with a lot of caveats around that where some studies are well designed, they have very large cohorts of people who are enrolled, and they are replicated in other populations.
So those are really the -- what we call the established research. But there’s still a lot of information that comes out in what we term preliminary research, which we put these caveats around it and we have a star rating system to make it very easy to understand for consumers how they should be looking and viewing this information.
And we’re now up to over 78 different Gene Journal articles from 14 in November. And that’s, you know, that seems to be satisfying people. And we’ve overheard people talking where they say, “Oh, that’s just a one-star study,” I -- you know, we’re already hearing that they’re starting to --
MS. DYSON: (Inaudible).
MS. AVEY: -- take this information in and discriminate based on how we’ve been able to categorize it for them.
MS. DYSON: So do you have any sense of how much people use it for the medical side and how much for the, like, the fun part -- your ancestry, seeing how you’re related to your siblings. That may change over time as more people sign up, more family members, but can you talk about that distinction?
MS. AVEY: Well, we just had a very interesting story come up where a woman who was -- she also had breast cancer in her 40’s and she’s been -- she’s a very well- educated, very articulate woman, and she took her information back from 23andMe to her oncologist. And I think she speaks to people at Memorial Sloan-Kettering and a few other clinical centers, but her interest was that she thought she was English, Irish, Methodist from her background, but it turned out her maternal haplogroup, which is information she found on the ancestry side of our tools, indicated that she might have some Jewish ancestry.
And so she wanted to take that information back to see, well, you know, I’d be interested to know, should I have the BRCA test because of, you know, I might have this part of my ancestry. So I think people are seeing this now all in context. It’s a very holistic way to look at your genome, and you can't really separate out the two.
MS. DYSON: Yeah. Well, let’s -- I want to come back to that because I think narcissism is actually underrated as a -- yeah. I see this happening -- I come not just from the health care world, but from a more general world where people are fascinated by the music they like, the travels they take, their financial information, and to some extent, your genome is just another piece of consumer information about how fascinating you are. And I think that’s real, I don't know -- whatever. I’d like to see if Rebecca has any response to what we just said.
MS. FISHER: To the narcissism comment?
MS. DYSON: No, the other thing.
MS. FISHER: I’m sorry. I missed it.
MR. HOLT: I have a mirror for you.
MS. FISHER: I’m for it. You mean, how --
MS. DYSON: No, the other stuff --
MS. FISHER: -- oh, everybody’s --
MS. DYSON: -- not just narcissism, yeah.
MS. FISHER: Oh, well, I wanted to say that I think 23andMe’s information support is really good, and I’ve looked at it and I think it’s a beautiful, beautiful effort. And so I also want to just say, I think the convergence thing that’s going on is really a great thing, and I’m very excited about it. My daughter has BRCA1 also, so it means a lot to us to have this information.
But I guess I just am still stuck on the fact that when I look out there, I don’t see what Matt referred to as, like, an advocate. I don’t see ombudsman, I don’t see that, and I’d like to see that.
MS. DYSON: Yeah. Well, I think -- I mean -- sorry, I’m not supposed to think, I’m the moderator. So let me ask a question. If nobody’s educated, does having more of this information out there, and especially information in the context of individuals, help people get educated so that there will be more advocates in the future? I mean, how otherwise can we foster this education happening?
MS. FISHER: That is an excellent question. And I think that what -- that question actually occurred to me over the weekend as well in slightly different form, but it’s kind of something that came to Africa having 50 countries and 34 of them have more cell phones than landlines. I mean, it’s kind of, like, you know, you don’t have a phone book anymore but you have all this connectivity. So I think what ends up happening is that you have to come at it from both angles and make sure that the information has an understanding under it. So it’s not just lots of this, but it’s a deep understanding. And I keep coming back to this term, legitimate complexity, because people don’t like that, but it’s real. And if we could somehow help people to understand, you know, we have a star system, we have an evidence system, we have a rating system. But guess what? It’s harder than that.
And we just need to somehow get people to understand that.
MS. DYSON: Okay. Let me try an audience question again. How many of you enjoyed studying statistics? Ah, this is not a representative audience. [LAUGTHER]
MR. HOLT: This is (inaudible).
MS. DYSON: How many of you found statistics easier to understand in the context of sports -- baseball averages, whatever? Okay. How many of you found it easier to understand in the context of your own genome?
Okay. Leading question, but anyway, it was a try. Do you --
MR. HOLT: (Inaudible) -- say something?
MS. DYSON: Yeah.
MR. HOLT: So there’s actually a really interesting comment. There’s a group called the Information Therapy Center in D.C., whose job it is is to try to help, or to force, depending which way you look at it -- the promotion of information as a therapy given at the end of each clinical encounter. Same as a prescription is given at the end of many clinical encounters. And they had a conference last year, and they actually had a group of sort of marketing people explaining how you would make information about health care fun and interesting. And I asked the question, which is, okay, if you have to do this at a sort of fourth grade reading level -- write information for health care that because people find it very complex at a fourth grade reading level, how is that, you know, you can do -- the sports pages can have this incredibly complex information about, you know, gun magazines, trucking magazines -- this stuff is written at, like, a, you know, post-graduate reading level and yet people get it. And part of it is interest. And interest in health care, unfortunately, correlates very much to, it matters to me now because I have whatever condition. And part of what’s going on in general in health care, especially with the evolution of the sort of the social networking and elsewhere, is that we’re seeing, you know, people helping each other through that explanation when something happens that matters because they typically have to make a decision.
I’ve just gone through this in my own household, trying to find a surgeon who could do a particular type of surgery, and there’s really very little good information out there. And I think it’s a two-step process. One is that we have to put out more and better information and more and better raw data, which means that data somehow has to be collected. And there’s only two ways it’ll be, sort of, forced out of the health care system; one is by regulation or one is by, sort of, consumer and payer demand. And both of those have been slow, but they’re both coming.
And the second thing is that once that’s out there, we’re going to see these advocates emerge. Now, at the moment they’re doing it kind of ad hoc, online, unpaid. If you look at the ACOR, which I’m sure, Rebecca, you’ve been involved in this. Which is the online American Cancer Online Resources -- did I get that right?
Which is, you know, a million-and-a-half emails sent out each month with people informing each other about cancer and all different types of cancer treatment. To me, that is, you know, unpaid advocacy. And what we haven’t yet had is the thing that we’ve had in financial services where, you know, there’s now Charles Schwab, you have people you can talk to who will help make, you know, the mumbo jumbo of the stock market explainable to you. And I think that’s going to happen, and if, you know, if the health care professions don’t start getting involved in that in a big way, Fidelity or Charles Schwab or somebody else will do it for them.
MS. DYSON: I just read a piece in the New York Times about some minors somewhere who were suing somebody for Morgan Stanley for giving them bad financial advice.
MR. HOLT: Look, no one’s going to say that all these advocates are going to get it right, or that there aren’t going to be self-interested, but that already happens now. I mean, let’s be honest about. We understand there is (inaudible) practice variation in most different types of medical care at the moment across the U.S., if not more. And, you know, it’s quite obvious that there’s self-interest going on there.
MS. DYSON: So maybe if you can go online and get a second opinion that’ll help?
MR. HOLT: That would be a very good start.
MS. DYSON: Okay. Linda.
MS. AVEY: Well, I think it’s -- this is one of the things that we are excited about is using the web to present very complex information because you can do it in layers and you can start out with, you know, kind of a ranking system that gives people, kind of, the first pass at the importance or the weight they should take that information. But then, what we’ve tried to do -- and we’re just at the beginning of this and we’re developing and hopefully improving our product every month that we have a new release -- but is to just build in these layers where if somebody wants to get down to the SNP level, the rs numbers that are part of a gene that were discovered in a paper, we even give the references to all the papers, it’s all there. But it’s just that we don’t necessarily want to confront everybody with it right up front, so having this layering system we think is proving to be a good model. And it’s something that if you put out the cookie crumbs for people, they will follow it to the level that they’re comfortable, but they don’t have to at the same time. So it’s really, really hard what we’re doing.
I’m sure Mari would say the same thing. And the folks from deCODE, that this is highly complex information, but, you know, just like the baseball statistics and everything else, we think people once they get familiar with it they’re going to be more comfortable with the information and they will start diving down deeper and deeper into it.
So we’re actually very excited and think it’s a huge opportunity to educate everyone and bring up the whole playing field so that we’re all ready for this day when we all hopefully have access to our genomes, whether it’s 5 years from now or 20 years from now, and we can take that information into our doctor and they’ll know what to do with that. But we can't sit and wait for that to happen.
If we wait for the medical community to be educated, you know, the Scripps Med School is one of the first, but I will be very curious to see how long it takes the other med schools to step up and decide, this is really important for our futures. And we, you know, we just don’t want to wait. And so this is one opportunity -- we think 23andMe is completely optional. You -- this is people signing up who are really interested in this information, and it is about you. It’s about you and your genome, and it is narcissistic in a lot of ways, but we’re human beings, we’re selfish creatures. That’s the way we operate. And we’re very selfish about our families.
We’ve talked to some people that, you know, initially when we were first starting the company whether or not they were interested and they said, “No way. I’m healthy. I’m fine.” And one of those guys had a son who was diagnosed with autism, and he came around full circle and said,“Sign me up. Sign up everybody in my family. Anything we can do, we are interested in participating.” So things change for people when there’s a change in their health, and they suddenly want more information.
Look at Michael J. Fox who, you know, turned is whole life around and created his foundation which is doing some amazing work. So we see that all the time, and it’s just -- it depends on where you catch someone in their life.
MS. DYSON: So you start off as a benefactor and become a beneficiary later. Let me -- I have one more question right now, but then I’d like to encourage you to raise your hands and the microphone people will show up.
So this question is kind of an essay question to a yes or no -- an essay response to a yes or no question. You take a person, they’re slightly overweight, they don’t exercise enough, they don’t get enough sleep, they drink too much, they’re your sort of typical person who knows --
MR. HOLT: You’re kidding me. [LAUGHTER]
MS. DYSON: -- who knows they should be behaving better. So now they go online and they get the results back. Maybe they don’t have a higher risk, maybe they have a lower risk -- do they -- how do they react? Does it make it easier for them to “behave better”? Do they say, “Oh, I’m at risk, I’m going to behave better,” or do they say, “Oh, I’m at risk, I guess I’ll stop even trying” if the risk was low. Can you just -- how do people actually respond? And anybody - -I mean, I’m sure you don’t have total data, but I’d like to hear how you think -- what the dynamics are.
MS. AVEY: Well, for us, it’s still early. You know, we just launched last November, and we are having a user gathering Tuesday night -- tomorrow night, which unfortunately, I’m going to miss. But we really do want to start gathering that data. We really do want to ask people, “What are you doing with this information?” and hopefully we’ll start learning that. And that will really help us shape our tools going forward of how can we make sure people are using this information properly, that they’re not over-using it, but that it’s also informative to them in ways hopefully that they can positively impact their lives. But we have heard, you know, one case where a guy who’s in his 30’s found out he had really high risk, you know, the highest risk that we can see with our SNPs that we have for Type 2 diabetes -- very healthy, fit, great shape, and found out through his wife going in -- because she was pregnant to be tested for gestational diabetes -- that he thought, “Oh, I’ll prick my finger, too.” And he found out his blood sugar levels were higher than hers, so for him it was a huge wake-up call that, you know, he just had no idea. And then he now is watching -- he works at Google, so he has to really watch the free food and, you know, has to be really careful about his intake. So it’s something that we’re hoping that, you know, as we can really stress the preventive measures that are positive things that people can do, certainly talking to their doctors about that. But, you know, like everyone says, we all know all the things we’re supposed to be doing. But when you see that you do have a bit of an increased genetic risk for something but you can do something with your environment, I think that empowers people even more.
MR. HOLT: Okay. You (inaudible) Esther, because that’s exactly what happened to me and that’s exactly what, you know, my situation when I had my genome tested. And I don’t know what to do because, yeah, I need to go to the gym more and I need to drink less and eat less, and the problem is I also have the life I have which involves, you know, I’d have to make some changes and -- better than I was ten years ago, but -- and this is the situation that most people are in. I mean, we’re probably in that -- what was the category from the Yankelovich, the sort of “Can try harder” --
MS. DYSON: Yeah.
MR. HOLT: You know, could be better, whatever it was. You know, a lot of people are like me in that situation, so it’s part of it. But this is part of a wider -- for most people, this is part of a wider issue, which is to do with, you know, general wellness, general lifestyle, all kinds of things which taken massive changes into behavior change, which we’re very, very bad at doing and there’s no support to help us do that because all the economic and cultural incentives are going in the wrong direction in this country. So, you know, to my mind, for most people, that’s how the direct-to-consumer genomic testing is going to be. It’s going to be, yeah, it kind of helps me, and maybe, you know, I did actually have -- you know, I did it with -- in conjunction with another test where I found I had a high blood sugar rating or whatever. But, you know, I’ve had my labs done recently and I’m basically in the normal range for most things, but I have some evident genomic risks. I don’t -- it’s hard for me to say, “Okay, I should change my life,” because of something absolutely urgent. But I think there’s another category of people -- and obviously, Rebecca, (inaudible) apply to you -- for whom it really does matter because it really is urgent and this stuff is absolutely crucial information about decisions they’re making today or now.
And so I think you have to look at those two categories of people different --
MS. DYSON: Right.
MR. HOLT: -- and then kind of assume that, yeah, fat 44-year-old guys who don’t get out and exercise enough, you know, that’s a more general problem and just knowing the genome isn’t going to solve that problem.
MS. DYSON: So the specificity of the information didn’t change your behavior?
MR. HOLT: No. Because I knew I should have been exercising more and drinking less anyway.
MS. DYSON: Okay. I mean, I personally have found I feel less embarrassed about avoiding fried foods and, you know, taking the fat out --
MR. HOLT: So the reason you were eating fried food was because, you know, you were embarrassed?
MS. DYSON: No. I’m less embarrassed. I still -- I don’t do it, but now I don’t feel embarrassed about taking the skin off the chicken, whatever, because I’m spending so much time with health care people. Rebecca.
MS. FISHER: Well, like Matt was saying, when my family gets sick, we really get sick. So I don’t know what I would think if just a casual finding came back, but I think it kind of speaks to the whole phone book in Africa thing, whereby, you know, the patient is going to be curious. I mean, for lack of a better word, they’re going to be curious so they’re going to agitate for more information and they’re going to bring that to their doctors. And the doctors are going to hopefully learn so that they can do their job better. And I think that’s actually a good thing.
MS. DYSON: Okay.
MS. FISHER: Thanks.
MS. DYSON: So do we have some questions here? Yes? Great. Can the mic people -- if you all raise your hands -- I don’t know how many mics there are, I’m going to try and -- Eric -- the beard over there and then the guy in the aisle. And remember to follow Michael’s instructions. Eric has already been introduced, but --
DR. TOPOL: Thanks very much, Esther. I -- just a few comments. I agree completely with Matt about the vacuum of people to help with patient advocacy. But I wanted to go on a couple point. One is in the diabetes story that you ran through and that Linda mentioned. That is that not only do we know about markers, but we now know different pathways of diabetes and we know if some are particularly sensitive to medicines that exist today that can be used to prevent the diabetes. So we haven’t done those types of studies to use Metformin or ACE inhibitors or (inaudible), so that’s opened up a whole new area is to finding the specific type of pathway that engenders risk of Type 2 diabetes.
And then I wanted to ask Rebecca, because Myriad was one of the early entries into this whole environment, and you would think that this test which costs $3500 or $4000, they would fess up and say, “This is not a classic mutation,” to have had it colored your experience of this intron perhaps private mutation in your family. Was that communicated? Because if it wasn’t, that was really unfortunate.
MS. FISHER: Dr., do you mean did Myriad communicate that it was on an intron?
DR. TOPOL: Yes.
MS. FISHER: Yes.
DR. TOPOL: And that it wasn’t a classic, prior --
MS. FISHER: Yes.
DR. TOPOL: -- described --
MS. FISHER: Yeah. I probably didn’t communicate it very well, but what happened was that the university setting was testing the mRNA. When it went to Myriad, when everybody got fed up with waiting, and it came back -- having paid the money -- they did disclose that. And they are the ones that told us that this had occurred. What’s interesting there -- and I’ll make this very brief, but Dr. Barbara Weber is a good friend of mine, and she was at Penn at the time. Her lab is the one that was testing the mRNA, not the genomic DNA. She felt that that was such an important aspect of the testing, that had a patient outcome, she brought me back to her med students for four years running to tell them story. So I don’t think she would mind my sharing that with you today.
MS. DYSON: Okay. The gentleman in the aisle and then the gentleman with the beard over there. Great.
DR. LICINIO: Hi. I’m Julio Licinio, I’m Chairman of Psychiatry at the University of Miami, and I’m also editor of two journals on Molecular Psychiatry and Pharmacogenomics Journal. So Molecular Psychiatry, which I started 13 years ago, I was just doing the back of the envelope numbers here, would probably publish, like, you know, 1500 papers in these 13 years. And I go over each one of them and (inaudible) the ones that are not accepted, so I probably went over 5000 papers in psychiatric -- most of them in psychiatric genetics, and there is a lot of, like, non-replication and things come now and then they’re not there and the (inaudible) now is this and then it’s that, and the relative risk, you know, is 2 percent, 10 percent, 20 percent, and varies from paper to paper. Then another one doesn’t find it and the field just goes, and that’s how we proceed because, you know, there’s always a new report proving or disproving or, you know, non-confirming or confirming something.
So my question is that even though the idea is very attractive, the issue of clinical validation, I find, is very troublesome, at least in some fields. I know that if you have the monogenetic gene if you have the, you know, breast cancer or something like that, but for common complex diseases, what comes out in research does not necessarily apply to a real life clinic. So I’d like to tell you just briefly, Linda, that I went to 23andMe to the site, I am the most technologically, you know, addicted person. I live in the internet, I do everything virtual, I go for everything new so I filled in everything, you know, pulled the (inaudible) in front. At the very last moment, you know, confirm this -- I didn’t confirm. I quit, which was the very first time I think in my life that I quit something that’s technologically based.
And my thinking was this: my family risk is heart disease, so everybody in my family has heart disease, people diet in their 40’s and have a little bit of some atypical pain and it’s a horrible heart attack and they die like flies. So anyway -- so my thinking -- and correct me if I’m wrong, is this: if I have a genetic predisposition, I am going to become more neurotic and I should lose weight and have a better life and exercise, et cetera. If I don’t have the risk, should I just be lazy and fat? So, which I don’t think I should. So I didn’t take the test and I dropped 14 pounds and I exercise very regularly, so I actually thank you for it, you know, for the service which I benefited from without being tested. [LAUGHTER]
And then my other concern is this. Are you 100 percent certain, you know, mathematically, you know, absolutely, you know, convinced that the data will not be hacked, stolen, passed on to somebody else, or, inadvertently, you know, gotten by some third-party? And that was another factor for me, so I thought, you know, it’s not really going to change my life because for me specifically, I don’t have any monogenetic disease -- we don’t have in the family (inaudible) complex, you know.
So for those, I have to do what I have to do anyway whether I have the risk or not.
MS. AVEY: Great.
DR. LICINIO: And then I have a potential problem with the privacy.
MS. DYSON: So, 100 percent? That was a question.
MS. AVEY: Well, first of all, I mean, just the -- you know, It’s interesting to hear that you went through the whole process and ended up not signing up. And that’s something that we like to hear, that people do go through and they really think about it . And if you decide at the end that it’s not for you, then you absolutely should not do it. So that’s -- it’s good to hear that people do come to that conclusion because we do say that this is not for everyone. And so one other thing that’s been interesting watching my father -- my -- on the male side of my family, the men also die like flies. And my dad is turning 79 at the end of this year, and he’s frankly shocked he’s still alive. He thinks he’s going to drop dead of a heart attack every single day. So it, you know, it’s different for every single person. And I think this is just an option that people have, who really are curious and do want the information that this is -- that we make this available to them. So, you know, I can't argue with your decision. And if it was helpful, I’m glad you went through the process, but --
MR. HOLT: I do think you should send him the $900 if you got fit anyway.
MS. AVEY: Yeah.
MS. DYSON: $999.
MS. AVEY: Or donate it to a charitable cause. But -- and then just on the security of the data, you know, we put so many measures into place. And I think the banking industry has done a phenomenal job of really developing online tools that people have gotten comfortable with. You know, when we first came out with websites to buy things online I think people were very afraid to spend to money, but they’re, you know, “I’m going to put my credit card online?” and what we notice is that people question new things. But if you look back at the old way of doing things, it’s just as, you know, there are just as many issues. If you let someone walk away with your credit card in a restaurant, who knows what they could go buy? So it’s something that, you know, I think we really are very concerned about that and we look to other industries that have already played in this space and have developed a lot of the technologies and we -- you know, we -- that’s first and foremost for us is the maintaining the privacy and the security of our customers data. But that said, what we’re also finding is that because we allow people to share certain portions of their genome -- we have two different levels of sharing, either more the modest and the basic versus a little bit more extended sharing -- almost -- it seems like we’re, you know, a lot of people are opting into that. So it does seem like the minute people get their genetic information -- and we find this within 23andMe, that the minute a new paper comes out, we’re all running around the office, “What do you have? Here’s what I have.” And people want to know, you know, what do you have, what are your risks for something, and it’s -- I think it’s going to become more the common vernacular, that people are going to start talking about this.
MS. DYSON: Yeah. Okay.
MR. HOLT: Can I just jump on that privacy --
MS. DYSON: Sure. Yeah.
MR. HOLT: -- thing for a second because that’s really important to realize, that there is a big divide amongst consumers about this. And there’s another company which is not in the genetics space, but (inaudible) patients like me -- MS. DYSON: Yeah.
MR. HOLT: -- which has -- which many of you may be aware, which is the social network for people with very severe chronic conditions like Parkinson’s and ALS, and they’re very explicit there. When you sign up for this site, you are going to be giving to other people in that community but basically anybody can join, incredibly detailed information about incredibly personal parts of your life when you have that disease. And yet they’ve found that people have found it so valuable that they’re sharing all kinds of (inaudible) about themselves. And it comes back to the core problem, what if this data got out?
Because, you know, banks do get robbed, sites do get hacked, data does get left around, even though, you know, we know that happens -- what is the possible consequence of this data getting out? And I think the main issue here is most people in this country are mostly concerned about their future ability to get access to health care and access to health insurance. And that’s a separate problem which we need to fix anyway.
MS. DYSON: Yeah. Then I must say, I was really disappointed. I -- my COBRA ran out last month, so I went through the process of getting personal individual insurance. And I asked these guys, “Would you like a copy of genome?” And none of them wanted it.
MR. HOLT: None of them have a clue what to do with it. They (inaudible) --
MS. DYSON: So --
MS. AVEY: On the flip side, I just want to throw something else in because the -- it seems that the government and the NIH’s answer to, you know, being -- full disclosure and being transparent, is putting a lot of genetic profiles on the web. And dbGaP is a place now that’s going to be collecting all of these bits of information on many, many people. And to me, what seems to get lost is who are the people whose genomes are being put out on the web. And if you talk to people like Neil Risch and others that are statistical geneticists, they will say, “With about two SNPs and a little bit of phenotypic information, I can identify that person and suddenly I have their entire genome.” So this answer that we’re putting out de-identified information -- you can't de-identify --
MS. DYSON: Yeah.
MS. AVEY: -- genetic information. So, you know, I think it’s more important that the consumer maintains the control of their information. If they want to share it, it’s completely up to them, but to have this answer that it’s going to be -- that you sign up for a study, you kind of sign away your life and your genome goes up on the web, we just don’t know that that’s a viable option.
MS. DYSON: Over here. And then the --
UNKNOWN: Yeah. I’d like to respond to that.
MS. DYSON: -- purple shirt.
MR. PODOLSKY: Doug Podolsky, Consumer Reports. Linda, have you found that your customers want genetic counseling, and do you offer one-on-one genetic counseling?
MS. AVEY: So, so far, again, it’s really early in the process. We haven’t had any direct requests for genetic counseling, per sé. Some of the questions have come up -- interestingly, most of them have come along the lines of the genealogy side, where, you know, people have gotten their haplogroup assignment and are really surprised by the information and they find that very interesting and compelling. And they may have done another service where they got a little bit -- not quite the same information because our mito -- especially for the mitochondrial markers, we study more than just the ones in the hyper-variable regions. So sometimes people get a little bit different haplogroup assignment. So, so far, we have not gotten a lot of requests for genetic counseling, but that said, we’re wanting to work -- again, we look very broadly at this. We want to do education on a very broad level, and because we’re compiling and aggregating all this information together anyway, we might as well leverage it to produce tools and to have conversations with genetic counselors, physicians, whomever we can have discussions with in a big way. So we’ve had several webcasts with NSGC; anyone who’s interested can sign up and be part of the webcast. Now that we have a demo account, people can sign up without having to pay anything, and through the genomes of the Mendel family, we’ve had some interesting comments on that. Like, the Mendel family are part of your demo account when you set it up and one woman wrote in saying, “I’m related to the Mendel’s,” and she was very excited.
So we had to kind of explain that they’re there for demo purposes, and she’s probably not related to them, but --
MS. DYSON: Maybe she is, they’re real people.
MS. AVEY: Could be. So, you know, I think having that tool available now let’s people sign up, they don’t, you know, they can get access to our tools and see all of the information that we share with our customers, how it’s formatted, how you’re able to look across different generations, compare siblings; there are so many tools that we have for families that we’re finding people are very interested in. So it’s a good question, though, and we’re kind of anticipating how we can work with all of the different groups in the genetic counseling field.
MS. DYSON: And in a 23andMe survey it would be really nice to ask people, have you talked to a doctor beforehand? Now that you got results, will you talk to a doctor? And just do some genuine data collection on that point.
MS. AVEY: Yeah. And we’ve gotten quite a few researchers already who want to write some grants and come get funding to do some work with us, where we’re happy to develop those types of surveys. Exactly.
MR. EVANS: Yeah --
MS. DYSON: The purple shirt was first.
MR. EVANS: Right. So --
MS. DYSON: But you have to say who you are.
MR. EVANS: I’m -- yeah. My name is Jim Evans, I’m a medical geneticist and I’m a naysayer. [LAUGHTER]
I think that the --
MS. DYSON: Great. Nice and clear.
MR. EVANS: -- I think that the emphasis on mass marketing the appeal of individual genomics takes our eye off the real value of this type of endeavor. I think that GWA studies and understanding our SNPs and the association with disease has incredible potential for illuminating disease, for medicine from the public health perspective, for drug targets, et cetera. But I would submit that the slide we saw, for example, of Dr. Topol’s risk as defined by 23andMe, telling him that he has gone from a 42 percent to a 54 percent risk of a coronary artery -- of coronary artery disease, is essentially meaningless information.
And if everyone embraces that information with the same enthusiasm that I hear being advocated, and those individuals who embrace with the same enthusiasm a reduction in their risk from 54 to 42 percent, we’re going to have a lot of people using that as reasons to not exercise, et cetera.
And I think this kind of effort takes the eye off the ball of where the real benefit of genome-wide association studies, SNPs, et cetera, are. I think that before we start marketing it, perhaps we should actually find out -- we’ve put the cart before the horse. We should actually find out if people will respond in the ways that are so, kind of, magically suggested, that they’ll exercise more, that we hear anecdotes when they find out that they’re at increased risk.
MS. DYSON: But let me ask you about myself. Why don’t you think I should be able to do this without --
MR. EVANS: Oh, I think that’s fine. What I think you deserve, though, is I think you deserve a clear explanation and not, kind of, a marketing ploy that this is useful medical information because it really has not been shown to be useful medical information. It’s fine if you want to do it from a recreational standpoint; I’m all for that if you want to spend your money that way. I would argue that, again, finding out that you’ve gone from a 42 to a 54 percent risk of heart disease is essentially meaningless for you. For the population it’s important; for you, it’s meaningless.
MS. DYSON: Yeah. And I don’t really see anyone telling me that that eight point differential is significant.
MR. EVANS: Oh, I think that’s the entire -- I think that’s a huge amount of the appeal that these companies are banking on to get people to send them $1000. There is this real appeal to, this is going to be useful medical information, and I think that it’s rather disingenuous to suggest that, oh, we aren’t really giving you anything that’s medically useful. Of course you’re trying to maintain you’re giving people medically useful information. And I would just debate that there really is substantial meaningful information here medically.
MS. DYSON: Have you read the content of these sites carefully?
MR. EVANS: Oh, very carefully. Yes.
MS. DYSON: Okay. Well, we’ll just have to disagree, and I’ll ask the guy next to you to give his question.
MR. GUTTMACHER: Okay. (Inaudible) although I do agree with everything Jim just said. [LAUGHTER]
I think that you can, if you read the sites carefully -- this is not what I actually -- if you read --
UNKNOWN: Speaking off microphone.
MR. GUTTMACHER: Oh, excuse me. I’m Alan Guttmacher, Deputy Director of the National Human Genome Research Institute at the NIH. If you read the sites carefully, it’s extremely well worded. If you walk away from the sites with a general impression, it may not always match exactly what the wording is. But what I’ve actually asked for the mic for is just to comment on something that Linda said about Dr. Risch’s access to dbGaP. Of course, that is a limited access database. He would have to show his -- what his (inaudible) research use of it was before he was afforded on that information, and he would have a users agreement before he did that, which amongst other requirements, would require that he said he was not going to use it try to identify individuals. If he did that and the federal government were of his doing that, then we would take a number of steps to follow up on his misuse of such information. Just to give some -- does that mean it can't be done? Of course not. But it would violate research ethics, et cetera, just as other violations of research ethics, it would be fought up to -- with quite fully.
MS. AVEY: And I’m just curious if the -- when the people signed up to be part of the studies, if they knew that they’re information would be accessible?
MR. GUTTMACHER: Well, if the informed consent process for the studies was not appropriate for its use in this way, then in fact it is not placed on dbGaP. That’s something we look -- we look at all of the studies which apply to be listed on dbGaP, and we’ve rejected a number because the consent was not appropriate.
MS. AVEY: That’s great.
MS. DYSON: The waving hand right in the middle of the room there. Thank you. No, no -- actually, yeah. Right there.
DR. KHOURY: My name is Muin Khoury, I’m the Director of the National Office of Public Health Genomics at CDC, and I’m one of the naysayers according Dr. Topol’s slides.
Actually, the word naysayer is more like what Rebecca was talking about, this sort of, being careful, proceeding with caution type person. And want to echo a little bit what Jim Evans said. And I don’t have any problem with people spending $1000 or $2500, I mean, we buy a lot of useless equipment all the time anyway. But in this case, genomics can really make, sort of, the next 10 to 20 years very exciting if we do it the right way.
There is a lot of discoveries being happening, and the value of the information that’s currently out there is not there yet. And I have to echo Jim and Alan Guttmacher and others, and the reason why I say that is because the -- from three fronts, just want to summarize briefly what I said in that New England Journal of Medicine paper. We don’t know if the information we get from one company is the same we get from another company. We don’t have a good handle on the oversight and, sort of, the analytic performance of these essays. Because of the changing in the literature, if you tell me today my risk of heart disease goes from 42 percent to 51 percent, tomorrow you might say the reverse based on the next paper that’s published.
More importantly, we have really no clue as to whether this information provides additional value to your existing risk factors for that disease. As a matter of fact, from all we know, I mean, I’ve seen the Type 2 diabetes literature, the heart disease literature, prostate cancer, and all these wonderful papers that Dr. Topol was mentioning earlier -- if you do a good analysis of the area under the curve, there is no more prediction to be had for all of these diseases on top of what you already know, which is your family history, your age, sometimes race and ethnicity, sometimes traditional risk factors. I mean, we know that from the Framingham risk factor profiled for cardiovascular disease.
I have no problem with people spending money, but people have to exercise, eat well, and do the right things from a public health perspective -- work, and reduce the burden of disease at the population level. And whether or not your additional 1½ or 2 percent is going to make or break, you know, that, has to be researched, and I sort of applaud the effort to do more research to figure out the impact of this information. But whether consumers should pay for that while research is being done, I have a problem with that because research by definition means -- I’ve been cut off. [LAUGHTER]
Thank you.
MR. HOLT: We’re out of time, but I’ll just say quickly, I mean, it seems to me that you’re kind of in a sensible place, which is that, yeah, it’s a question of who pays for this, right? Because there’s a lot of stuff that comes out of the health care system in general. When I say stuff, I mean both diagnostic tests, procedures, and who knows what, which is a very limited or debatable value. And, you know, we know this from Joe Winberg’s (phonetic) work at Dharma (phonetic) for over the last 40 years. So the question is, you know, which side of the line is this NIH funded research studies -- is this like the rest of the world where we have private enterprise, you know, using consumers or not using consumers, funding research (inaudible) whoever bought, you know, many information technology products. If you bought a Windows product within the last 20 years you probably actually, you know, are a consumer paying for research. You know, it seems to me that -- it’s a question of who funds this and at what point does this become part of the general medical mainstream? That’s the question. This is going to really explode when Medicare and insurance companies decide that, you know, paying for one of these $2000 genetic tests is going to be the way to go and it’s some natural thing that gets done as part of the general medical procedure process. And that happens with many different technologies and many different types of activity in health care when their clear value has been assessed. So it seems to me that’s the dividing line of the question, not whether or not, you know, it should be paid for by consumers or private industry or NIH. It’s question is when does it become part of the general mainstream that, you know, the whatever society it is recommends that 50 or 30 or 20 or 0 years of age you get this -- you get your SNPs done. And it seems to me, that’s the real dividing line questions because that’s when we’re going to start spending real money and making our friends here very rich or not.
MS. DYSON: You must have a response to this, Linda.
MS. AVEY: Yeah. Just really quickly. I think Eric wants to say something too. But I, you know, I just -- I feel like we always do these research studies, and I’ve been looking at these and working with people for over 20 years who do this kind of research, and I think that there’s time and it’s an opportunity now to do it a little differently and to try something new because we’ve been doing the same thing for a long time. And this is why whenever somebody tries something new, that a lot of naysayers pop up and say, you know, let’s question this.
Which we’re very open to the questions and we welcome the debate because we want to do this well, we want to do it right, we want this to be meaningful for people. We’re not just doing this to make a buck; believe me, that’s not our goal whatsoever. We’re here to make a difference.
Individuals seem to want to participate. When you talk to people who have been sick, who have had cancer, who feel like they can now participate in something that might be meaningful, that they could be -- you know, that they could have an active role. And the traditional research paradigm, unlike things like the Framingham study which are more unusual and atypical, we don’t have a real way of tracking people prospectively. And being able to develop a long-term relationship with them and find out, when did you get the disease, when did you take the drugs, track all that information in a very concise and centralized, standardized way. Most epidemiologists would love that, so we’re -- we just want to create a mechanism to enable that, and then we’ll work with the researchers and the experts in the field and say, “Here we are, we’ve got x number of people in our database who are willing to share information; what would you like to ask them?”
So it’s a new twist, and we knew we were going to get arrows in the back. We’re still going to get arrows in the back, but we’re going to do it.
MS. DYSON: Okay. As I said, benefactor today, beneficiary tomorrow. I’ve been asked to read as the final question a question from Kenneth Offit of the Memorial Sloan-Kettering Cancer Center; he’s Chief of the Clinical Genetic Service. And if you want to put this into the record, you might. But let me just -- it’s a bit too long to read, I’m just going to end with the conclusion which is really the conclusion question for the panel.
Is this -- and trying -- this is your chance to summarize, say something witty, you know, whatever.
“I would ask the panel” he says, “is this the time for,” and I guess this “either caution, consumer and provider education, and not-for-profit marketing of research data?” I’m sorry.
UNKNOWN: (Inaudible).
MS. DYSON: I’m just trying to read this thing. I think, basically, it’s the -- I can't really see whether this means not-for-profit marketing or not-for-profit marketing of research --
MR. HOLT: (Inaudible) -- is it a time (inaudible) --
UNKNOWN: (Inaudible).
MR. HOLT: Not for profit marketing. Right.
MS. DYSON. Yeah. Whatever. You can answer it whichever way you want, so, Rebecca.
MS. FISHER: Don’t look at me. (Inaudible) give it to Matt --
MS. DYSON: Should --
MS. FISHER: I guess I --
MS. DYSON: I think there’s a question whether it’s proper to bring profits into it. And --
MR. HOLT: I mean, great, good luck, welcome to America. I mean, what part of the health care system does not have for-profit marketing in it? And that includes, by the way, almost everyone in the non-profit sector of the health care business. I mean, you know, great idea -- Memorial Sloan-Kettering. You’ve seen that building, I mean, come on. [LAUGHTER]
That’s not how this country works. I mean, you know, fantastic in other places, but, you know, and we need to have naysayers, we need to have debate, we need to have sort of people shining bright lights at this -- as they should the rest of the health care system as to exactly what’s going on and where the money flows, and, you know, whether it’s doing good or not. Exactly. But to say that people shouldn’t do for-profit businesses in this is ridiculous given everything else that happens in health care and the rest of society.
MS. DYSON: Thank you. That was clear.
Rebecca, anything else?
MS. FISHER: I’m still not sure that I understand the question. But, in general, I agree with Matt. I think, you know, free enterprise is -- has made us a really great country and we should continue with that paradigm. We just need to do it carefully.
MS. DYSON: Thank you. Linda?
MS. AVEY: Yeah, I’ll voice the same thing. That we, you know, we really think at the end of the day what will make 23andMe a successful company is having a really great user interface where we make this information really clear for people. We hope the costs continue to drop, which we think they will. It’s the -- historically, if you look at the cost of genotyping over the last ten years, which was shown, it’s dropping tremendously. And so we really think the value of this is having a lot of people engaged and willing to share information. And as long as they’re willing to do that, we think there is a way to do this. And if you try to do this in a not-for- profit way, which we talked about when we first started the company, can we either split out a not-for-profit side of 23andMe or do something a little differently, and the problem is that when you’re running a not-for-profit, it’s really hard to hire really good engineers, it’s really hard to build a really strong team to build what you need to get people to want to participate in the first place.
So you’re kind of between a rock and a hard place. So we felt like we can be a company that does good and does well, and that’s really our mission. And, you know, we’re going to be voicing that more and more and wanting to do our own studies that hopefully we will be able to do some funding and as -- hopefully we’re successful. So we’re sensitive to that problem, but we’re -- you know, we think it’s free enterprise; it’s America.
MS. DYSON: Let me thank the panel for being a great panel. I think we need to move forward with free enterprise, free consumers -- all in the context of having more panels like this so that people understand what they’re doing and what the implications are. Thank you very much. And thank the audience. [APPLAUSE]
DR. COWAN: Thanks to the panel. We’ll have a break now. we have a 15 minute break; that’ll bring us back at five till 3:00, please. If you can do that; I know 15 minutes is short. [BREAK]
Panel 2: is the Testing Process Reliable, and is the Information?s Privacy Maintained?
Dr. Reed Tuckson is going to chair the panel. This panel will be addressing -- where’d I go, lost my -- addressing issues on genetics, health, and society. Dr. Tuckson chaired the Secretary’s Advisory Committee on Genetics, Health, and Society. So without expanding anymore, I’ll let him take over, introduce his panel, and we’ll get started.
DR. TUCKSON: Thank you. Good, good. Good afternoon. Good afternoon.
AUDIENCE: Good afternoon.
DR. TUCKSON: Now, you’re all going to wake up one way or the other, so we’re just not having -- and if we could the people in the back to come on in because I’m not going to have my first panelists talk to confusion.
Now, we’re going to change the order a little bit because we decided that we wanted to. And so we can do that. There are two issues really before us in this section. And again, just to orient you -- is the testing process reliable and is the information’s privacy maintained? And so I’m going to break those into two distinct sections. And we’re going to start with this question of is the testing process reliable? Well, this has been, as all of you as astute observers know, a fundamental issue in this field for many years. I think most of you are familiar with the work of something called the Secretary’s Advisory Committee for Genetic Testing, which was formed several years ago. And that Advisory Committee’s whole function was to try to get at this question of the adequacy of the oversight of genetic tests. And that is work that continues forward to this day.
The question then becomes is, is it in fact true that genetic tests are reliable? And one of the good things that is occurring in this area to give us a better sense of it and to give us greater assurances, is the new Secretary Advisory Committee on Genetics, Health, and Society. The audience being extremely populated by many of those good people and former colleagues of mine, I see.
They have put forward an important report to the Secretary, which is now being analyzed by the Secretary’s Office. I will tell you that that report does raise some important issues about the adequacy and the reliability of the oversight of genetic tests. In fact, I see government holding up a copy of it right now. Government is in the room. And so the real issue is that there are issues here, and so without going further into it, we have some perspectives. Question related number two is, do consumers really care if it’s reliable? Do consumers really have a position on this? Don’t most people just say, “Well, of course the government has taken care of all of this.” Don’t most people say, “I don’t know any difference between genetic tests and all the other tests, I just assume it’s a holistic -- big hole -- and somebody is taking care of it.” So the question becomes, do consumers really care if it’s reliable, or do they just expect it.
And then finally, do consumers actually perceive that there is a problem? Whether they care about it or not, do they think there is a problem? And if they think that there is a problem, do they perceive it in a way that is determinant? Does their perception of reliability cause them -- or their perception of non-reliability or uncertainty -- cause them to act or not act in a certain way?
And so those are some of the questions that would logically derive from our organizers asking, is the testing process reliable?
Let me then switch to the second half of their challenge to us. Is the information’s privacy maintained?
Well, a big contextual issue here is of course whether or not you will be discriminated about because of the information. We are all, I think, celebratory of the GINA Bill, and that was a long-fought effort by a lot of people, many of whom are in this room today. And so at least that starts to give us some sense as we frame this conversation around protection of misuse of the information. Still, is privacy maintained in fact, and is it maintained in a way that is more or less stringent than in other areas of medicine? We come back to this question of genetic exceptionalism; is the privacy of information in genetics more or less maintained than in other areas of medicine. And secondly, is this an issue of concern for consumers, and is their concern determinant. Do people worry about this privacy of information, do we have any sense that the GINA Bill has taken away the concern around misuse, and now it’s a question of essential, just privacy for its own sake. And is that concern determinant? Does it result, or will it result, for example, in an unavailability of this information for coordination of care and disease management resources.
I think most of you in the audience are aware that today’s health care system with chronically ill people -- the health care system is organizing itself to be able to use data and information to help navigate people through a fragmented care delivery system, helping to get people to the full array of the services -- the comprehensive services that may be associated with their clinical condition. Will concern for privacy cause the unavailability of that information to be used for these critical purposes and have an interesting inadvertent result? And that being, that people with chronic disease, people with complex illnesses who need lots of care coordination, won't be able to get it because of people’s concern around privacy, thereby not making the information available, and then compromising health status.
Will people’s concern about privacy result in the unavailability of this information to share with family? And so what will it do to family dynamics at Thanksgiving dinner? And will it mean that there will be some people at dinner who will hope that certain people at dinner, like the moderator, will be quiet and just not talk about things. Will there be some family members who will notice that other family members have gotten prophylactic surgery, and thereby will have information that they wished that they had not had. And what does that do to the dynamics of family life? And finally, will this concern cause a chilling effect on public health surveillance and population-based prevention and research? And so the question is, ultimately, do anxieties have determinant outcomes in this field?
Well, with that as a table setting, let me turn to our speakers. Do not be distracted by the agenda on your program because it’s wrong. Our first speaker is going to be Jeffrey Gulcher, who is the Chief Scientific Officer for deCODE genetics. Not only is he here because he’s one of the founders of deCODE, but he’s also here because his colleague is stuck in Switzerland.
DR. GULCHER: Iceland.
DR. TUCKSON: Same thing. [LAUGHTER]
DR. GULCHER: Not if you’ve been to Iceland, sir.
DR. TUCKSON: A long way away is the point. So we’re very pleased, though, that Jeff is here. And Jeff is going to really focus in a bit on this issue of reliability of clinical tests. And we’re very happy that you are here, Dr. Jeffrey Gulcher.
DR. GULCHER: Thank you. I just want to point out that we’ve spent a lot of time thinking about genetic risk test because we’re also a diagnostics company and we make available through our reference laboratory, laboratory-derived tests for genetic risk for individual diseases in addition to deCODEme, which sums up those diseases and adds some additional diseases that we have not yet developed tests for, and offered it as an individual set of tests. But when it comes to reliability, it’s really important to emphasize that the genetic risk tests that we’re all putting together are risk factors, they’re risk markers. They are not pathoneumonic for a disease, so therefore they’re not really a true diagnostic from that point of view. They’re certainly not a determinative test either from a genetics point of view because this is not like the Huntington’s disease gene, that if you are positive for that single gene for Huntington’s, you will get Huntington’s no matter what you do. Conversely, if you don’t have a mutation in that gene, you will not get Huntington’s disease no matter what you do. For the common diseases it’s an interplay between genetics and the environment, and no single gene is going to determine absolutely whether or not you’re going to develop a heart attack or a stroke. So really these tests, when we put these tests together, and in some cases these are single or two-marker tests, and other cases they are eight-marker tests. For example, in our prostate cancer test, eight markers together define risks compared to the general population of developing prostate cancer, anywhere from .4-fold up to 7-fold. So for a patient who has a high risk for prostate cancer, they’re not going to be told that you’re definitely going to develop prostate cancer. And for somebody who has a lower risk based on a genetic profile, that patient is not going to be told that you are immune from prostate cancer, and therefore you should not get PSA testing, for example, as a screen.
Just as physicians -- Dr. Topol will tell his patient who has the upper quartile, quintile of LDL cholesterol, he’s not going to tell the patient, “You are definitely going to develop a stroke or an MI,” right?
“But we need manage that risk factor for you.” And the patients that he has -- or the lower quintile of LDL cholesterol, he’s not going to tell them, “Let’s not pursue any other risk factors or manage your other risk factors” because the number one cause of death in patients with a lower quintile of LDL cholesterol is still MI and stroke. Right? So as physicians, we know how to deal with risk factors, we know how to manage them low-risk or high-risk; the key is to be able to put those together in the context of other risk factors and use them to prioritize patients to those who deserve maybe more attention when it comes to earlier diagnosis of cancer, or to motivate them to change their lifestyles or manage those other risk factors (inaudible). If indeed the information does add new information that’s not already being assessed, and Dr. Khoury would suggest that maybe some of this information is redundant with what we’re already capturing today with either family history or the other risk factors. And I would contend the important thing to realize is these common genetic risk factors are adding something much beyond family history. They do not account for the vast majority of family history, these are not rare variants of high effect, and if you look at paper after paper, our own discoveries and others, it does not capture family history. So family history alone will not substitute for this genetic profiling.
Conversely, 95 percent of prostate cancer patients do not have a family history of prostate cancer -- of diagnosed prostate cancer. And so you can't rely just on that. If these tests are useful for those who don’t have a family history, it’s adding -- by definition, it’s adding additional information -- risk information beyond family history, so they’re not substitute, although they can be interchanged.
So when it comes to reliability, it’s important to communicate to the patient and to the physician that these are not determinative. So if somebody says, “Is this a reliable test? This is going to predict that I’m going to have a stroke?” No, you can't say that. You can only say that this is a risk ratio compared to the general population risk and there are other risk factors that need to be measured -- environmental risk factors and other things -- and there are many genetic risk factors that we do not know yet. But still this information may in certain cases be useful to act upon through your physician.
Now let’s move back -- so that’s reliability in terms of the interpretation of the information, but then there’s been some suggestions by others that maybe we can't measure the genotypes -- the genetic information, very accurately, or that we can't really tell the FDA or CMS how accurately we do measure. That’s what the so- called analytical validation component of a diagnostic.
The analytical validation for a genetic test -- the reliability of that measurement of that information is much easier to measure yourself or determine it’s accuracy yourself as a laboratory, much easier to demonstrate to the FDA or CMS that you are accurate because genetic information -- it’s pretty cut and dry, at least these single-based changes that Dr. Topol mentioned. Very easy. Sequence-based -- you sequence the genome -- or sequence that one little location in a set of patients, which is considered by the FDA the gold standard for genotyping, and match it with your genotyping platform. And what’s the concordance rate? And the concordance rates I would guarantee for all three of our companies is very, very high indeed -- 99 percent -- 99.9 percent plus. But it’s easy to demonstrate to the regulatory bodies how accurate that is, and to communicate that to our patients. So when we talk about reliability we can measure reliability; much more reliable than demonstrating how reliable can we measure CRP or other -- or even LDL cholesterol or other biomarkers that fluctuate and have interfering substances within the sample that you’re measuring. A lot easier to describe that and document that.
Let’s move on to the clinical validity; that’s the second piece of CMS or FDA when it regulates a diagnostic. The clinical validity -- and if you move to my first slide -- I just want to summarize. The genetic risk tests that we provide are very well clinically validated indeed. If the definition of clinical validity is that you discover them in one population and then you replicate them in multiple populations. That’s the definition. We’re not talking about clinical utility; we’ll get to that later. But clinical validity, does it replicate, does it have the same effect in multiple populations? And so for the markers that we provide, these same set of markers have been replicated in multiple populations. In some cases they’ve only been tested in Caucasian populations; other cases, they’ve been tested in other ethnic populations and been replicated, but the point is, they are clinically validated in the populations that are being claimed.
So when you sum up all of the patient populations that are behind, let’s say the diabetes markers or the prostate cancer markers, you realize that the number of patients and controls together, are in the tens of thousands. In many cases, you have over 10,000 patients behind that. For the MI test, for example, or 5,000 patients versus 30,000 or 40,000 controls. So you have a lot of data behind them -- larger data sets behind these tests than for most FDA approved diagnostics and therapeutics. So they are well validated from the standpoint of replication, and then when it comes to estimation of what that risk really is, we’re not using 200 or 300 patients to estimate what is the true relative risk of this particular genotype in these Caucasian populations, we make use of these full tens of thousands of patients to estimate that relative risk. Right? Just as a clinical trial uses thousands of patients to define what the relative risk reduction is due to a drug, all right, but these are tens of thousands that are estimating this particular risk across populations, and we think that’s a pretty good estimate. To have a higher precision than that, we’d have to 500,000 patients or so. Right. So we think that the clinical validity for many of these tests is already there. And I should also mention that these markers can -- you can demonstrate with these large population sets that they are independent of each other, meaning that they don’t -- they’re not synergistic or redundant with each other. And so therefore you don’t have to come up with complicated models of how to put these eight different prostate cancer markers together to define the risk for that particular patient, you can first convert the odds ratios that we typically report in all of our publications to risk ratios -- relative risk compared to the general population so that you have a standard population by which the risk is compared. And then because there are independent risk factors for prostate cancer, you can simply multiply the genotype specific risks for each of those eight markers together to define the composite genetic risk compared to the general population. This is what physicians have been doing for a century -- multiplying independent risk factors together to define composite risk. So we think that’s a way in which -- think it’s easy for physicians to in general understand how we’re doing this, as long as we’re transparent on how we define clinical validity.
If you go to the next slide, this answers the so what part, which is really important to have in this discussion about analytical and clinical validity because if this stuff -- if this information is not useful in certain circumstances, then why are we even having this discussion? Should we wait until another 50 different genes for Type 2 diabetes have been discovered? Or is this information useful today? If we had waited for the assessment of HDL or some subparticle sizes for LDL -- should we have waited before we measured total cholesterol or even LDL cholesterol, waited for the additional nuances of cardiovascular risk? No. We use the information as we discover it as long as it adds something new; and I would contend that it does indeed add something new. The heart attack variance that Dr. Topol mentioned -- we’re talking about a 1 -- it’s a modest risk, 1.3 to 1.5, depending on the age of onset of risk. But this is an independent risk factor, independent of LDL cholesterol, hypertension, smoking, family history -- risk factors that are routinely measured but this is not routinely measured. It adds something. There is a recent study that showed -- prospective study that showed, yes, there wasn’t much of a change in the AUC for cardiovascular risk, it only went from 62 percent to 64 percent, not significant. But there was a significant re-classification of patients between the low, intermediate, and high-risk categories based on ATP3 criteria, which most physicians use today.
Substantially -- about 15 percent of patients got reclassified. So here’s an example where there is something you can do differently about it; you can change the target level of LDL cholesterol if a patient rises to a different class. Prostate cancer, eight markers that define this risk that I mentioned. Breast cancer, we’re about ready to launch a test for breast cancer -- individual test. Eight markers that -- 5 percent of the general population is at 2-fold risk for breast cancer independent of BRCA1 and BRCA2. This is for more of the late-onset breast cancer, which has a much bigger public health impact than the rare form -- early-onset form of breast cancer. And so it provides another way of assessing risk that compliments BRCA1 and BRCA2 for the different -- for the usual form of breast cancer. Type 2 diabetes, 10 percent of general population -- or pre- diabetics, actually convert at a very high rate to Type 2 diabetes. Fifty to 70 percent absolute risk within three to four years; this is based on the DPP and DPS study, a clinical trial where the genetic markers were added.
And then finally I want to mention before I go to the case study, atrial fibrillation, we discovered markers for atrial fibrillation that we then asked the question, what’s the clinical utility? Applied them to a series of stroke cohorts, and identified that there’s a large portion of patients with cryptogenic stroke that are not being diagnosed with having atrial fibrillation. They go in and out of atrial fibrillation. The public health impact of not making the diagnosis -- proper diagnosis of a fibrillated stroke is immense because anti-platelets do not work very well for prevention of stroke related to AF.
But Warfarin does, it cuts down stroke risk by about 60 to 70 percent. If -- in order to use this test today in the health care system today, we estimate 150,000 patients would be diagnosed with atrial fibrillation related stroke that are not already being diagnosed, and it could save Medicare $1 billion a year if applied in that particular manner. So it can have an impact, but only if you pick certain niches where there is a clinical utility that you can demonstrate.
Next slide. So finally, I just want to give you a case study, which was my own. I have a family history of prostate cancer, but it’s the late-onset version. My father had prostate cancer when he was over 70-years-old, a benign form. The AUA Guidelines would not suggest that I be concerned about earlier onset prostate cancer because my father had such late onset, and the guidelines suggest that if you only have a family history of a father or a brother over the -- of prostate cancer with onset younger than 65, that you consider doing PSA testing at an earlier age than normal. Normally, it’s recommended that you start getting PSA testing at 50; if you are at higher risk, it’s suggested that you get PSA testing at 40.
Since I’m more compulsive, I went ahead and got my PSA tested anyway at 42, and I was completely below normal. Then I got my deCODEme results back when we updated it with the eight markers, and my relative risk was now 1.88 just on the basis of my genetic profile alone. Lifetime risk for a white male is 16 percent, so I’m double that risk. And by the way, there are no other risk factors for white males when it comes to prostate cancer. There’s not some other identifier that can help my physician decide, do I want -- should I test or not? Also, the markers suggest that I had moderately increased risk for aggressive versus non-aggressive prostate cancer. So the high risk prompted my primary care physician to refer me to a -- sorry -- the high risk prompted my primary care physician to go ahead and measure my PSA. I’m only 48- years-old so I normally would not have had my PSA tested at this time. My PSA was high-normal at 2.5; the range is from 0 to 4. Some people use different cutoffs depending on additional risks, like family history. But the high risk prompted my primary care physician to refer me to a urologist. The high risk prompted him to recommend a ultrasound-guided biopsy, which was positive for intermediate grade prostate cancer with about 20 percent of my prostate is filled with cancer. If I had not had this information, my primary care physician probably would not have ordered the PSA, he probably would not have referred this normal range PSA -- high-normal range PSA to a urologist for additional evaluation, and maybe my urologist would not have recommended an ultrasound-guided biopsy. Two weeks ago, I was scanned -- I had a bone scan and I had a normal CT, so it doesn’t look it has spread as far as we know. And then in two weeks I’ll have my prostate taken out with a radical prostatectomy. But here’s an example where this information can indeed be useful, but only in certain circumstances. We’re not suggesting that everybody be screened, but in certain circumstances, this information can interact or work together with already established guidelines.
DR. TUCKSON: Well, thank you very much. And thanks for sharing such a comprehensive range, not only of the technical but the personal, and we very much appreciate that.
When we get to the question period, I’m going to ask you some issues regarding, again, from the consumers perspective, how does the consumer know that the test -- and your test -- do what they say they do? You’ve also opened up the Pandora’s box of the reliability and the interpretation of information which we may get to. But at a very fundamental level, you seem like a nice guy, deCODE seems like a pretty nice company. But again, how does the public know, and is there adequate oversight that says that somebody is checking on you despite the fact that you’re such a lovely person?
Ryan Phelan, founder and CEO of DNA Direct, would you carry this on for us?
MS. PHELAN: I’ll try. Thank you for including me here today. My company, DNA Direct, does a little bit of a different service in the genome-wide arrays that you’ve heard about here today. We actually offer services that we call medical diagnostic tests -- genetic tests that help people make health care and medical decisions.
We’re not the lab; we are genetic experts, we’re comprised of medical geneticists that act as our medical director and guide our clinical protocols, and genetic counselors that interpret and provide information to consumers. I started this company just over four years ago, and so we actually have real on the ground experience talking with consumers, patients, and providers every day.
And I thought what I would do is share with you a little bit about what I’ve learned from our customers. And also, I’m (inaudible) with all these things that I’m thinking about in response to so many of the thoughtful questions raised here today.
Now, I’m going to start with actually something that Rebecca raised, which is, our company does BRCA testing. Now again, we work with Myriad Genetics as our lab, and we help people with that very important decision early on, of whether or not testing is important. I’m going to talk about that a little bit because to me that’s what is involved in, is the testing process reliable? Dr. Gulcher has done a great job talking about the accuracy, the clinical and analytical validity of these tests, which run 99.9 percent molecular diagnostics. But it’s the whole process that I think consumers need greater understanding and awareness of, and in a sense, should actually drive for even a better quality and standards in this industry. So I started the company because I knew people were not getting access to some of the medical genetic tests that I thought were really useful, that medical guidelines were established saying people within a certain protocol with -- where testing would be relevant.
And our company does the same kind of assessment for determining who is appropriate for testing by demonstrating the pros and cons of testing and helping people really make an informed consent. And I believe that that has to be a really important part of any testing process.
So what I have up here on this slide is just a handful of the questions that consumers raise every day. And we know this both from phone calls we’re getting, from emails, and from where people look on the site. So obviously, can I trust this test? Can I trust this company? Will my results be kept private? What is your privacy policy? Will this test actually help me make a better health care decision? Is this test going to be covered by insurance? Will this test give me peace of mind? These are the kinds of questions that consumers have, and that companies have to responsibly provide answers to, with transparency. And I believe that where our industry is going, now that there are even more reputable companies, I believe, coming into the space, is really trying to create some industry guidelines, sort of a best practices. So our company provides full transparency around our policies on our site, and I believe this is going to be an increasing standard that will happen.
You have the next slide? I also think that it’s important here today to talk about this field of genetic testing with a little bit greater distinction. And so I’ve done sort of a sampling of a very crude way of categorizing testing. So on the very bottom, I’ve put down diagnostic testing for very targeted genetic diseases, and I’ve included in that as an example, Huntington disease. And as Jeff mentioned, this is a highly deterministic test, it’s one where people who are carrying the mutation will in fact at some point in their life develop Huntington’s disease. And what I’ve put on the right-hand side are examples of support services that I believe have to be provided in order to offer that testing in a responsible manner. So on the very right- hand side, it says, “in person consultation;” I’m assuming in a physician’s office with health care professionals doing some kind of physical and mental and emotional assessment of this particular patient in order to determine whether or not Huntington’s disease testing would be relevant and useful to them. That’s standard clinical practice, and that’s part of the medical guidelines. But as we move further up the ladder of genetic testing -- and where we’re going today into the consumer world, we’re seeing predictive testing (inaudible) for serious health care conditions, like BRCA.
And probably many of us in this room would debate whether or not BRCA testing needs to be done in a physician’s office face-to-face. Well, the truth is, in major academic centers all over this country, even they are having to often utilize genetic counselors by phone. Some people prefer them -- prefer the phone to a face-to-face, and in addition, it can reach a much greater audience of people with very limited genetic expertise. At DNA Direct we do everything by phone, but we do pre and post-test counseling by phone and by web. So that’s an example of where we’re starting to see a virtual provider actually filling a clinical need. And as we go up the ladder, I’ve got genetic (inaudible) carrier, risk assessment for things like Cystic Fibrosis or for pharmacogenetic testing, for Warfarin, or for Tamoxifen testing. At DNA Direct we do that without a phone consult, per sé, being required but with physician oversight. Those are supposed to be tic boxes by the way, I’ve got to fix that and with web support. And then as we go up that ladder where you see genome-wide testing, I’ve included genome-wide arrays, like some of the companies that we’ve discussed here today -- but also, full gene sequencing. I think that what’s going to happen is there’s going to have to be a different level of support in order to responsibly provide that service. At some point, today we may say that there are, you know, a handful or a dozen tests of SNPs that have clear, clinical implications, but if we fast forward 18 months, 5 years, those tests are going to become more and more predictive and they’re going to have greater and greater weight. And the question is, at what point does that testing require physician involvement, at what point should it require a genetic consult or medical advice, as Rebecca was mentioning -- or a health advocate. At what point are there intermediaries that help some of these consumers: 1) make a decision whether or not testing is going to be helpful and relevant and appropriate to them; and 2) what are they going to do with the information once they get that result, do they have any kind of safety net of people that they can actually to?
DR. TUCKSON: Ryan, before you go on --
MS. PHELAN: Yeah.
DR. TUCKSON: -- before you go on, let me just make sure -- because you mentioned that some of these, you said, should have check --
MS. PHELAN: Yeah. Yeah.
DR. TUCKSON: Are you saying that the --
MS. PHELAN: Those little funny boxes on the right.
DR. TUCKSON: So -- oh. The funny boxes on the right?
MS. PHELAN: The -- those little --
DR. TUCKSON: Okay. So they’re the stars?
MS. PHELAN: The stars were meant to be stars.
DR. TUCKSON: Okay.
MS. PHELAN: That I’m saying are condition dependent.
UNKNOWN: They look like little windows.
MS. PHELAN: And the little windows are --
DR. TUCKSON: Those are checkboxes.
MS. PHELAN: -- or doors were supposed to be checkboxes. Sorry about that.
DR. TUCKSON: Okay. Good. So it’s right in the handout, people have, by the way, over here.
MS. PHELAN: Yeah.
DR. TUCKSON: Good. Keep going.
MS. PHELAN: Okay. So this is kind of a wild leap at -- with really no setup for this. But this is an idea; it’s called DNA Perspectives. It’s a concept that DNA Direct is working on, we’re inviting industry-wide collaboration with non-profits, with academic institutions, and others to actually help consumers identify whether or not a test is going to be useful, responsible, and relevant to them. So this is really a placeholder; we’re starting this around -- just with gathering information from different experts on Alzheimer’s testing. That would be with the APOE gene.
And what you see here is an expert’s rating system. So this would be actually provided -- this information, this score, would be done by a dozen or so medical experts from around the country. Their discussion regarding whether or not they believe the APOE gene has scientific validity, would be completely transparent to anyone who wanted to look on this wiki. And we’re in the process of doing this. So we did a placeholder here in this mockup saying the community could probably agree -- the scientific community -- that APOE gene is highly correlated with the scientific validity for Alzheimer’s. But the predictive value, I’m just -- we’re giving a random 25; it’s probably a lot lower in predicting who will actually ultimately get Alzheimer’s and who will not. And hence, the clinical utility with there being no known therapeutic intervention for Alzheimer’s, would probably be viewed by the scientific and medical community very low. But I show you as an example the personal utility. With a score of 75, if we ask consumers -- and there have been studies called the REVEAL Study that show this -- that consumers would actually say, knowing my predisposition for Alzheimer’s disease would be highly useful to me as a consumer. And what I’m trying to do here is to show that there are going to be services like this, whether or not it’s DNA Perspectives or DNA Perspectives grows and it morphs into something that could be something that the industry actually comes together with, with government and non- profit agencies actually really build an independent ratings system. This is where we have to go because this question about how do you know what one test, one company, one service, one variant -- what’s the real usefulness of it? I think there’s going to be a lot of public debate on this. And I don’t think we can wait and say this all has to be done before anybody does any testing. The testing is happening, information is happening, it’s getting to the consumer. But meanwhile, we need to be able to figure out how can people actually start to look at what experts are saying about this, and then ultimately, how can consumers wade in and provide their own information, their own feedback, on the usefulness of these tests and of the actionability of these tests.
So we’re going to be launching this fall with literally just this one gene variant with our scientific community inviting consumers to participate in this discussion, and I’m really putting this out as a placeholder to people here in this audience who may know of other industry-wide initiatives. People have talked a lot about the need for a ratings system, but I believe that we need to start to make this happen and to see what are the components that are really going to make a difference for the end-user, who is the consumer, the patient, and the provider, I think.
DR. TUCKSON: Well, thank you very much. I’m going to come back and ask you to delve a little bit more -- when we get to the question period -- around those consumers that are on the phone. What are they really saying to you about what level of ease or dis-ease they have about this reliability business and this privacy business. So just know I’m going to come back.
Panel 3: What is Currently Useful to Consumers, and What Can They Expect in the Future?
MS. MCGRAW: Okay. Thank you very much. I wrote down a couple of things in that during the first panel that I thought were really interesting. The one was from the Yankelovich survey data -- people assume medical privacy. I think that’s a really interesting point, and I’ll come back to it in a minute.
I think the other piece that was interesting was that to the extent that we’ve delved at all into privacy and security issues, we kind of went sort of more to towards the security pieces -- the data is secure, people can't hack into it or it’s encrypted or whatever. And we see a distinction between privacy and security, but both are quite important and I’ll go into that in a little bit of detail, too.
And the other thing that I thought was so interesting about the marketing presentation that we got and what the different types of consumers, is just how valuable data that could target marketing and advertising would be. Which, if none of us had a sense about just how valuable that identifiable data about what people might be predisposed to get in the future or even what particular conditions they have, would be to advertisers, you know, there’s certainly good evidence for that. So I don’t think anyone in this room would disagree with the statement that the privacy component is very important, as is the security. And the truth is is that what protections we have are a bit of a mixed bag. There’s some better news today than there was in the past because of the passage of GINA, but what so often is the case is that we are either understating or overstating the amount of protection that we do have. And the protections really are important to think about in two ways. One is, what can people do with the information? This is the privacy piece. What are the permissible uses of health information, whether it’s genetic information or information about health status? The second question is, if you’ve got that information, to what extent can it be used in ways to harm you? And this is what people tend to focus on most; can it be used to discriminate against me?
Can it be used to hurt me in terms of getting health insurance? Can it be used to hurt me in terms of employment? Can my employer fire me or not give me promotions? Et cetera.
The good news about GINA is that at least with respect to health insurance and with respect to employment, you can no longer use a piece of genetic information for discrimination purposes in health insurance and in employment. But we didn’t quite finish the job; we still have some work to do because, number one, if you have the manifestation of the condition for which you have the genetic marker, the information -- that is that you’ve been treated for a certain condition, that you have a chronic condition -- isn’t in fact protected under GINA, and the extent to which a health insurer can use it for underwriting -- sorry, Reed -- underwriting purposes or the extent to which an employer can use it if they are able to obtain it for employment purposes kind of depends. You know, we have the Americans with Disabilities Act on the employment side; there are some protections on the insurance side under HIPPA, some under some state laws, but it’s a very incomplete picture. So while we have taken care of some things with respect to genetic information, we still have the problem that Matt raised, which is that the information once you actually have a condition can often be used in ways to harm you.
Now, getting to the point about HIPPA, that privacy is assumed. It’s so interesting because the point there, I think, is that people often assume that when they are entering their health information on a website or even with respect to the information that their physicians or hospitals have about them, that that information can only be used in certain ways. And typically people really significantly underestimate the extent to which health information can be lawfully used. And the point I’m making more than anything is that I’m the transparency point -- is for consumers to have a much better understanding of what are the permissible uses of their information, and not so that when they’re seeking care, when they’re seeking to get a genetic test, they have an absolutely complete understanding. And I couldn’t agree more with the folks who said earlier that if you’ve met one consumer with respect their privacy concerns, you have met one consumer with respect to their privacy concerns.
There are an awful lot of people for whom -- who are willing to disclose a fair amount of information about themselves in the interest of whether it’s furthering research, whether it’s as part of a social networking site, et cetera. Again, since I’m a privacy advocate, I think that’s nuts. But there are people who will do that, but the policies about what that information even to the extent that it’s disclosed by those folks will and won't be used is really important, and it’s not just a matter of what the legal regime is. I mean, how many -- you know, in terms of even just looking at a privacy policy and understanding what it is that the company that you’re entrusting your information with can and can't do with your data, you know, there’s lots of evidence out there about how people don’t tend to read them and if they read them they don’t understand them. I don't know when the last time was that you signed up for something and, you know, just scrolled through that privacy policy and clicked that box at the end. I’ve done it myself. It’s not the most protective way to do this.
So getting to HIPPA, that is the federal law that we have that governs the uses of information, the privacy protections, the security pieces that need to be in place for covered entities. A lot of the folks who are obtaining medical information now are not currently covered under HIPPA. HIPPA’s coverage is pretty limited - - it’s hospitals, it’s physicians, it’s pharmacists, it’s labs, but it’s not everyone who’s now in this space to protect this information, which then puts the onus on the consumer to be that much more aware of what are the potential uses of this information? Again, because it is so valuable. I think the other thing to keep in mind with HIPPA is that because genetic information by itself without a link to some other piece of information isn’t necessarily identifiable, again, depending on its context.
Identifiable information is also not protected health information under the law. So the bottom line being is that we sort of have a patchwork of protections here, so when the question comes up for consumers, you know, “Will my information be kept private?” The best advice that I contend to give people in this context is, “Well, that depends. Who has it? Who’s holding it? Is it linked to other identifiable information? For example, is it part of your medical record or is it part of a research study where it’s in a great big databank?” So I think we have some work to do in terms of being able to assure people that when they’re getting these tests their information will in fact be kept private and secure, and that to the extent that there will be uses made for it to treat them or to help pay for their care. These are the ways that the data can be used, and these are the ways that the data cannot.
DR. TUCKSON: Very good. Thank you for a very interesting first round. As the audience starts to think about what it wants to ask you about, let me -- as promised, Jeff, let’s go back to this issue of getting beyond your competitors ex who’s not as nearly as nice of guy or company as yours is. How do we -- what is your view as a private-sector person trying to run your business and provide an important service to the American people -- what is your view of the adequacy of oversight that can give a consumer, your momma out there somewhere, confidence that the test does what it’s supposed to do?
DR. GULCHER: Good question. I think, you know, currently the oversight for a test sold to an American or whose results are given to an American is that CMS or FDA have to have certified or -- they’re the ones that regulate laboratory derived tests or testing kits, and those are already in place. What we’ve tried to do is emphasize that we’re CLIA compliant in the context of CMS and FDA. And now whether or not consumers understand all of that, you know, that’s a different story, but we try to emphasize that there is a regulation that covers analytical and clinical validity with laboratory-derived tests and that’s the extent of it. But if the question then becomes, is there a need for further oversight or beyond what oversight already exists, I guess that’s a different question for the consumer.
DR. TUCKSON: Let me just ask, Sarah Carr (phonetic) just remind me for my information, is the report from the Secretary’s Advisory Committee with its recommendations to the Secretary, is that up yet online or is it dependent upon waiting for the Secretary’s Office?
MS. CARR: It’s online.
DR. TUCKSON: It is online. So I would urge -- first of all, I would urge all of the private-sector companies that are doing this work to review, if you would, the Secretary of Health’s Advisory Committee on Genetics, Health, and Society -- easy to find; and look at the report on the recommendations regarding the adequacy of oversight. And I think that the question becomes, if private-sector is convinced that there may be an issue here of a few holes, that we might want to have private- sector come forward and partner with public-sector to hurry up and plug those holes and try to get this thing done. I won't say any more as the moderator because I’ll start to sound like what I am, which is an advocate. But I am concerned, and I believe that this needs to get dealt with in an expeditious way and that the Secretary’s office shouldn’t be down here trying to figure this thing out, I think the public-sector should step up to the plate and help to close that deal. Do you have a comment to make on that?
MS. PHELAN: I do. And I think the private- sector is stepping up to the plate and, to some degree, trying to figure out where the regulatory environment currently has left off and where the industry can try to help create guidelines and, you know, best code of practice and things like that. So I think you’ll be hearing more about that.
DR. TUCKSON: Good.
MS. PHELAN: But can I take a cut at your answer on do we have enough regulation for the consumer to decide?
DR. TUCKSON: Yes.
MS. PHELAN: So, right now, the unfortunate thing in this industry is that these terminologies: FDA oversight, FDA approved laboratory tests, CLIA -- these don’t mean anything to the consumer. So at DNA Direct, we offer tests that are done in CLIA labs and with medical guidelines established. And we put all that on the site, but it doesn’t stop a consumer from looking at another website for a genetic test for -- I’m just going to use a random thing like, you know, for baldness -- male-pattern baldness -- something that may or may not have scientific rigor, and looking at it and saying, “Well, my assumption is this -- it’s on the web, it should be regulated by the government.” And I think this is really what caused California to actually step up with its cease and desist letters that it issued to a number of companies over the last two weeks. Is, you know, a question was, “Are they providing these with medical oversight or are they doing it in CLIA labs,” but also, this big question that ultimately all of these companies, all of us have to demonstrate, is are the tests that are being offered scientifically valid?
DR. TUCKSON: Well, I appreciate the point. And we’ll get into some -- obviously we’re getting into some very interesting issues here. You know, you said, “I have to wonder,” I was very much impressed by your slide of your test case that you’re going to do on Alzheimer’s, APOE, I thought that was pretty good. I kept wondering the level of education that the consumer would have to have to be able to deal with that. I mean, you’re right there, you’ve got the words, and I’m sure there is an explanation of clinical utility and clinical validity --
MS. PHELAN: Oh, yeah. It’s --
DR. TUCKSON: I’m still trying to think back to the so-called average American. It’s like, you know, you’ve got to work your way through it, so unless you can figure it out to know whether you’re in a risk or not risk, I mean, in some level it seems to me there ought to be a common (inaudible) that says, “Hey, this is legitimate.” And you shouldn’t as a consumer have to sort of be lucky enough to be able to stumble into whether or not you’re in shaky ground or not.
But let me ask you, when the people call you all -- and I’m not sure what population of people call you -- what are they saying? Is there anxiety on their part around -- and I doubt it, but let me just ask -- you know, reliability, validity, and/or privacy; what happens in those conversations?
MS. PHELAN: All right. I think that looking at the Yankelovich study, I have to say that our population has always been what I refer to as the rightly worried, which is not a particular category that you had, but it’s one we use. And these are people who have a known personal or a family history of a medical problem. And to answer the literacy question, it cuts across all educational, social strata. And it’s because they have an underlying concern about a health care issue, and what they do is they read up. And so, believe it when somebody has a family history of cancer -- early-onset breast cancer -- they are going to learn about the BRCA gene.
Now, you know, should they all have to wade into that level of depth? No. But for those who want to, they need to. And any site or service has to be able to provide that.
When consumers are approaching testing, they do it very thoughtfully. This is not a booming business of people throwing down $3000 for testing for no good reason. Believe me, people think through genetic testing. They think through the pros and cons, they think through the privacy issues, they think through privacy even in their own family. They want to know, if I test what does that mean to my other family members, do I need to provide them with the information around the results? I mean, these things have a lot of implications. They think through their insurance, what’s going to happen if they have not yet had cancer or known anything symptomatic, and that’s part of what we do and it’s called counseling. It’s to help people really way that and think through it.
DR. TUCKSON: Well, thank you. I must say, I was very impressed with Eric’s example that he went out and -- Dr. Topol -- and did his own and he looks at these probabilities and -- and you’ve talked about probabilities and you’re made some decisions based on probabilities.
And I continue to wonder, how does the public know that those probabilities are right? I mean, upon what -- who’s -- you’re making a -- I mean, there’s so much subjectiveness here for a person, and at the end of the day, okay, it’s 1 in 6; well, who says it’s 1 in 6? How do I know that’s right? Who are these people that are saying these things? And is there any argument about -- is it really 1 in 7 or 1 in 8, did somebody’s paper disagree with Bob Smith’s paper on that? How do I go back and actually know that? This is fundamental. But Deven, as you look at this stuff from a macro-policy point of view, “If you’ve met one consumer,” you say, “you’ve met one consumer.” How does one then suggest to the extent that you would advocate for any level playing field of public policy; how do you make public policy when you have this range of, not only genetic variation, but personal decision-making variation?
MS. MCGRAW: Right. Well, you know, one thing is to consider that there ought to be a baseline below which -- you refer to it yourself, the sort of baseline of either oversight, a set of sort of ground rules that all the companies in this space, the health care providers have to follow. That’s certainly the pattern that we’ve got, you know, in terms of our own privacy laws in this country. There’s the federal baseline of HIPPA and some of the states have chosen to go beyond it, and some providers in fact even go voluntarily beyond it. And then the ability of folks to, with all of the right information and tools in hand, to be able to make decisions that are sort of very individually centric and be able to say, you know, for me, I’m okay with sticking my entire genetic sequence on the web. I’m okay with that; I’m even okay with sticking my name on the end of it. You know, you’re permitted to do that, but that doesn’t -- even if there are some -- there is some variability in terms of consumer taste and concerns, it doesn’t absolve us of the responsibility for creating at least a set of rules below which, you know, no one should fall. So --
DR. TUCKSON: All right. Well, the floor is open, and I can't believe it, but Kevin -- Father Fitzgerald is first in line. And we can only go wonderful from there. And you’ll be next.
FATHER FITZGERALD: Thank you. Kevin Fitzgerald from Georgetown University and also from the Secretary’s Advisory Committee on Genetics, Health, and Society.
Question which could be for any panel, but since this panel is more focused on the consumer, I thought it was more appropriate here. People are talking about doing the good; no one doubts that someone wants to start a company to do something wrong or evil or bad. All right. So no one’s questioning that; the question is, how do you determine the good? Who is good? Who is deciding what the good is? Especially in a situation where we have such problems that we see all the time, in research in particular, with what we call therapeutic misconception.
Is that a concept familiar? This is basically, you know, someone comes into a phase one trial, you go through all the informed consent forms, you sit down with them, you go through the entire thing, they go through the six months of chemotherapy or whatever it is -- if it’s oncology -- they come out, six months later you go back and you ask them, “Why did you go through that?” And they say,“Because I thought it would do me some good,” in spite of the fact it was a phase one trial. So what -- and this, again, we heard before, you know, “This is probabilistic, it’s statistics.” True. It is statistics, but it’s not baseball we’re talking about. If you have a debate between whether batting average is better than on-base percentage is better than slugging percentage, that has some significance in some part of the world. We’re talking about people’s health, their own understanding of their well-being and who they are. How do you address that concern in your industry? Do you address it, and if not, what are you going to do?
DR. GULCHER: Yeah. And let’s (inaudible) our industry, I’m not sure what you’re referring to. If we talk about the need, the un-medical need for risk assessment, okay, that’s a medicine-wide issue. Right? And a demand for that, that’s the basis for why all these studies have been done -- the genome-wide association studies have been done. That’s what we’re searching for here, right, risk assessment. So it’s not just somebody creating a new industry out of -- and trying to create a need that doesn’t exist; there is a need. Right? As I mentioned with prostate cancer, you have very limited information that you can impart to a patient to help decide how vigorously do you search for cancer. All right. And the best treatment for prostate cancer is early detection, so I would contend that actually there is more of a demand from physicians and the health care system for this kind of information, rather than the industry sort of pushing it on to consumers or patients or physicians.
FATHER FITZGERALD: Well, okay. But that’s still in a sense doesn’t somehow recuse you of the responsibility for addressing it.
DR. GULCHER: Oh, no. Yeah, okay. Responsibility to make sure the information that we create is reliable, and I think we described that. Is it useful? That -- whether or not it’s useful really is between the physician and their patient, right, or a guidelines among professional societies or whatever, and this information feeds into those guidelines, right, because it’s setting an additional risk -- it’s adding additional risk to other things that are already being assessed, and that may trigger whether or not you do something different with your physician. But we’re not telling patients what to do with this information other than act on it only in the context of a physician, right? We don’t -- we offer genetic counseling, but we don’t pretend to think that our genetic counselors are going to tell patients what to do with this information. They may help try to frame what risk means, but it’s really the physician who can work together with the patient to act on that information, just as physicians act on other risk information. It’s just another clinical risk, there’s nothing new about that.
DR. TUCKSON: Then the issue then ultimately -- and we raised it earlier, is, again, how do we educate the physician to know what to make sense of it and upon what database does the physician make those choices? I think it gets down to, again, I think that as we get ready for Muin’s question, it’s the notion of how much oversight does there have to be with, you know, heavy-handed government looking out for vulnerable people. And the Lord knows that if there were ever a vulnerable people, this is a case of vulnerability versus having the industry (inaudible) large, sort of, say, “Okay, we’re going to put some best practices” -- I think you used that word, Ryan in your -- you know, in terms of industry standard best practices, so you don’t have to have the poor Secretary of Health have to come in and ride roughshot over this thing.
DR. KOUHRY: Just to elaborate a bit more on your clinical validity and utility issues. I mean, if we’re looking for credible information that -- as a consumer who is savvy with numbers -- I mean, I love numbers, but it’s, you know, I’d like to get sort of the most up to date information that’s credible for my own health care and disease prevention. Now, the problem with the existing literature right now -- and you’ve alluded to it -- it’s risk factor information. I mean, right now, we have a database of about 35,000 genetic association articles, and, you know, you do the meta analysis and the, you know, all the GWA’s, and you put them together. And then you try to go from replication to a risk estimate for an individual. And the three companies do it in slightly different ways, and I’ve had reporters talk to me where they took the three tests and they got three different, you know, sets of advice from three companies. It’s the same genome; however, it depends on how you read the literature and how you put the information together. I mean, one company puts out lifetime risk estimates. The other company puts out incidence rates over the next ten years. Basically, the data that are used for the second tier analysis is not from these papers, it’s from existing data sets, like, see registries for cancer incidents in the population. And then you extrapolate from here and there, and when you say your lifetime risk for Type 2 diabetes is 1 out of 3 as an average, that’s an average risk for Type 2 diabetes for a person born today, not the person who is 50-years-old who might be taking your test tomorrow morning. So I think playing with numbers -- this is not BRCA1 or Huntington disease anymore where you follow the modes of inheritance, its chromosomic dominant and recessive. This uses extraneous pieces of information to arrive at these clinical validity estimates, and without industry wide standards -- even with the best possible intention -- there’s going to be severe variation that is going to be translated to different sets of advice from one group to another, and perhaps different courses of action.
One more thing. Clinical utility, you told us your wonderful story about prostate cancer. I mean, I don't know if we replicate your story a 100 times and we do a clinical trial about a situation like yours, whether or not there will be clinical utility from having had your genetic test done. I mean, I’m not questioning your own, sort of, decision for what you’re going to undergo -- and that’s strictly a personal decision, but there has to be some clinical trials to accompany this kind of individual thinking because at the end of the day somebody has to pay for these procedures. And if we’re going to label the whole population into risk strata across thousands of data points, we’re all going to be at increased risk of something and decreased risks for something else. So unless we standardize this and collect the kind of information that we’re going to use for medical practice, it’s going to be a mess out there.
DR. TUCKSON: That’s a very, very thoughtful question. Let me ask you, Ryan, how --
DR. GULCHER: Are we allowed to respond to it?
DR. TUCKSON: Go ahead. Okay. Sorry. We got a couple minutes. We go -- by the way, I was given leeway since we started late, we get to go until 10 after, so, but Ryan let me just -- and before we get to you. How freaked out are your counselors by the first part of Muin’s question and saying -- I mean, do you feel like when your folk are sitting there on the phone doing this counseling interaction, that you’re sometimes sitting on what could or could not be a shaky database around which you are giving this kind of advice.
MS. PHELAN: No, not at all. But that is because we’re not in a shaky territory. We’re not doing genome-wide arrays across the board, so I’m really not the one to answer your question. But I do have an opinion about the difference between clinical utility and personal utility, and you saw that up in my slide. I think that personal utility is something that we’re all going to have to wrestle with here. As all of this testing is coming aboard, people like Jeff -- if I can just use you an example as a consumer for a minute -- are going to find value with some of this information, that they may make health care choices with. It may be very different than what would be reviewed as clinical utility down the road, and I think that is something that -- you know, that is going to be a tension that we have right now because it’s going to take a long time for some of these new technologies to actually get all of the way through to where there’s proof of clinical utility. And so it’s not what I do.
DR. TUCKSON: Great. Thanks. Jeff.
DR. GULCHER: Yep. Well, first of all, the last statement you made that, oh, if we do deCODEme or 23andMe or Navigenics, we’re going to find out that, oh, I’m at higher risk for some things and I’m a lower risk for other things. Well, that’s the nature of the beast, right?
That’s the whole point, right? You have -- we have differences in risk, right? Some of us will be at higher risk for cancer, other people will be risk for cardiovascular disease, et cetera. And isn’t it better to know that, understand that risk early on so that you have the opportunity of either preventing those diseases, or maybe you’re more highly motivated to finally quit your drinking and excess eating. Okay. Or you can do something about it with management with your physician, or early detection in the context of cancer. So that’s the whole point. The question is, is that -- is the magnitude of this risk high enough to act on, does it save money in the health care system overall, does one need to do a 15 to 20 year outcome study, right, those studies don’t exist. Same thing for prostate cancer; there is no such thing as a long-term outcome study for prostate cancer, right? But yet, there are guidelines that suggest once you achieve a certain risk -- 20 percent lifetime risk for breast cancer, then you should have -- and I think a lot of companies like Reed’s pay for extra attention, extra MRI screening in addition to the usual mammography for breast cancer. But you have to reach a certain risk, right, before that happens, and that risk is dependent on various things, which can also include validated markers for genetic risk that can put you up to that threshold.
And then you fit into the established guidelines that say, once a woman achieves a certain five-year risk or lifetime risk, these are what the recommendations are.
DR. TUCKSON: Well, this is fascinating, and I think that Muin’s ending point was, at the end of the day, somebody’s got to pay for these assumptions. And so, does CMS, with all of its active budget problems right this second, does CMS actually start to say, okay, if you have this kind of a mathematics that you put on your slide -- something like it -- and I don’t want to make it personal to you, but just say you put mathematics up there that come up with a number, at what point should the public insurance reimburse that prostatectomy, and how do you, sort of, make those decisions as a society struggling with some real choices. So I think your answer was responsive, and I think Muin’s ending thought was also very important. As we get to these last couple questions, Deven, I just want to make sure that I ask you real quick, though, one thing I was going to make sure we get at, and that is -- I’m -- so I’m going to flip this whole thing around. Where everything here has been cautious and conservative, and at the end of the day, how do you -- as somebody who I think is an advocate for caution -- can we pile on so much caution that we just stifle this whole dangone (phonetic) thing and we don’t wind up with diddly squat?
MS. MCGRAW: Well, I certainly hope not. I like to label myself as the privacy advocate who, like was said in the very beginning, I don’t believe in using the word balance because I think you can have privacy protections and advanced medicine through increased knowledge and grabbing on to the most promising information that’s out there, whether it’s genetic testing or what it might be.
But you have to really focus on both because without consumer trust in either the testing enterprise or the use of the information, we really won't be able to move this forward in ways that we want to. And too often, the balance question means that, well, we won't -- you know, we have enough privacy and security and we need to --
DR. TUCKSON: Diddly squat, by the way, is a highly technical concept.
MS. MCGRAW: It is.
MS. AVEY: I just thought I would comment on -- I don't know if this is on -- but Muin’s point. We take that --
DR. TUCKSON: Right. Would you tell us your name?
MS. AVEY: I’m sorry. Linda Avey with 23andMe. And the comment about a person getting the testing done with the three companies and getting some differential data back, we fully admit that that is the case. And in fact, Mari Baker and Ryan Phelan and Jeffery Gulcher and I, along with the Personalized Medicine Coalition had a breakfast this morning that -- and this was really started by Navigenics -- they realize the importance of all of us working together in this new nascent industry, that we need to develop standards. So that is something that we’re working on. We’re really excited to have the PMC take the charge on this because they’re a neutral body and they can bring in some of the other stakeholders in this space who really want to have a voice in how we set up these standards. But we do realize that that is a problem right now, and that’s why we need to work together, because we do have to make certain assumptions. Do we look at lifetime risk, do we look at risk over ten years?
Those are assumptions that we can all come together as a community and decide what is the best way to do this, and then we will conduct it that way. So I just wanted to make that point.
DR. TUCKSON: Well, I think that should be applauded. And I would just say, ya’ll better really start moving fast. [LAUGHTER]
Because it’s so necessary. And that’s responsible behavior, but ya’ll got a whole big gap to hurry up and close or else somebody else is going to try to close it for you. Last comment.
MS. JOHANSEN: Katie (phonetic) Johansen from the American Medical Association. Two quick questions -- one for Jeffrey. I’m curious about what the reaction of your primary care physician was when you brought in your deCODE results, and whether you think that that is -- was a general reaction or whether that was specialized because you obviously were an employee of deCODE. And then the next question is for Ryan, and maybe it’s more of a comment, but I question the appropriateness of having on your DNA prospective sheet, the last question about personal utility because I think by including that question about, you know, would this information be helpful to you, with a test that has very low predictive value and low clinical validity, I think that question implies that that test is going to give you the answer to that question when really the low predictive value and the low clinical validity just don’t add up for that test.
DR. TUCKSON: Those are good. First and then second. Good.
DR. GULCHER: Yeah. So you would say I was stacking the deck on my primary care physician, I guess. Although the -- when it comes to -- and he was very of course intrigued by the reports that I brought him. But the urologist, I think, is the more interesting -- how his behavior changed -- that normally, somebody with a PSA of 2.5 in my age range, he would not have acted on, and he was more interested in the genetic profiling as being the determinative of whether or not he would biopsy or not.
But I should mention, there was a preventive cardiologist that had a patient brought in a PSA of 3, who was 55- years-old and dint have any other risk factors, and he had ordered deCODEme for the patient in the context of cardiovascular profiling, and then the patient had higher risk for prostate cancer. And he was just biopsied last week and had even more cancer in his prostate than I had.
But when it comes to, you know, this type of information, how do we educate physicians, or inform them at least of this, we try to encapsulate what the information is. We try to document the clinical validity, okay, with all of the different articles. And we’re not talking about the 35,000 different genetic association articles that Muin was talking about. We’re talking about, this is a different era, which I think Dr. Topol addressed. We’re now talking about markers that do indeed replicate; we’re not talking about the articles that end up somehow on molecular psychiatry that don’t necessarily replicate, right? We’re talking about articles that get published in peer-reviewed journals like New England Journal and Nature Genetics where the standard now is much higher, admittedly, than even two or three years ago --
DR. TUCKSON: Jeff, one just -- just -- would your -- based on your guesstimate on your conversation with your urologist, what would he or she have said if the biopsy had a 1 percent of --
DR. GULCHER: Right. Or was low-grade?
DR. TUCKSON: Would you think that he or she would have changed her advice to you?
DR. GULCHER: Oh, absolutely. If it were a low- grade tumor or there was no tumor, then, of course he wouldn’t have recommended a prostatectomy. Because it was intermediate-grade and had, you know, 15 percent in my prostate --
DR. TUCKSON: Okay.
DR. GULCHER: -- that by itself, you know, indicates --
DR. TUCKSON: Okay. Last half, and then we’re closing off.
MS. PHELAN: I’m going to partly answer his question about what do physicians do with this information. We do outcome studies -- not a study, but outcome research on our customers. What do they do with medical information that they get from DNA Direct? The vast majority share it with their physician, no surprise with the Yankelovich document. And when asked, did the physician find it of help? Very high -- 80 percent satisfaction. And did they use it to make a better health care choice? Very high numbers. So these are people who take that information to their doctor and use it for health care decision-making. And yes, the personal utility is a little confusing up in that one, but again, it was a placeholder so we’ll work on that one.
DR. TUCKSON: Thank you. And would you give our good panel a round of applause? [APPLAUSE]
Looking to the Future of Consumer Services (moderated Discussion)
We will wrap this up at 5:30. I know people have airplanes and transportation arrangements, so we will not let this drag on. But if you could help me by taking your seats so we can get started with the last panel I would rally appreciate it.
There will be some overlap here; we’ve gone over many of these issues with the other panels, and certainly with the questions and answers. But as I said at the beginning, some things are simply worth redunding.
This is a panel on what’s available now and what’s available in the future. It’ll be chaired by Nancy Johnson, who is currently a Senior Public Policy Advisor at Baker Donelson. Her background is 24 years in Congress -- I heard a Congressman say one time that being in Congress working is like dog years, so one year seems to last as long as seven. So I don’t know what the math is on 24 years, but congratulations on such a wonderful career and thanks for being here. She’s had a long-term interest in health care, being a sponsor and supporter for things like mental health, (inaudible) legislation, Patients’ Bill of Rights, and my personal favorite, Taxpayer Bill of Rights; has had many awards to include the National Patient Advocate Foundation and as with the other panels, she will introduce the other panelists and we will go ahead and get started.
MS. JOHNSON: Thank you. We are the last. And as we start -- but -- the questions have been wonderful, the presentations have been wonderful; I certainly have gotten a lot out of my afternoon. And I want to put this last discussion -- well, in fact, the whole afternoon’s discussion in a slightly different context.
If we are to continue to see breakthroughs in medical science, if we are to speed the delivery of those breakthroughs to patients, if we are to provide access to affordable health care to all Americans, then we must abandon our illness treatment model of health care. Furthermore, if we are to afford the kind of health that science and universal coverage will provide for this nation, we absolutely must abandon our illness treatment model of health care. Over the course of recent years -- recent decades, I guess I would say, we’ve learned to keep a lot of people alive. And we have fundamentally altered the kind of care that most people need. And in so doing, we have created what I call the 80/20 problem. Some people say it’s not quite 80/20 it’s 75/25, whatever. The bottom line is that whether it’s the public system or the private system, 75 to 80 percent of our dollars go to 20 to 25 percent of the people. And that’s because we are trying to manage people with multiple chronic illnesses only after they get sick enough to go to the doctor -- in other words, with an illness treatment model. So the good news is that in a reformed -- in a health and wellness centered model, patients have to be more active. You cannot manage someone’s chronic disease if they do not want you to manage that chronic disease because you can’t take their medicine for them. So it’s very different -- very simple, but it’s absolutely going to be a dramatically different system from the point of view of the patient. They will have to be far more involved in their health than they are now in their health care. So that’s the good news. The bad news is that being involved in your health care isn’t always easy. And furthermore, we have never involved patients much in their health care; we have told them what was wrong and what they needed to do to get better. You cannot do that in a health and wellness system, so I see this conversation about how do we talk to people about genetic issues as part of this whole larger issue of how are we going to talk to ourselves about a patient-centered health care system in which, truly, the patient is a part of the care delivery system.
One of the things I worked the most on was the development of chronic disease management demonstrations. And the hardest thing was how do you get this into a fee- for-service system. And when you look at what’s happened, you see all of those systems, and that’s why the call-in system, the telephone advising system that we’ve heard something about is something we actually know about. But we also know that you have to change the way you talk to patients and you have to constantly change the way you talk to patients because patients are experiencing different things as they manage their own diseases. So this issue of, how do we talk to ourselves and what are the implications, is something that’s extraordinarily important to us not just in terms of how are we going to integrate genetic medicine into a more holistic health- oriented care delivery system, but because in that area almost more than any other area, honesty is hard, transparency is difficult, but if your communication isn’t both honest and transparent, we will not be able to generate the quality health system that we have the science and technology to enjoy.
So I consider this an extremely important discussion that we’re having here today, and I’m delighted with the people that we have in this panel, as we have had excellent people all day long. There are different characteristics to these conversations; when I talk to doctors who do pharmacogenetic testing for patients that have mental illness or some other things where there’s quite a specific relationship between the testing and the medical treatment, you certainly get one kind of response.
But in the larger arena, how do we make sure that what we begin to -- how we begin to talk to ourselves and how we begin to handle this new knowledge in that communication does indeed deepen not only the health knowledge of those who take the tests, but the health knowledge of the general population. And how do we deepen their ability to judge value from the kinds of information that they are going to get in the future, whether it’s about how to manage their diabetes or in this rather more complex but very important area. I mean, what is the relationship between genetic testing and diabetes? And if it comes to where there’s a pretty good relationship, how does government foster that? How does the private sector react? What are we doing to motivate? So it’s really a big and important conversation, and I’m delighted with the kind of people that are going to do the big talking here.
But we’ll start with Ronni Sandroff who is Director of Health and Family for Consumer Reports and responsible for the health coverage across media products, including Consumer Reports magazine, Consumer Reports on Health’s monthly newspaper, CR TV, and the new health website, consumerreportshealth.org. Thank you very much, Sandy (sic), for being here with us.
MS. SANDROFF: Oh, thank you. I’m so happy to be here. It’s been a very interesting meeting so far. Someone asked me in the ladies room, why is Consumer Reports here? And I’ve been health editor there for nine years; we’ve covered health for 70 years, but we are best known for the car ratings.
So one of the things we’ve been doing for the last few years is trying to apply some of the methods we use for ratings and recommendations of products to rate more health care products and even to get into rating treatments, drugs, hospitals, and so on. It’s not the same thing at all, but I think what is the same is our ability to communicate to consumers the relative value of various products, and in some way, that’s kind of, you know, dealing with relative risk is one of the hardest concepts, but that is what we do all the time.
So how do we evaluate health care service? We look at the research and we look to you for good summaries of the research; we don’t do it ourselves. But we’re also aware of something I call flopability. We do an article every couple of years on overturned health truths, and we never have trouble finding material. So we’ve done estrogen, antibiotics for ear infections, I could go on and on. So I’m concerned about that -- the genetic test results that you get today, what will they mean in five years or in five months? I also am concerned as the many panelists have raised on the usefulness of the results, both medical -- and I do think there is personal usefulness if there’s good predictive value, but perhaps not that much you can do about the disease right now. I think it’s up to the individual to decide what they might do with that information.
But the thing that really attracted me to this meeting was the prediction from the organizers that there was going to be an explosion in direct-to-consumer advertising for genetic testing over the next few years. And we are very concerned about the power of dtc ads. I mean, we’ve seen in the pharmaceutical area over the last ten years, perhaps some good education coming from all the pharma ads, but also maybe a waste of medical time. I mean, how many times a day are you told to ask your doctor about something that might not really be your most important issue? And very much concerned about the over- prescription of some new drugs because of the advertising.
I’d also like to support a point that Ryan made earlier. People will assume when they hear these ads that they’re on the up and up; they will assume that the results are valid, that the government has kind of taken care of it. I mean, we’ve surveyed -- and many people have surveyed consumers about the use of dietary supplements, and an amazing amount of people assume that the FDA has tested them for safety and efficacy. After all, they allow them on the shelves; they allow them to be advertised on TV. So I think that that’s an area that we will be watching. One of the great things about working for consumer Reports is that we don’t take ads, which means we can criticize your ads. So we’ll be watching that.
But what I was asked to do for this panel was to -- as we’re looking to the future, to come up with some cases. And we kind of do this in publishing; we try to imagine the consumer. So I’ve imagined two consumers who in -- maybe two -- maybe not now, but in two years, three years, might be attracted by direct-to-consumer ads to get their genomes tested.
My first victim here is Adam (phonetic), 42, and he’s a bit of health nut. He wants the best of everything. Both his parents are overweight, they both have diabetes, they both have heart problems, and they’re on multiple medications. And he’s already exercising and dieting and doing everything. But he’s gotten a promise that he’s going to get customized advice about how to lower his risk. So my question to the rest of the panelists is, you know, will that promise be fulfilled?
Is it worth his $1000?
And then let’s go out a few more years into the future and think about Jack and Jill, and they’re both 32 and they’ve just become engaged. She has a brother with autism, he has a family history of Type 1 diabetes, although he doesn’t have it himself, and they’ve decided to have their whole genomes tested and scoured for risks. So will a genetic test give them useful information about whether to have children? If they decide to adopt, will those adopted children be screened for genetic risks? I’m not sure, but I’m very interested.
MS. JOHNSON. Thank you very much. Our next panelist is Angela Trepanier; she is a certified genetic counselor and has led the development of genetic counseling at at least two universities and is currently the President of the National Society of Genetic Counselors, responsible for leading that association and being its chief spokesman. It’s a pleasure to have you here today, Angela.
MS. TREPANIER: Thank you. So I’m not going to answer your questions, but present my own cases for consideration because the points that I wanted to make is that for personalized medicine really to have promise, you have to have a personalize approach based on the patient.
So I’m going to start with two patients: Alice (phonetic) and Mary (phonetic), both of whom had a father who was diagnosed with diabetes in their 40’s. With Alice’s case, her father was diagnosed after a routine physical exam revealed that he had an elevated blood glucose. He had the appropriate follow-up testing and was found to have the disease, and then managed his condition through exercise, diet, and medication. So he got that information about his health through routine health care and was proactive about the way he managed it. And so the message is that Alice got from her father is that diabetes is manageable, you just have to do some things, but these are very reasonable things to do to prevent complications from the disease.
Mary, on the other hand, her father was diagnosed with diabetes after being hospitalized with severe elevated glucose and ketoacidosis, and he almost died at the time of hospitalization. She was 7-years-old at the time and remembers that critical event very well.
Her family is Italian; their diet consisted of pastas, meat sauces, not things consistent with a good diet for a diabetic. And her father was obese and didn’t comply with diet or exercise regimen, but did take his medication.
But he unfortunately died at the age of 65 from congestive heart failure, basically, complications of diabetes. So if you take these same two women who at face value have exactly the same family history in terms of at least the person affected, their needs are going to be very, very different. Alice, I know -- now know that you can classify her as one of these “lead the way” people because her family taught her that if you have a condition or a risk, you manage it aggressively and it’s easy to do that. She’s already dieting, she’s exercising, and she might present for genetic testing and genetic counseling because she wants to know what else she can do. She’s already scoured the internet, she’s found out about the genomic testing that’s available. We’ll assume this is three or four or five -- I’m not sure how many years down the road -- when we know that what the value of the information is when it’s coming from one of these tests.
And not only does she want to know what her risk is, because she sort of already thinks she’s at increased risk, she wants to know if there’s something else she can do. She’s happy to diet, she’s happy to exercise, but she wants to take it to the next level.
Mary, on the other hand, is referred for genetic testing and counseling because her primary care physician is frustrated. She’s overweight, she doesn’t exercise -- for years he’s been telling her that those are risk factors for the disease that her father died from, but she won't budge. So he’s sending her off to genetics to get information and hopes that that will trigger some change in her behavior.
And so the approach that you need to take with these two women or two men or whoever it is, is going to be completely different. With Alice, who is very proactive and has done the research, she is coming in potentially for information and she wants to be proactive. With Mary, if you don’t deal with the anxiety that she has about the diabetes in the family and figure out why it is -- with the assistance of other health care providers, that she won't comply with diet and exercise and what the issues are that prevent her from doing that, then giving her a genetic test may not provide any benefit whatsoever. And so the important message here is that it’s not enough just to have a genomic or a genetic test, you really have to take into consideration the person presenting in front of you and what their concerns are and what they’re capable of doing. And then you also have to -- for both women -- present the information that you’re providing in a way that’s conducive to how they learn information. So we know from genetic counseling that some people are visual learners, some people are oral learners, some people want face-to-face consultations, some people want to do telephone counseling, some people want internet resources, some people want written materials. All of those things have to be available if you want to provide good information to all the people who might potentially benefit from genetic testing and counseling.
You also want to make sure that -- here in this example I have you in the example of two people who have a family history, so they have a context for the disease for which they might be at increased risk -- there are going to be a lot of genetic tests, like we’ve already heard, where there’s no family history and all of the sudden somebody’s told -- assuming that it’s credible information -- that they have an increased risk for something that they have no experience with. And they’re going to need something more than your word to make them believe that that information is valid information.
So what we need to do now in getting ready for good genetic testing, is start to educate people about the broader applications of genomic testing. It’s not just about these single gene disorders that other people have, it’s about chronic diseases that any of us can get, and we need to start including that information in our health classes and also in our textbooks, and then also -- and most important -- we need to make sure that all health care providers are educated about the availability and the validity and the credibility of genomic tests. Because coming and getting a test result and just meeting with a genetic counselor who tells you this is what you need to do, is meaningless if the rest of the health care team that needs to be there to help that person act on that information doesn’t give them the same information and validate what they’ve heard.
MS. JOHNSON: Thank you very much, Angela. You can certainly see through her comments how the system has to change -- how profoundly it has to change. Katherine Johansen is the Senior Scientist at the American Medical Association’s Program in Genetics and Molecular Medicine. Before joining the AMA, her main focus was laboratory research on molecular cell and developmental biology projects. At the AMA, she leads the development of physician education programs on medical genetics, including pharmacogenetics, the genetic basis for Warfarin dosing, the genetics of common disorders, and the translation of genetic technology into the clinical setting.
Thanks, Kathy.
MS. JOHANSEN: Thank you. So, like Nancy said, our main focus at the AMA and the Program on Genetics and Molecular Medicine is to provide educational resources and support to physicians as they integrate new genetic technologies into clinical practice. And so because this area at this dtc genetic testing area has really exploded in the last few years, it is an area that has become an area of interest and concern even for the AMA.
So recently the Board of Trustees of the AMA decided to study this in a bit more detail and recently set forth policy on what the AMA feels should be the next few steps in dealing with direct-to-consumer genetic testing.
So in 2004, which is when our old policy was established, the AMA House of Delegates which is the policy setting chamber of the AMA, really just decided to generally oppose direct-to-consumer genetic testing. And one can imagine that there were many reasons for that and probably one of them is something that we heard a bit earlier, which is that there’s kind of an old fashioned view that physicians want to be the sole source of health information and don’t want to give that up. And that is certainly possible that that was the reason that our old policy existed, but I think that physicians now are realizing that that is just not going to be effective.
It’s not going to be effective to just blanketly oppose direct-to-consumer genetic testing because it’s here and it’s something that needs to be dealt with. So at our recent policy-making meeting in June, a new policy was adopted which really still encourages patients or consumers to come to their physicians with questions, but doesn’t blanketly oppose direct-to-consumer genetic testing anymore. So instead, like I said, the AMA encourages consumers with questions to come to their physician. And it also addresses advertising, which is something we haven’t really talked about a lot yet. There -- a lot of the information that consumers are getting about direct- to-consumer genetic testing is through advertising, and so one concern that physicians have is that they are getting truthful information in that advertising since that advertising is partially what drives consumers to decide that they want to take this test.
So the AMA, along with some other organizations, would really like to come up with some good criteria for direct-to-consumer advertising to make sure that the advertising is truthful and not misleading, it presents a fair balance of the tests’ capabilities and limitations so that the consumer -- and all at the consumer level so that the consumer really understands what it is that they are about to undertake.
The policy also advocates for education of physicians. And I think is going to be key to making sure that consumers know what to do with the information once they have this genetic test. The problem that we’ve seen, however, is that there are some physicians who are not ready to deal with these test results when their patients bring them in.
So we realize that we are advocating for physicians to provide education for patients that come in with these types of tests, but we need to make sure that the physicians are knowledgeable, first of all, in basic genetics, which we see is not the case sometimes. And we also need to make sure that they are knowledgeable in how to interpret a genetic test.
So the -- I guess the point of this panel and the questions that we were asked to answer is, what kind of resources do consumers need? And so I’m just going to take a step, sort of a different attack on that and ask, what is it that the physician can do for the consumer? If we are indeed advocating that physicians should be a source of information for consumers who want to undertake this testing, what is it that the physician can provide to the patient. And before that even can be answered the physician has to understand some intricacies about these tests. The physician first of all has to understand whether a test is even indicated for this patient, and that hinges upon a basic knowledge of genetics in the first place.
The physician needs to be able to tell the patient whether a test is worth getting. Is there any scientific evidence that this test is really worth getting, and is this test going to tell the patient anything? And again, that goes back to the physician being able to understand what the predictive value and the utility of a test actually is. And then the physician also has to be able to use results that a patient might bring into them to come up with a therapeutic plan. And again, that gets back to, does the physician know enough about genetics to use that information in the context of other health information of that patient to come up with a therapeutic plan for that patient.
So in the future I think we really are just going to see more and more of this direct-to-consumer genetic testing and I think that just underpins the need for physicians to be educated a bit more on this topic. And we also need to make sure that this is not a question of physicians just wanting a piece of the pie and not wanting to let go of that power of being able to have the control over ordering a genetic test. This is something that really does have the potential for benefit for the patient of done in a proper way. And so if we can sort of convince physicians to accept this information and understand whether they should accept the information when a test is actually valuable and when it might not be, we might be able to actually give physicians another tool in their, sort of, arsenal in diagnosis and therapy. So, thank you.
MS. JOHNSON: Thank you. As the wife of a physician, I remember those discussions or the conversations that didn’t have a clear to-do list, and that is hard. Mari Baker is currently the Executive in Residence at Kleiner Perkins Caufield & Byers. But before that, she was President of the BabyCenter of Johnson & Johnson Company which was the leading website for new and expectant parents winning numerous online health awards, but expanding also significantly offline and internationally. Equally interesting was her work as Senior Vice President at Intuit where she was the product manager for Quicken and led it’s growth into the number one personal finance product in the world along with international expansion and the launch of Quicken.com. So she comes to Navigenics with a lot of experience. Mari.
MS. BAKER: Thanks, Nancy. And actually, currently I’m President and CEO of Navigenics and have been since early on in the company’s days and had the opportunity to be involved with the company before it actually got funding from it’s investors. And the vision that we’ve always had with the company that our founders, Dr. David Agus and Dr. Dietrich Stephan brought to the table was exactly the line of thinking, Nancy, that I think you used in a lot of the introduction is that there was tremendous opportunity to use genetic information to improve health outcomes, to identify people at risk for disease, and begin to have them work with their physicians to identify potential courses of action, if relevant, that can be taken pre-symptomatically to delay or prevent the onset of disease. And, you know, as we look at -- and in answer to the question about the usefulness of this data today, you know, first of all, when we look at some of the data that we have back from our early participants early on as we developed the product, nearly half of the people who got -- 46 percent of the people who had gotten their results participated in our study indicated they had made a change in their daily life as the result of having this information. Their genetic information does in fact create a tremendous teachable moment for people that can lead to changes in diet, exercise, visits to the doctor, working with their doctor to look to see whether they needed any follow-on tests or changes in medication or anything of that nature. It causes, you know, people to think when there is an issue that’s identified and causes people to think.
One of those stories I’d like to get a chance to also share is an early customer that we had who identified a high risk for colon cancer, previously had no known of the classic risk factors which, you know, you might look at a BMI over 30 which provides a predictive odds ratio of 1.7 towards 1.75 towards colon cancer, being a current smoker provides an odds ratio of 1.32 towards colon cancer, and having a family history of colon cancer delivers an odds ratio of 2.24. She had none of these situations and yet her genetic data came back showing a high risk for colon cancer. And when you look at the SNPs that we’re using for that condition, they have odds ratios of 1.47, 1.37, and 1.7 being just as good as any of the classic risk factors, and when taken together, provide a maximum potential odds ratio of 2.54, just as good if not better than a family history. So she took this information, talked to her doctor, her doctor in consultation -- which, again, is what we find our customers will do is if they find something they want to do something about, they’ll talk to their doctor about it, which is the right next step. And they decided to go ahead and do a colonoscopy where they found a 1.5 centimeter polyp which she got removed. And, you know, it is now, you know, going to be on a path of being able to watch for this in the future and, you know, the important thing is she was 39-years-old. Now, you know, the normal, standard practice in medicine would have been she would have not even been offered a colonoscopy until she was 50, and, you know, who knows what would have transpired in the following decade with the polyp that had been identified.
And it’s a story like that that we believe to Katy’s point, does help to provide an additional tool in the toolbox for a physician to look at the patient in front of them, to look at the information that they know about that patient, and the additional insights that genetic information can provide to determine an appropriate course of action, if any. And I think we’ve heard about a number of those here today.
And so I think, you know, it’s evident that the data -- given the nascent nature of this industry, or at least, you know, many of us here today that there are real examples of people deriving real benefit from these services. And absolutely, there are real issues that these companies need to grapple with; we are working together to grapple with those and to come up with solutions. But there is real benefit being delivered and real usefulness today.
MS. JOHNSON: Well, we’re going to open it now to questions from the floor. We can start circulating the microphone. Yes, back there in the back.
DR. LESTER: Yes. My name is Jeff Lester, I’m board certified internist; also I’m doing a medical genetics fellowship at University of Miami. One of the things I wanted to mention, we had been talking about doctors sitting down with their patients and talking to their patients. With doctors, you know, primary care doctors, pediatricians, and internists seeing 10 to 15 patients a day and what they do and how they manage this information, I think it’s important to remember and understand that doctors -- the internists and the pediatricians care about a couple basic things, you know, one thing is what is a diagnosis for this patient, what test do I need to order to get the diagnosis, what drug do I prescribe to the patient to make them better, and also, am I going to get paid for this for this service that I’m providing for them. Those are the key issue is that they want to know. And then, you know, another question that they have is, you know, if the person comes with a printout from a company and today their risk factor is a 25 percent lifetime risk of getting breast cancer, and then they get a bilateral radical mastectomy and then a couple, five years later they find out that their risk factor was only 15 percent -- am I going to get sued, you know? And what happens? Am I going to lose my practice? Am I going to lose my medical license because I’m sitting down and talking with them? You know, is the information I’m giving them good information and is it something that can be put out, you know, in the next 10, 15, 20 years for them.
So, you know, when somebody sits down and -- you know, we’ll have to make sure that the information that the doctors have is quick, easy, succinct information. I know that’s almost impossible to do at this point, you know, there are thousands of diseases out there, but having a one or two page synopsis for that patient, that disease, is what the primary care doctor wants in order to make sure that when they talk to somebody that they’re not spending an hour trying to figure this out, that they have something very concrete to talk about to make sure they’re effective and they can give good information. But, you know, doing it in an efficient and effective way.
MS. JOHNSON: You know, you’re absolutely right. The current system is set up that way and that’s the way it -- doctors have to work in order to get paid, in order to protect themselves from malpractice suits. So how do we get from here to there? You can't move from here to there with today’s level of knowledge. We just don’t know enough. So what happens is people will, through their own free will, decide to do this. And from what the scientists learn in the lab and what all the schools of medicine -- I mean, there are groups all over the country that are doing really remarkable work and it’s a credit to HHS incidentally that they even thought of having this meeting today. And in the fall in Utah, they’re going to get those communities together that are working on translating genetic information, genetic research into medical practice, and from all those things, we as a society will begin to know different things. And then we can translate that into payment policy and into, hopefully, liability law. But it is a process. And part of the reason electronic health information technology is so important is it begins to build those teams of communication. And the communication between multiple members of a team around this kind of issue is critical to a good outcome. So, you know, what you’re really asking is, how does a society go about making major change? And the policy makers don’t lead change, knowledge and experience lead change. So it is very important for us to do these conversations and for them to have good communication with the government, and for FDA and other regulators not to jump in there and regulate without a better understanding of what you’re doing. But fundamental -- and this is something that really is different about today’s world than it was 5 years ago or 10 years ago or 20 years ago -- the pace of change is so rapid that we have to accelerate the communication between the private-sector and what’s happening in this kind of an area, and the regulators and policymakers because otherwise they will make mistakes. They will regulate this the way they have regulated the world of the past, see? And so if you don’t want that model, then we do have to move. But we can't move without constantly keeping in mind exactly all the points that you have made about today’s world. But I have found -- and when you look at what’s happened in chronic disease management, you don’t see it very much because nobody reports on it. But anyway, the dynamic of the conversation that develops, both in those communities where electronic records are widespread and so you have a team sport here of caring for people, and also, where chronic disease has been the focus, it is a different dialogue. It is a different team. You see this in the big systems of Kaiser and Mayo and some of those. So that does have to spread but this conversation is part of that, and we can never forget the sort of now anachronistic barriers that have been put in place by the old system of illness treatment and by the old liability system that presented a different kind of thinking.
But there was another one down here and then we’ll go over there. Yeah.
UNKNOWN: Yeah, I have two questions. One kind of a more wacky one, one more serious. So you decide which one is which. So why have -- the first question is this: why have academic health centers stayed so behind the private-sector in terms of incorporating genetics into health care, particularly in the areas of risk, early intervention, as we said with the prostate situation here, and prevention?
And the other question, since everybody’s using this nice case studies, I’m going to give another case story for 2018. So Mary (phonetic) goes to a dinner at her boyfriend Joe’s (phonetic) house. She gets there, the young brother is autistic, an uncle that’s there at the dinner had colon surgery at a relatively early age with no symptoms, and there is a second cousin once removed who is bipolar. So they are driving -- he is driving her back home, and then he asks, you know, about the family; she says, “Oh, they are wonderful people. I like your father and mother,” et cetera, “but I’m 37-years-old, I don’t have a lot of time to waste here, and if we’re going to go on dating, I want to see your Navigenics profile.” [LAUGTHER]
So since the sea of Navigenics is here, what should Joe say to Mary? [LAUGHTER]
MS. JOHNSON: Mari?
MS. BAKER: And that one wasn’t the wacky question? [LAUGTHER]
Well, I think that part of what you touch on is this notion that at some point, you know, I think the point has been made here today that at some point out in the future, you know, this stuff is moving along. We will have these sort of insights into, you know, what’s in our genes and, you know, hopefully, you know, if a move was made on colon cancer, it would be because, you know, a physician believed that that was the right thing to do for a -- to do any sort of -- any surgery on anything, obviously involves a physician that requires a thought process that says this is an appropriate step to take.
The, you know, there’s a wide range of, you know, issues that go on, including -- I think back to the prior comment about, you know, the, you know, not only do we have to get people on electronic health records, which still are not uniform and universal in this country, but we also have to develop some of the decision support systems that start to take the information in those health records, combine it with family history, and combine it with genetic information so the decision support systems are in place to be able to give those insights and red flags, or, you know, questions for physicians to know and to think about in interacting with their patients. And so these are all things that have to be put into place. I think the question of what Joe answers back to Mary is much more fundamental and has to do with the reasons why even though we meet our in-laws, we still get married, and it probably falls in the similar bucket.
MS. JOHNSON: Yes.
MS. AVEY: I’m now moved to tell my own personal story, which is very briefly, that according to 23andMe, I have a very low risk of colon cancer. And I put this in the 23andMe blog for what it’s worth, yet I happened to know that I probably have a rather high risk; my father continually has polyps and I’ve been tested a few times, so -- three or four years ago I went and discovered I, too, have a polyp. It was a flat one and it got removed.
So I know for practical purposes, I probably have a 98 percent risk of dying of colon cancer if I don’t continue to get checked and if I don’t die of something else first. And the point of this story and the point of me putting it on the 23andMe blog is what really needs to happen is people need to understand statistics and probability and risk. And that’s really, really tough. The way they’re going to understand it is if you have the early adopters, the people who are really interested, the guys with $1000 and more, doing this now and understanding what it means, which in many cases is very, very little. As someone said, the difference between a 52 and 42 percent risk is - - it’s meaningful but not for the individual because your risk is either 100 percent or 0, but you only know that after the fact. And for people to understand what this does and what it doesn’t do, for them to understand that it’s going to help them probably pick better drugs and better treatments, but that a risk is only a risk, you -- nobody can tell them if they’re actually going to get it or not unless it’s something that’s completely predetermined. That’s really what we need to teach society, and I think the way we do this best is by having these discussions not just among people who already know all this, but in the pages of the New York Times, in public hearings with the state of California, in not just the New York Times but the, you know, the (inaudible) Gazette and in People magazine let’s have some intelligent discussion of the celebrities risks, and then people will be able to apply their own lives just the way they understand football scores. It sounds intuitive when you talk about football, it needs to become that way -- that genes.
MS. JOHNSON: Yeah. In other words, in a new arena, remember what may look like danger is opportunity, so New York and California are opportunity for this industry. Ronni.
MS. SANDROFF: Yeah. I just wanted to say that I think what would really be exciting for consumers would be to get a genetic test and find out that you didn’t ever have to have a colonoscopy. And that there was something you didn’t have to do, and you didn’t have to worry about. And I think people kind of -- that’s kind of the implied promise. It’s not, you know, if you’re just going to find out -- everybody’s shaking their heads who knows more than I do --
MS. JOHNSON: That’s wrong.
MS. SANDROFF: -- so they’re probably -- so that’s never happening, right? You’re only going to find out you have more things to do.
MS. AVEY: No, I think that that issue is that these tests all try to be clear if there’s environmental impact and there’s genetic impact. And that -- I think that’s, you know, one of the reasons we’ve all tried to, you know, present information in a way that helps people to know even if there is a lower genetic based risk, you still need to pay attention to the other risk factors and to the other things you need to do because there’s two pieces to the equation.
UNKNOWN: Speaking off microphone.
MS. AVEY: Yes. Is this on? Yeah. Just two comments really quickly. One, there is some times relatively good news. For example, with BRCA, somebody with a known family mutation and the offspring or sibling does not carry that same mutation, that is really good news. But those are rare and few between in genetics, I understand.
UNKNOWN: Speaking off microphone.
MS. AVEY: Well, they could still, but they won't get the same one that their mother died of. Okay. That’s a big deal.
Unknown: Speaking off microphone.
MS. AVEY: Just the average risk. So -- but the other thing I wanted to say, I was at the U.K. Human Genetics Commission last week on the same topic that we’re all talking about on the voluntary code of practice for direct-to-consumer and I noticed that day in the British press that the first couple in England to have PGD for BRCA had happened. And, I mean, I don't know that that made it in the American press, but that’s a big deal. And that is -- let’s go fast forward on your case study -- where’s it going to go? Well, actually, it will go to PGD.
MS. JOHNSON: Over here.
MR. RACKOVER: Mike Rackover from the American Academy of Physician Assistants. I just think it’s important to -- that when we talk about patient care that we do include nurse practitioners and physician assistants. Our organization, we’ve partnered with the genetics community and the National Human Genome Research Institute to institute education that physician assistants will be educated in the genetics that we’re talking about today.
I also have other concerns here, but we’re forgetting about the other 40 to 50 million people that don’t have health care insurance. We have to balance out the information that you’re talking about today in every day reality of patient care. And we’re moving very quickly into forgetting about the challenge of everyday medicine. The Navigenics -- the type of patients that are now getting direct-to-consumer testing are typically an educated population and it’s a biased population in the type of information that they’re going to get. So, I mean, what do we do for the patients that obviously that we see that cannot afford these type of testing; what do we do with these type of patients? We can't ev -- we write prescriptions and they have to -- they can't even afford the prescriptions that we write. So, I mean, we’re -- it’s a bigger challenge here. And in fact, I realize the importance of what we’re talking about today, but we’re still forgetting about the everyday population that comes to see us.
MS. JOHNSON: Yeah. Yeah. Don’t forget, though, that currently risk does drive payment policy, so we pay for mammograms with women with a history of breast cancer in their family and some other things. I mean, it’s very embryonic, you know, and it was a different kind of analysis at risk, but the more you begin to know about genetics and the more the testing turns up more increasing the uniform results, I mean, that will reflect itself in payment policy.
MR. RACKOVER: But my specialty was oncology. When we first started evaluating patients that have cancer, it was obviously imaging, x-rays, CT scans, MRI. Now every patient gets a PET scan. So we’re now spending $5000 to $7000, $8000, for every time a cancer patient is diagnosed. It’s -- there’s something wrong with the system. Nobody questioned the fact of the integration of radiological imaging in the treatment of cancer -- or evaluation process. Here, we can't get passed genetic testing. The, you know, we’ve been spending years sitting -- hearing all these committees being able to talk about genetic testing and the treatment of cancer certainly has moved to the cost of what it costs for cancer, it’s huge. But we can't do basic genetic testing.
MS. JOHNSON: but in those numbers of years, we have learned a lot about where the costs are located in the system, and if we could begin to weed those out and move them and use them -- use modern science to move us forward to -- so -- it’s not hopeless, but I’m -- I certainly recognize that today’s system doesn’t differentiate between appropriate care and inappropriate care or needed care and unneeded care. (Inaudible) --
MR. RACKOVER: Another thing, we have to pass a law. We have to pass a law to basically get some type of preventive testing done.
MS. JOHNSON: Well, it shouldn’t be that way. That is the way it’s been, but see, as you -- and if you develop a health system, it won't have to be that way anyway. I won't -- we have an illness treatment system so then we have to make special provision for prevention. But as you change the laws and the systems, you can get away from that.
MS. JOHANSEN: Can I just make a --
MS. JOHNSON: Yeah.
MS. JOHANSEN: So I -- can I just make a quick comment about a few of the questions that I have heard?
MS. JOHNSON: Sure.
MS. JOHANSEN: I think there have been some really related questions, and Rocky’s question just sort of brought it up again. And that’s that, you know, there’s a question about why there is really slow uptake of genetics in some medical centers and there was also a comment by a physician saying that they’re very time constrained and don’t have time to do this. And I think some of these questions are actually --
MS. JOHNSON: (Inaudible).
MS. JOHANSEN: -- answering each other. I think there’s been very slow uptake, number one, because physicians don’t have time to add -- especially primary care physicians, are so time constrained and don’t have time to add another, sort of, fancy, new test to their limited five minutes with patients, and are not going to do that until they see evidence that that test actually impacts clinical utility. But that evidence isn’t quite there yet; there might be some hints that that evidence is there, but until that is really shown, I think that might be a shove in the right direction for physicians to start using that information -- the genetic information. And so I think Rocky’s point also about, you know, who’s going to pay for patients that don’t have health care; that’s another question that physicians have to confront when they’re -- when they think that a genetic test might be appropriate for their patient. How are they going to say to their patient, “Well, I think you should get this test, but it’s going to cost you $500 and I don’t know where you’re going to et that money.” That’s another reason that I think there’s been some slow uptake.
MS. JOHNSON: What about medical education? You certainly have a hand in that from the AMA. Do our medical schools even -- are they even training our doctors in how to use this information?
MS. JOHANSEN: That’s a question. Right. The - -
MS. JOHNSON : The answer is pretty much no, isn’t it?
MS. JOHANSEN: Right. Well -- there are movements. Right. I mean, there are movements in some parts of the health care world, like the physician assistants and the nurse practitioners have been very good about integrating some genetics education into their curriculum. But medical school education is a bit harder to crack. The exams, the qualifying exams, and on other exams that are along the way are set very far in advance and it’s hard to change the questions on those and because it’s hard to change the questions on those, it’s hard to change the curriculum that is taught in order for the students to be able to answer those questions. And that is absolutely something that needs to be addressed.
MS. JOHNSON: We can change that if we choose.
UNKNOWN: I’d like to make a series of statements and see if the panel would like to comment on. It’s sort of like a sweeping generalization of the field of personal genomics, and see if you all agree with my assessment or not. And I say that with passion because I don’t want the field of personal genomics to suffer the same fate as total body scans had a few years ago when, you know, there was a craze, people went in, they had all kinds of procedures -- some of them necessary, some of them are not. We’ve heard some anecdotes about the usefulness of this information both good and possibly not that good in terms of the potential harms and benefits.
And so the way I look at the field right now, it’s in a state of flux. We’re in this teachable moment where what we need to do in addition to discovery research of finding new genes and genetic risk factor, is to do the translational research to allow the kind of -- that kind of information from both clinical validity and clinical utility perspective to be shown, you know, the balance of harms and benefits, do the clinical trials that need to be done. Unfortunately, this will take time and it will take money to do it. But we’re already spending billions of dollars to do the $1000 genomes and, you know, if the public and the private-sector can come together to do translational genomics and in the sense to allow us to do the kind of research that shows really the added value of genetic information in a health care delivery system that is already crumbling under it’s own weight, I mean, we might be suffering the same fate of other new technologies. So, I mean, that’s sort of a plea that I have. I don't know if people agree with that assessment.
But in the meantime, clinical validity is low because it’s probabilistic information, no matter how many new genes you add, it’s still going to be, you know, 51 versus 47 percent. And it could be misleading, like we’ve seen, because some information is not in the genome so the -- we need to look at the benefits, sort of, the balance of harms and benefits. And we all think that there could be benefit that will come out of this, but there could be some real harms, especially if implemented on a population basis.
MS. JOHNSON: Comment?
MS. JOHANSEN: I don't know where you draw the line in terms of determining when this technology is available for primetime, but I do think that if you offer it prematurely when there’s a lot of flux and a lot of variability in terms of what the results might mean, then you stand to lose being able to get consumers to buy into the technology. So if you use the information prematurely and you get a lot of results that change pretty dramatically over the next five or ten years, then people are going to start to think that this is not good technology and they might not use it in the future when it is good technology. So I think that’s my cautionary note, I think, and it goes along with what you’re saying.
I mean, right now is probably the time to build the infrastructure, find out what the questions are -- I’m not saying, “Don’t do it,” but people need to know what they’re getting into and what the limitations are, and it needs to be presented in multiple different ways because even if you think that you’re presenting information in a way that suggests that it’s probabilistic, numeracy in this country is horribly low. So you -- we just have to be very careful in how we proceed, and I do think that we need to keep in mind that if we lose consumers -- and when the promise is met in the future, they might not want to use this information. And that would be a tragedy because I think in the future this information will help cut health care costs, will help us target health care, and there is tremendous promise.
MS. BAKER: I want to just add a note on that too, which is, it’s a little bit of this, you know, discussion of, well, how can we make this more accessible to people? But yet we’re not sure that broad populations know how to deal with statistical data and make these tradeoffs. So one might argue that for right now, having these services be at a relatively high price allows the opportunity for, you know, learning and knowledge and education among an educated audience who is paying for this out-of-pocket. And for, you know, for us to be able to learn these issues as we move along, and I think, you know, one of the things that was pointed out earlier is, you know, an example of somebody having taken multiple tests from the three different services and getting different answers. Well, it’s not that people are calculating things differently, which is certainly the case, but underneath there, you know, right now people are using different SNPs to determine, you know, results for a condition. Those things clearly need to be standardized.
And, you know, the ability to be able to look at this information and see these differences enables us all to work together to come up with these industry best practices and to be able to move forward on this. You know, I think that, you know, the question on the translational genomics, we would love to see that funded.
We would love to see that worked on. We would love to see the clinical studies done around all these points. But we shouldn’t forget that medicine changes, as well. You know, it wasn’t that long ago when in some case -- right, doctors were advertising to -- in smoking ads, right, for cigarettes. And look how long it took us to decide that smoking was actually bad for you. You know, it took a long time for mammograms to get reimbursed. You know, it takes -- some of these things take a long time, and medical knowledge changes, science knowledge changes.
It’s something that’s a fact today and will continue to be the case with this. This makes it more transparent, this, you know, these sort of services help people keep up to date, you know, with this information. And, you know, that’s -- you know, I think there’s a value in that for people to know that, you know, there will always have the latest rather than being subject to things that might be 20 or 30, you know, tested, or 20 or 30 years old and haven’t been updated in time.
MS. JOHNSON: The best protection against that danger, which is real, I would say, is for the private- sector and the public-sector to work together more aggressively than we have in the past when new things came forward, and in a more kind of intelligent fashion so the industry, if they have any sense, won’t indulge in an explosion of direct-to-consumer advertising, particularly until the -- I mean, we use the New York and California experiences as an opportunity, an opportunity to talk about what they’re doing, an opportunity to work together to get more standardized tests and talk about the need for that. You know, transparency and openness and directness will save this industry, but if there isn’t that kind of openness, it well erode trust and it will all -- I mean, we’re talking about -- among enlightened people, we were talking about the top level of consumer users. Not only can they afford the $1000, but they’re interested and they care about their health for the most part. But if you talk -- remember all the people out there who would be panicked if they knew they had any propensity, any risk whatsoever toward any serious disease. And we aren’t prepared yet to differentiate between levels of risk. So there’s a lot of public educating to do, and what could be a better forum than the two biggest states in the nation at each end of the country as a way to talk about this and begin to think publicly -- help the public think through what do you gain and what are the risks you take? And unless the private-sector better understands this issue of public education, then in today’s world and with it’s, sort of, volatility and the simplification of messages that’s typical of every kind of media, we will lose the opportunity in this area and it won't come back for five or ten years. So we have time for one more question; I’m being signaled. Is there -- there it is.
UNKNOWN: Speaking off microphone.
MS. JOHNSON: Well, two more if you’re short. We’ve got five minutes.
UNKNOWN: I’ll be quick. Well, it’s about medical education, so, some data. Through the end of 2005, 15 percent of medical schools, as reported by their deans, said they teach no genetics. And of those who teach genetics, 17 percent teach less than 60 hours throughout the four years of medical school. So you could argue good, bad, or indifferent, but the key piece is what Mari said, which is it’s a changing field. And one of the key issues that was meant as I understand to deal with the changing field is continuing medical education. And 48 states, I believe, have CME requirements on a regular basis. Over the last ten years one of the key things that I’ve -- AMA and others were involved in, is requiring most states have 5 to 10 percent of those CME hours have to be on risk stratification, to look for abuse or other challenges in the home. So what about the idea of using - - and I clearly have a point of view here, but the idea of using this CME process as one that acknowledges that the world is changing so we can't teach everything, you know, in four years or two years of medical school and expect those physicians to be up to date 10, 20, 30, 40 years later, but using a system that already exists with potentially some requirements around a percent of that is on genetics or emerging technologies, so -- because as I look at -- I guess this isn’t short -- but as I hear something that came up on every panel today, it’s doctors today, health care providers starting with physicians, need to be able to lead the way because whatever you get in a personal genomics, you can't do your colonoscopy yourself. You can't write the prescriptions for the most part; you need to go through your physician. So one organizing issue that I saw is that we need to educate our providers in a better way for any part of this industry to become fully transparent.
MS. JOHNSON: (Inaudible) so many other things, we know more about this than we think; Marshfield is a big system, they do translational research, they -- every year they set aside a day-and-a-half for the education of their physicians and what they’re doing, and I’d be surprised if some of the other big systems don’t too. So we could inject that into the medical schools more rapidly, if we cared to.
UNKNOWN: I just want --
UNKNOWN: (Inaudible).
MS. BAKER: -- one comment on that because I think that’s exactly the right point. And, you know, we have attempted to make a tiny step, you know, in that direction. But that one tiny effort -- I think that the results are emblematic of the interest and gaps that exist in this space. So we funded Medscape to create a CME course in personal genomics and in clinical practice. And, you know, they went out, found somebody to develop the course, and I think it’s 25 CME credits, so it’s not huge, but it’s, you know, reasonable, it’s something. And in the first -- so I have data through the end of May and it went out I think in late -- like the last couple days of February, so -- March, April, May -- three months of data. Over 5,000 Medscape members, health care professionals, read the course, and 2,500 completed it for CME credits. And I, you know, it is acknowledgeable a very small, simple first step, but I think it shows the amount of interest among health care professionals in absorbing this information, learning about this information, and I think a lot of the benefits of an online venue, too, and make it easier for people, which Medscape is, is an online venue for taking -- getting CME credits, to be able to have access and get that learning in the time they have available.
MS. JOHNSON: Excellent. Last question.
MR. MILLER: Just a quick question. My name is Paul Miller, I’m a law professor and a professor of disability studies at the University of Washington in Seattle, and also a member of the Secretary’s Advisory Committee. I wanted to jump in to sort of a side conversation we had a couple of conversations ago about PGD and sort of where all this information is going. I think one of the underlying -- and sort of put on the table -- an issue; the underlying, sort of, assumption with all this information is that information is good and that people that we’re talking about conditions that either today or in the future, somebody can do something about, that these are sort of health outcomes. There’s another perspective from the disability point of view that people with disabilities -- that parents are going to -- or others -- are going to begin to look for genetic anomalies, genetic disorders, and sort of, take those out of the system to basically use PGD, to use these genetic markers to eliminate people with or to reduce pregnancies of people with disabilities. And I think that that’s something that both the genetics community, the physician community, and others interested, really need to be sort of aware of and to sort of think about the impact on people with disabilities, both as members of society, the move of and support of social services and government services to disability programs and the relationship between PGD and genetic anomaly identification, and pregnancy and birth. I think it’s an important issue and I just wanted to put it on the table.
MS. JOHNSON: Thank you. And thanks to our panel for bringing their rich experience of consumers to the table as we conclude this panel -- this day’s -- this half-days discussion. Thank you very much for your past work and your continued contribution. [APPLAUSE]
CONCLUSION/FINAL Remarks
We’re going to wrap this up very quickly. I’m going to make a few comments and then turn it over to Dr. Greg Downing; he started the conference, and he’ll end the conference and we’ll be on our way. This was to look at the future in this field and I think very clearly as we talked through this day, much of our future has arrived, it’s just lumpy. It just hasn’t arrived everywhere at the same time. There are elements that will affect our profession for 20 or 50 years; we see them, we know what they are. And then the day, I think, was really centered around, first, a convergence of thought -- that that’s a fairly desirable future from the point of view of consumers. From the potential of genomics, that there was divergence among the group over issues about regulation, oversight. What drives this? Is it the research and science that should drive it? Is it the market that should drive it? Is it both? What is this enterprise? I heard recognition on several things. One is that risk communications and effective communications, not only within the profession but with patients, will be key to whatever success comes out of our efforts; that there are very divided views on privacy, and they are very grate concerns over both privacy and the reliability and integrity of information.
There were additional concerns about the engagement of health care providers. How do we bring this future across our health care establishment? And there was a big question, what’s good enough? When is something good enough to be in the market and when is it not? I heard that least through this conversation all day. If I could sum this up, I would say that what we have here is a clear and predictable evidence of growing pains for a science moving very fast, turning pharmascience into a young industry, and trying to figure out how to handle the risk, the science, the motivations, the markets, the trust that have to be successful and have to come together in a system for all of this to be the benefit to have the potential that we all described at the beginning and thought we saw here. And I’m sure and still think we do.
Two observations: one, I am proud of my profession. I’m proud of my fellow health care providers and the scientists and the entrepreneurs in here who have all come together and had a very frank and open debate with a great deal of passion that’s sometimes sharp differences of opinion, but all done in a manner of most admirable mutual respect. I asked for no hitting and there wasn’t any. It just -- you followed orders very good. I’m so proud of you.
I did not hear the word ethics mentioned once. I heard regulation and I heard governance and I heard market and I heard the science and I heard the facts and I heard -- I never heard anybody talk about the ethics. And I think sometimes -- and I just throw this out for thought. Sometimes we have a tendency to take a scientific advance, make it work, and then we put it in a market or we take it to people, and then after awhile, then we start figuring out the ethics. But we often don’t figure out the ethics first, we often figure out the ethics after the governance has come along and been the third thing that’s kind of come in the wake of sometimes not thinking these important things through. And we are now reaching a point that the complexity and the power of our science is so overwhelming, that it almost butts up against the level that it begins to make a difference as to what we are as human beings. So I think as this community goes forward, the idea of developing an ethical framework, as you have developed these many other frameworks around these other issues, might be something to think about. Finally, all the thoughts here have been captured. We set out to have a conversation, we did that; I think my analogy to the Manhattan Project and the importance of the dialogue was not off at all. In fact, I’m more convinced of its appropriateness now than when we started. I think this will be a very great value to everyone concerned, and let me ask you to give one more round of applause to Greg Downing and his team who put this on. And I will turn the floor over to Greg for his final remarks. [APPLAUSE]
DR. DOWNING: Thank you, Michael. I think we have a small team of vested futurists within the department that worked over the last year to share ideas about how to facilitate a discussion that we think probably for everyone is at times uncomfortable, and perhaps that’s where the dialogue ends today is still with an unease but more reflective of an appreciation for other viewpoints that are exhibited here. And I’m sure if we came back a year from now we’re going to know a lot more about this terrain. I just wanted to finally thank Mike for helping work with the group that came together today. Obviously a lot of thought given to your remarks, and the appreciation that we have for being able to have a candid discussion about our own viewpoints is an important thing to start with. I think from the Department’s viewpoint there is a lot more work to be done and we’ve certainly been leaning on our Advisory Committees in a variety of different ways these past several years to help develop some of the boundaries about which the conversation and the actions that take place go forward. We’ll do that in the form of a summary from this meeting and it’ll be posted on the website and certainly the materials from this will be available to those who wish to utilize them for their work going forward.
I want to thank all the speakers again, and from Rick and everyone at the Department, we appreciate everyone’s engagement in this and hope that it builds on some of the foundations here about openness and transparency and the engagement that all of you had to ask yourselves the critical questions about whether we’re doing the right things in the right ways for the people that we’re all here for. So again, thank you for your time this afternoon. We’ve enjoyed the opportunity and hope that this has been a value to all of your efforts here as well. So thank you. [APPLAUSE]
Presentation by Steve Bodhaine
Presentation by Steve Bodhaine, MBA
Group President, Research/Product Development, Yankelovich
Prepared for:
Personalized Health Care Initaitive Workshop
July 7, 2008
Prepared by
Yankelovich, Inc
Disclaimer: The findings and conclusions of this presentation are those of the authors and do not necessarily represent the views of ASPE or HHS.
Background, Objectives and Methodology
Background
- The Department of Health and Human Services(HHS), Office of the Assistant Secretary for Planning and Evaluation(ASPE) is holding a workshop called "Understanding the Interest and Needs of Consumers in the Use of Genome-based Health Information Services" to discuss consumer demand for an interest in services that provide genetic testing/screening.
- This workshop will include major stakeholders in the field of genomics, including clinicians, genetic counselors, and consumer advocates. In order to provide specific consumer response to this issue, TSC a division of Yankelovich conducted qualitative primary research on the topic.
Objectives
In a qualitative fashion, the research answers the following key questions related to attitudes toward genetic testing among consumers:
- Awareness & Understanding of Genetic Testing
- What do consumers know about genetic testing?
- How do they define genetic testing?
- What words/concepts do they associate with the concept?
- Is their overall evaluation of genetic testing positive or negative?
- In what circumstances do they see genetic testing being used?
- Who wants/needs testing? Why? Where?
- How knowledgeable are they about online genetic testing sites?
- What do consumers know about genetic testing?
- Personal Interest in Genetic Testing
- What is the level of personal interest in testing?
- And Why?
- How would they react to positive /negative results?
- What is the level of personal interest in testing?
- Concerns about Genetic Testing
- What are the biggest concerns consumers have about genetic testing?
Methodology
- Interview Format: 90-minute in-depth interviews with consumers
- Sample: Total of 9 consumers interviewed
- Interviews were conducted in Raleigh, NC on June 25, 2008.
- A diverse group of consumers (age, gender, education, HH income) were interviewed
- Qualitative Research Caveat
- As with all qualitative research, these findings are based on a limited number of consumers. No assurances can be made that their attitudes and behaviors are projectable to the US population of consumers.
- Accordingly, the use of the terms "many," "majority." "some," etc., in this report should be interpreted directionally, with the understanding that they are descriptive of the study sample.
Key Takeaways
- Familiarity with the concept of genetic testing is limited.
- The majority of consumers say they have heard of genetic testing, but do not know much about it.
- Most can provide a definition for genetic testing, but aren't sure what it involves, how the information is used, what the results would look like, or how much it would cost.
- For many, they have neutral opinions about the genetic testing because they feel they don't know enough to fully evlauate it.
- Consumers are largely unaware genetic testing information companies exist that provide services via the Internet.
- The majority of consumers say they have heard of genetic testing, but do not know much about it.
- The healthcare professional is desired - and assumed to be - involoved in the infomration gathering, testing, and analysis processes.
- Most consumers say they would consider genetic testing, especially for conditions where they could affect their future risk by taking action.
- Though most are able to define what genetic testing is, consumer knowledge drops off around how genetic testing information is used.
- Most are open to genetic tests being perfomred, but have concerns about "what happens next?"
Hopes
- Understanding disease risk
- taking preventative steps
- Planning for the future
Concerns
Ability to understand results:
- Delivery of information
- Interpretation/explanation of results
Actions taken after testing:
- Risk of discrimination
- Religious /Ethical decisions(e.g., abortion)
Detailed Findings
Awareness and Understanding of Genetic Testing
Familiarity with Genetic Testing
- Consumers have heard of genetic testing, but feel their knowledge is limited.
| I have not heard anything about genetic testing | 0% |
|---|---|
| I have not heard of genetic testing, but I do not know much about it | 56% |
| I know quite a bit about genetic testing | 44% |
| I know quite a bit about genetic testing | 0% |
| I consider myself an expert on genetic testing | 0% |
Feelings Toward Genetic Testing
- Many consumers say their overall feelings about genetic testing are neutral, largely because they don't know enough about it to have an informed opinion.
| How would you rate your feelings about genetic testing overall? | |
|---|---|
| Extremely positive | 31% |
| More Positive than negative | 22% |
| Neutral | 56%1 |
| More negative than positive | 11% |
| Extremely negative | 0% |
1."I would have to hear more about it"
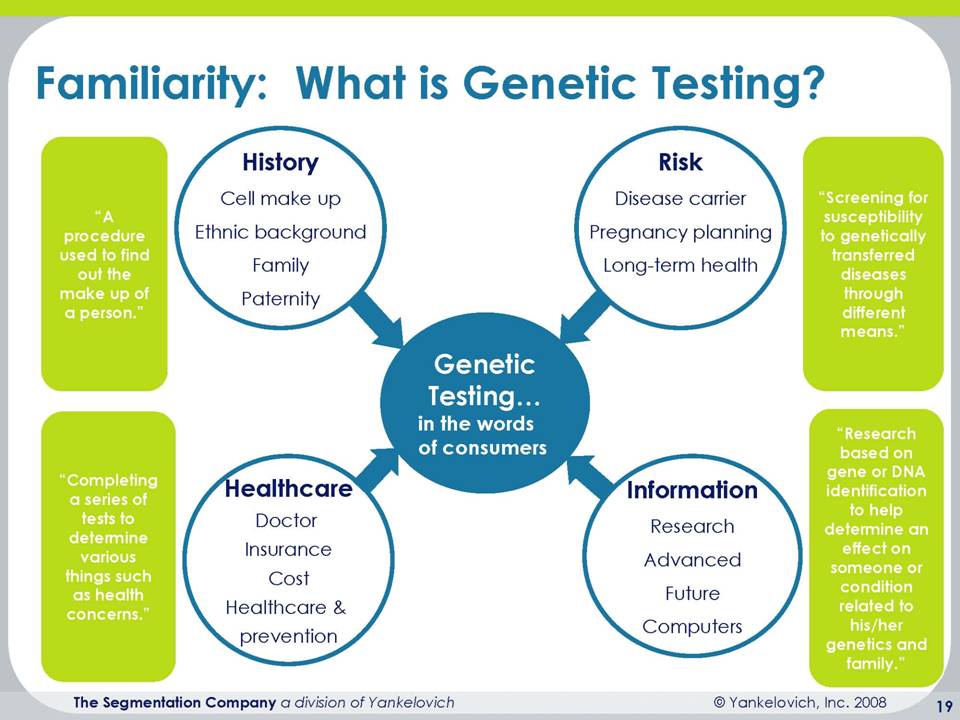
Familiarity: What is Genetic Testing --- Four green rectangles with text inside:("A procedure used to find out the make-up of a person");("Completing a series of tests, to determine various things such as health concerns");("screening for susceptibility to genetically transfered diseases through different means");("research based on gene or DNA identification to help determine an effect on someone or condition related to his/her genetics and family"), counter-clockwise, respectively . Inside the four triangle diagram are four circles with text inside:("History -cell make up,ethnic background, family, paternity"); ("Risk - Disease Carrier, Preganancy Planning, long- term health);("Information- research, advanced, future, computers);(Healthcare --doctor, insurance, cost, healthcare & prevention), clockwise, respectively.
| Who is likely to get genetic testing | Why do they need genetic testing? | Where do they go to have it done, and by whom |
|---|---|---|
|
|
|
Familiarity: Direct-to Consumer Services
- None of the participants in this study were aware that they could currently purchase genetic testing from an online company
- One respondent, though, was not surprised because "you can buy everything else online".
- They also had no idea about cost for the test, with estimates ranging from $20 to $2500 for testing for one disease.
- Most were skeptical of the accuracy of the test
- Worrying that their test would get mixed up with someone else's
- While most consumers said they would consider having the test done online, they remained skeptical
- And all said that if the online test came back positive they would go to the doctor to take another test
- All consumers also said that they would take their results to their doctor for interpretation
Perceived Benefits of Genetic Testing
- Consumers say that genetic testing is valuable because it provides information about their health, and gives them the opportunity to seek treatment.
- In contrast, a negative test result is not the end of the story. Many said that just because a test is negative does not mean that you shouldn't continue to have screenings and check-ups because "the test are not a guarantee -you could still get the disease" .
| "Genetic testing is valuable because....Mean, with scale:1 (Strongly Disagree) to 6 (Strongly Agree)" | |
|---|---|
| It can provide relief from uncertainty about the risk of getting a disease (you know your risk) | 4.9 |
| A negative test result can eliminate the need for unnecessary checkups and screening tests | 2.8 |
| A positive result can direct a person toward available prevention, monitoring and treatment options. | 5.4 |
Knowledge about Test Meaning
- Most consumers understand that genetic testing will tell them their risk level of developing a disease rather than a certainty of developing that disease
| If you have genetic testing for cancer-- which of the following describes what the test will tell you? | |
|---|---|
| Whether I will have a higher that average risk of developing cancer in the future --89% | Whether I will definately get cancer in the future-11% |
Desired Information Before Testing
- Above all, accuracy of the test was the most important information for consumers.
- Consumers also wanted to know about basic logistics of the test-- what is the process? Where would I go for the test? How much time would it take? What would it involove?
| If you were looking for information on genetic testing, how important is each of the following types of information to you? Mean, with scale: 1(Not at all important to 7 (Extremely important) | |
|---|---|
| Accuracy of the testing services | 6.3 |
| Costs | 5.5 |
| How a clinician would utilize this information in decision-making | 5.2 |
| Scientific basis for the testing | 4.9 |
| How information will be used (privacy) | 4.8 |
| Risks from testing (discrimination by others, insurers, employers, etc.) | 4.2 |
Information about Genetic Testing
- Most consumers believe that information on genetic testing is out there, and their go-to sources for information would be the Internet or their doctor.
| When it comes to fiding information that would help consumers use genetic information to improve their health, do think such information is: | |
|---|---|
| Easy enough to find | 44% |
| Somewhat difficult to find | 44% |
| Very difficult to find | 0% |
Where would you go to find information about genetic testing?2
- "the Internet"
- "a reliable approved website"
- "WebMD"
- "NIH website"
- "Google search"
- "someplace that is not trying to profit"
- "my regular doctor"
- "library"
- "a college, university, or other non-biased source"
note-size of lettering reflects more mentions, such that the internet, with largest font, was most often mentioned.
Information Sources
- Consumers believe that doctors, other medical professionals and genetic counselors should be involved in developing public information about genetic testing.
- In contrast, most consumers did not expect (or want) the government to be involved in developing information.
- However, if asked whether the FDA should be involved they were somewhat more open to the possibility.
- The concept of "government" did not elicit favorable reactions.
| Which of the following sources should be responsible for helping consumers be developing public information about genetic testing? | |
|---|---|
| Doctors & other medical professionals | 78% |
| Genetic counselors | 56% |
| Companies that provide genetic health information services | 44% |
| The government | 22% |
Regulation & Industry Standards for Testing
- Consumers wanted a "reputable organization" to create an industry standard to help consumers identify which companies were trustworthy for gentic testing
- The organizations mentioned included:
- FDA
- AMA
- Independent group of doctors
- Well-known medical journal
- "I would trust medical professional more than the government"
Detailed Findings
Personal Interest in Genetic Testing
- Consumers want to know about their disease risk, particularly if they could take steps to lower their risk.
- While interest was lower for learning risk for diseases for which there was no action, the majority were still interested.
- Interest was lowest in non-health related information.
| Which of the following types of personal genetic information would you be interested in?: Information that tells you..... | |
|---|---|
| Your risk for a specific disease (with or without your doctor knowing) | 89% |
| Your risk for a disease that you could do something about-- that is, by knowing that you are at risk you could alter the onset of the disease by using a medical intervention or making lifestyle changes | 100% |
| Your risk for a disease you could not do something about---that is, even knowing your risk you could not do anything to change your risk level, but it might influence your life decisions (such as having children or retirement planning) | 67% |
| Your ancient ancestors (which portion of your DNA that came from a particular region of the world) | 44% |
| Your physical traits (such as eye and hair color, type of earwax, height, caffeine metabollism, baldness, or odor detection) | 67% |
- Consumers were most interested in personal testing for cancer and heart disease.
- The general sentiment was that they would want testing for more "serious" diseases.
- In contrast, few saw need for testing for obesity because it was "about personal control" and "I can just look at myself to know"
- None of the participants in this study had undergone genetic testing for any reason, though several knew children who had been tested.
| Which of the following health conditions would you consider getting genetic testing for (or have been tested for) in order to determine if you are at increased risk? | |
|---|---|
| Cancer | 100% |
| Heart Disease | 78% |
| Diabetes | 67% |
| Alzheimers | 44% |
| A mental health condition (e.g. depresssion) | 22% |
| Obesity | 22% |
Genetic Testing & Family
- Consumers say that they would want to share genetic testing information with family members if those individuals might be at risk
- Likewise, they would want family members to tell them if they discovered an above-average risk.
| Mean, with scale: 1 (Strongly Disagree) to 6 (Strongly Agree) | |
|---|---|
| If I took a genetic test and discovered I had a higher-than-normal genetic risk for a disease, I would tell my family because they might have the same risk. | 5.3 |
| If a family member discovered that they had a higher-than normal genetic risk, I would want them to tell me. | 5.7 |
Concerns about Genetic Testing
| How concerned are you about each of the following aspects of genetic testing? Mean on scale: 1 (Not at all concerned) to 6 (Extremely concerned) | |
|---|---|
| Having standards and quality control measures available when interpreting generic test results | 5.3 |
| The ability for people to understand the results of their genetic tests | 5.1 |
| The potential for discrimination for those who have higher-than-average risk for particular health conditions (e.g., by insurers, employers, courts, schools, adoption agencies, and the military, among others) | 4.9 |
| The ability for healthcare providers (e.g., doctors, nurses, etc.) to understand the results of genetic tests | 4.9 |
| Fairness in access to genetic services (e.g., if cost is high then services may not be affordable to all) | 4.9 |
| The usefulness of genetic information (e.g., the ability to use the information to improve medical care) | 4.8 |
| The impact it would have on a person who learns they have a higher-than-average risk for a health condition that cannot be medically treated | 4.8 |
| Privacy and confidentiality issues related to finding out my personal genetic information | 4.7 |
| The impact it would have on how a person feels about him/herself (by knowing their risk levels) | 4.2 |
| The impact it would have on how others in society may fell about a person (by knowing their risk levels) | 4.1 |
| Effects of genetic testing on people's reporductive choices (i.e., they may choose not to have children if thay are predisposed to certain health conditions) | 4.0 |
Future Research Directions
Consumer-oriented research
- Segmentation to identify high-targeted audiences for genetic testing
- including prime prospect analysis
- and to identify what makes consumers more/less likely to undergo genetic testing
- Test around the information delivery
- What information will be conveyed, language and/or numbers used, etc.
- Follow-up based on results
- Price-elasticity
- How much would consumers be willing to pay for the test?
- When is it no longer "worth it"?
HCP-oriented research
- To determine charactersitics of strongest proponents of genetic testing
- Concept positioning for HCPs/How to engage HCPs as the consumer sees them as integral to the process
Presentation by Ryan Phelan
Presentation by Ryan Phelan
Founder and CEO
DNA Director
July 7th 2008
12:30pm-5:30PM
Ronald Reagan International Trade Center
Washington DC
Disclaimer: The findings and conclusions of this presentation are those of the authors and do not necessarily represent the views of ASPE or HHS.
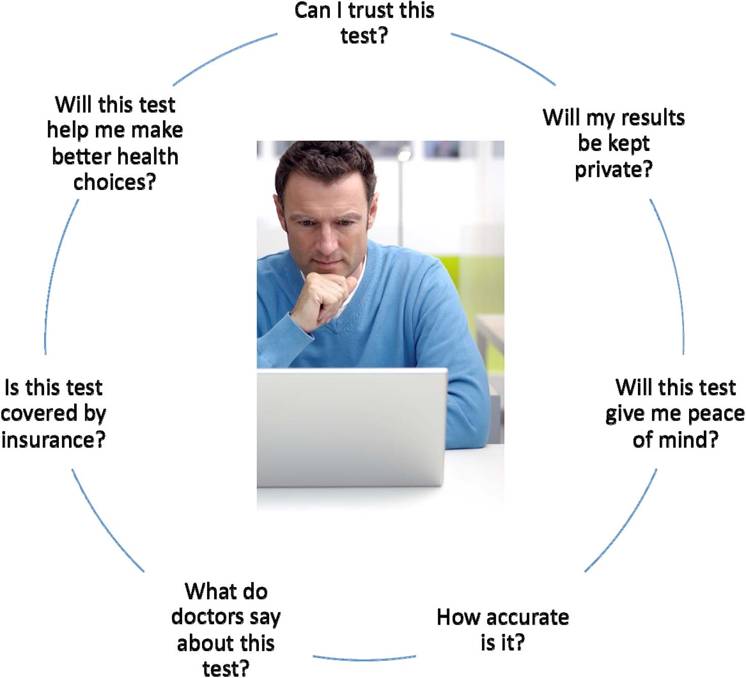
circle with text questions on it:"Can I trust this test?",(on top of circle, followed by these sentences in a counterclockwise direction); "Will my results be kept private?" ,(top right ); "will this test give me peace of mind?",(middle right); "How accurate is it?" ,(lower right);"What do doctors say about this test"?,(lower left);"Is this test covered by insurance?",(middle left);"Will this test help me make better health choices",(upper left).
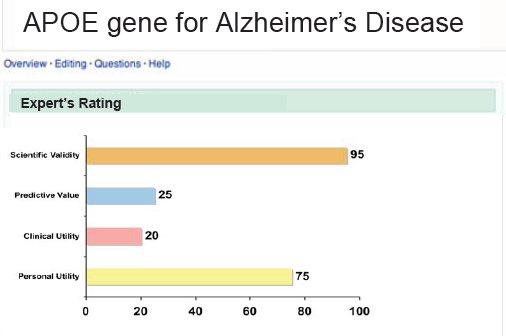
APO Gene for Alzheimers Disease(bar chart)- Experts Rating--Scientific Validity(95); Predictive Value(25); Clinical Utility(20); Personal Utility(75)

| Type of Genetic Tests | Examples | In-Person Evaluation | Physician Oversight | GC Phone Consult | Web Support |
|---|---|---|---|---|---|
| Genome-wide testing | SNP array | note1 | note 1 | note1 | note1 |
| Genetic Screening:carrier, risk assessment, drug response | Cystic Fibrosis Carrier | marked | note1 | marked | |
| Predictive Testing for serious health conditions | BRCA | marked | marked | marked | |
| Diagnostic Testing for Genetic Diseases | Huntingtons Disease | marked | marked |
Condition/ Situation Dependent
Presentation by Jeffrey Gulcher
Presentation by Jeffrey Gulcher, M.D., Ph.D
Chief Scientific Officer
deCode Genetics
July 7, 2008
12:30PM-5:30PM
Ronald Reagan International Trade Center
Washington, DC
Disclaimer: The findings and conclusions of this presentation are those of the authors and do not necessarily represent the views of ASPE or HHS.
Clinical Validation (deCode genetics)
- The genetic risk test for each disease is well-validated
- Each genetic marker has been replicated in 5 to 60 separate populations
- The realtive risk is derived from several thousands of patients and tens of thousands of controls - much larger than the data used to support most FDA - approved diagnostics and drugs
- Markers are shown to be independent and are therefore combined by multiplying the risks together using a standard, validated method
Useful Genetic risk tests for common disease- are we there yet?
- Heart attack -deCode MI (2 markers)
- 21% of the general population has 1.5 fold risk for early heart attack and 1.3 fold risk regardless of age of onset (comparable to risk conferred be LDL-cholesterol
- Prostate cancer-deCode proCa (8 markers)
- 10% of the general population has average 2 fold risk, 1% has 3 fold risk (lifetime risk 48% )
- Breast cancer - deCode BreastCa (8 markers)
- 5% of the general poulation has 2 fold risk, 1% 3 fold risk (lifetime risk of 36%)
- Type 2 Diabetes - deCode T2 (4 - 8 markers)
- 10% of prediabete patients have two fold risk for quickly progressing to type 2 diabetes (50% to 70 % absolute risk within 3 to4 years)
- Atrial fibrillation - deCode AF (2 markers)
- 25% of the general population has average two-fold risk of AF (1.5 to3.6 fold)
- Reveals that AF is a much greater cause for stroke than previoiusly recognized
- 150,000 stroke and TIAs per year may be due to undiagnosed AF - discharged on the wrong drug for secondary stroke prevention
- Focusing extra cardiac monitoring on test-positive patients may save Medicare Almost 1 Billion dollars after accounting for costs of tests
- Glaucoma - deCode Glaucoma (3 markers)
- 2.3 fold risk for more aggressive form of glaucoma leading to earlier blindness if unrecognized
A Case Study Demonstrating
Usefulness of a Genetic Risk Test
- 48 year old white male in apparent good health,
- His father diagnosed with localized prostate cancer ar age 70
- patient with low normal PSA at age 42
- deCode Prostate Cancer test results (8 markers combined)
- Relative risk= 1.88 fold compared to general poulation risk for white males
- Calculated lifetime risk= 1.88 x 16% =30%
- Modestly higher risk for aggressive vs non-aggressive disease
- High risk prompted early serum PSA test be primary care
- PSA was high normal at 2.5ng/ml
- High risk prompted referral to urologist
- High risk prompted urologist to perform ultasound-guided biopsy
- Positive for cancer in 3 out of 12 core biopsies - 15% volume
- Intermediate grade tumor (Gleason score of 6)
- Sugery after negative workup for metastasis -likely cure
